Allergy Asthma Immunol Res.
2014 Jan;6(1):22-26. 10.4168/aair.2014.6.1.22.
Effects of Methylprednisolone Pulse Therapy on Refractory Mycoplasma pneumoniae Pneumonia in Children
- Affiliations
-
- 1Department of Pediatrics, Chungnam National University School of Medicine, Daejeon, Korea. immlee@cnu.ac.kr
- KMID: 2166924
- DOI: http://doi.org/10.4168/aair.2014.6.1.22
Abstract
- PURPOSE
Mycoplasma pneumoniae (M. pneumoniae) is one of the most common causes of community-acquired pneumonia in children. The clinical course is typically self-limited and benign; however, rare cases of severe pneumonia can develop despite appropriate antibiotic therapy. We studied the effects of methylprednisolone pulse therapy on severe refractory M. pneumoniae pneumonia in children.
METHODS
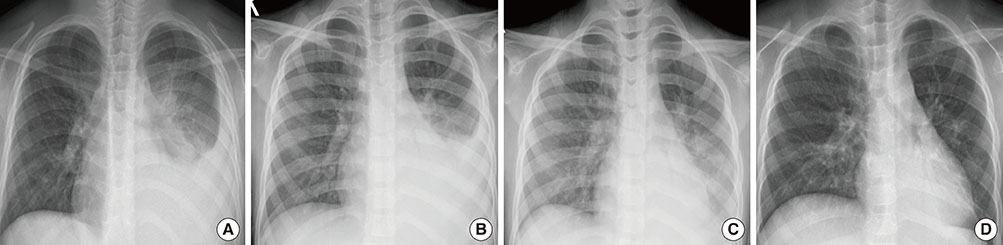
The clinical effects of methylprednisolone therapy were evaluated retrospectively in 12 children with severe refractory M. pneumoniae pneumonia, which was diagnosed serologically. All patients developed respiratory distress, high fever, and initial lobar pneumonic consolidation based on radiological findings. All clinical symptoms deteriorated despite appropriate antibiotic therapy. Thus, children were treated with intravenous methylprednisolone pulse therapy in addition to antibiotics.
RESULTS
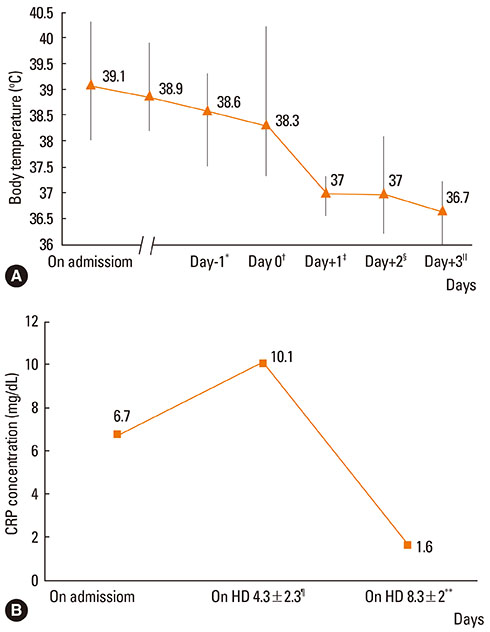
The average febrile period before admission was 4.9+/-1.7 days, and fever persisted in all children until steroid administration. Methylprednisolone pulse therapy (30 mg/kg) was given 5.4+/-2.5 days after admission. After methylprednisolone pulse therapy, clinical symptoms improved in all patients without adverse events. The fever subsided 0-2 h after initiation of corticosteroid therapy. The abnormal radiological findings resolved within 2.6+/-1.3 days, and the high C-reactive protein levels (6.7+/-5.9 mg/dL) on admission decreased to 1.3+/-1.7 mg/dL within 3.0+/-1.1 days after starting corticosteroid therapy.
CONCLUSIONS
Three-day methylprednisolone pulse therapy could be applied to treatment of refractory M. pneumoniae pneumonia despite appropriate antibiotic therapy and appeared to be efficacious and well-tolerated.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Early Additional Immune-Modulators for Mycoplasma pneumoniae Pneumonia in Children: An Observation Study
You-Sook Youn, Sung-Churl Lee, Jung-Woo Rhim, Myung-Seok Shin, Jin-Han Kang, Kyung-Yil Lee
Infect Chemother. 2014;46(4):239-247. doi: 10.3947/ic.2014.46.4.239.
Reference
-
1. Waites KB, Talkington DF. Mycoplasma pneumoniae and its role as a human pathogen. Clin Microbiol Rev. 2004; 17:697–728.2. Atkinson TP, Balish MF, Waites KB. Epidemiology, clinical manifestations, pathogenesis and laboratory detection of Mycoplasma pneumoniae infections. FEMS Microbiol Rev. 2008; 32:956–973.3. Heiskanen-Kosma T, Korppi M, Jokinen C, Kurki S, Heiskanen L, Juvonen H, Kallinen S, Stén M, Tarkiainen A, Rönnberg PR, Kleemola M, Mäkelä PH, Leinonen M. Etiology of childhood pneumonia: serologic results of a prospective, population-based study. Pediatr Infect Dis J. 1998; 17:986–991.4. Sinaniotis CA, Sinaniotis AC. Community-acquired pneumonia in children. Curr Opin Pulm Med. 2005; 11:218–225.5. Chan ED, Welsh CH. Fulminant Mycoplasma pneumoniae pneumonia. West J Med. 1995; 162:133–142.6. Lee KY. Pediatric respiratory infections by Mycoplasma pneumoniae. Expert Rev Anti Infect Ther. 2008; 6:509–521.7. Lee SH, Noh SM, Lee KY, Lee HS, Hong JH, Lee MH, Lee JS, Lee BC. Clinico-epidemiologic study of Mycoplasma pneumoniae pneumonia (1993 through 2003). Korean J Pediatr. 2005; 48:154–157.8. Morozumi M, Takahashi T, Ubukata K. Macrolide-resistant Mycoplasma pneumoniae: characteristics of isolates and clinical aspects of community-acquired pneumonia. J Infect Chemother. 2010; 16:78–86.9. Shimizu T, Kida Y, Kuwano K. Cytoadherence-dependent induction of inflammatory responses by Mycoplasma pneumoniae. Immunology. 2011; 133:51–61.10. Lu A, Wang L, Zhang X, Zhang M. Combined treatment for child refractory Mycoplasma pneumoniae pneumonia with ciprofloxacin and glucocorticoid. Pediatr Pulmonol. 2011; 46:1093–1097.11. Tamura A, Matsubara K, Tanaka T, Nigami H, Yura K, Fukaya T. Methylprednisolone pulse therapy for refractory Mycoplasma pneumoniae pneumonia in children. J Infect. 2008; 57:223–228.12. Lee KY, Lee HS, Hong JH, Lee MH, Lee JS, Burgner D, Lee BC. Role of prednisolone treatment in severe Mycoplasma pneumoniae pneumonia in children. Pediatr Pulmonol. 2006; 41:263–268.13. Ferwerda A, Moll HA, de Groot R. Respiratory tract infections by Mycoplasma pneumoniae in children: a review of diagnostic and therapeutic measures. Eur J Pediatr. 2001; 160:483–491.14. Radisic M, Torn A, Gutierrez P, Defranchi HA, Pardo P. Severe acute lung injury caused by Mycoplasma pneumoniae: potential role for steroid pulses in treatment. Clin Infect Dis. 2000; 31:1507–1511.15. Waites KB, Balish MF, Atkinson TP. New insights into the pathogenesis and detection of Mycoplasma pneumoniae infections. Future Microbiol. 2008; 3:635–648.16. Yang J, Hooper WC, Phillips DJ, Talkington DF. Cytokines in Mycoplasma pneumoniae infections. Cytokine Growth Factor Rev. 2004; 15:157–168.17. Denny FW, Taylor-Robinson D, Allison AC. The role of thymus-dependent immunity in Mycoplasma pulmonis infections of mice. J Med Microbiol. 1972; 5:327–336.18. Tagliabue C, Salvatore CM, Techasaensiri C, Mejias A, Torres JP, Katz K, Gomez AM, Esposito S, Principi N, Hardy RD. The impact of steroids given with macrolide therapy on experimental Mycoplasma pneumoniae respiratory infection. J Infect Dis. 2008; 198:1180–1188.19. De Pascale G, Bello G, Antonelli M. Steroids in severe pneumonia: a literature review. Minerva Anestesiol. 2011; 77:902–910.20. Rhen T, Cidlowski JA. Antiinflammatory action of glucocorticoids--new mechanisms for old drugs. N Engl J Med. 2005; 353:1711–1723.21. Niitu Y, Hasegawa S, Suetake T, Kubota H, Komatsu S, Horikawa M. Resistance of Mycoplasma pneumoniae to erythromycin and other antibiotics. J Pediatr. 1970; 76:438–443.22. Okazaki N, Narita M, Yamada S, Izumikawa K, Umetsu M, Kenri T, Sasaki Y, Arakawa Y, Sasaki T. Characteristics of macrolide-resistant Mycoplasma pneumoniae strains isolated from patients and induced with erythromycin in vitro. Microbiol Immunol. 2001; 45:617–620.23. Bébéar C, Pereyre S, Peuchant O. Mycoplasma pneumoniae: susceptibility and resistance to antibiotics. Future Microbiol. 2011; 6:423–431.24. Oh CE, Choi EH, Lee HJ. Detection of genetic mutations associated with macrolide resistance of Mycoplasma pneumoniae. Korean J Pediatr. 2010; 53:178–183.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A clinical study of mycoplasma pneumonia in children during recent 5 years
- Mycoplasma pneumoniae Pneumonia in Children
- Clinical Observation on Pneumonia due to Mycoplasma Pneumoniae in Children
- Clinical Study of Patients with Mycoplasma Pneumoniae Pneumonia in Children
- Clinical Consideration on Pneumonia caused by Mycoplasma Pneumoniae in Children