Cancer Res Treat.
2005 Oct;37(5):273-278.
Treatment Outcomes of Three-Dimensional Conformal Radiotherapy for Stage III Non-Small Cell Lung Cancer
- Affiliations
-
- 1Department of Radiation Oncology, Chungnam National University, Daejeon, Korea. mjcho@cnu.ac.kr
- 2Department of Internal Medicine, Chungnam National University, Daejeon, Korea.
- 3Department of Chest Surgery, Chungnam National University, Daejeon, Korea.
- 4Department of Cancer Research Institute, Chungnam National University, Daejeon, Korea.
Abstract
- PURPOSE
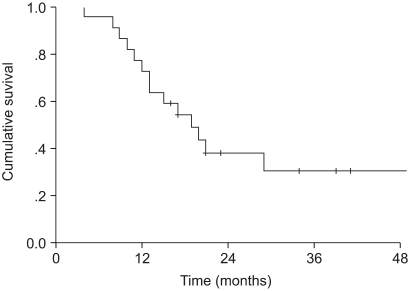
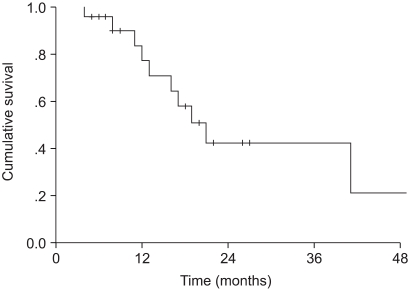
To evaluate the treatment outcomes of the three-dimensional conformal radiotherapy (3D-CRT), in conjunction with induction chemotherapy, for the treatment of stage III non-small cell lung cancer (NSCLC). MATERIALS AND METHODS: Between November 1998 and March 2003, 22 patients with histologically proven, clinical stage III NSCLC, treated with induction chemotherapy, followed by 3D-CRT, were retrospectively analyzed. There were 21 males (96%) and 1 female (4%), with a median age of 68.5 (range, 42~79). The clinical cancer stages were IIIA and IIIB in 41 and 59%, respectively. The histologies were squamous cell carcinoma, adenocarcinoma and others in 73, 18 and 9%, respectively. Twenty patients (91%) received induction chemotherapy before radiation therapy. The majority of the chemotherapy regimen consisted of cisplatin and gemcitabine. Radiation was delivered with conventional anteroposterior/ posteroanterior fields for 36 Gy, and then 3D-CRT was performed. The total radiation dose was 70.2 Gy. The median follow-up period was 17 months (range, 4~59 months). RESULTS: The median overall survival was 19 months. The two and four-year overall survival rates were 37.9 and 30.3%, respectively. The median progression-free survival was 21 months. The two and four-year progression-free survival rates were 42.1 and 21%, respectively. The prognostic factors for overall survival by a univariate analysis were age, histology and T stage (p<0.05). Acute radiation toxicities, as evaluated by the RTOG toxicity criteria, included two cases of grade 3 lung toxicity and one case of grade 2 esophagus toxicity. CONCLUSIONS: The radiation dose could be increased without a significant increment in the acute toxicities when using 3D-CRT. It also seems to be a safe, well- tolerated and effective treatment modality for stage III NSCLC.
MeSH Terms
Figure
Reference
-
1. Perez CA, Pajak TF, Rubin P, Simpson JR, Mohiuddin M, Brady LW, et al. Long-term observations of the patterns of failure in patients with unresectable non-oat cell carcinoma of the lung treated with definitive radiotherapy: a report by the Radiation Therapy Oncology Group. Cancer. 1987; 59:1874–1881. PMID: 3032394.2. Le Chevalier T, Arriagada R, Quoix E, Ruffie P, Martin M, Tarayre M, et al. Radiotherapy alone versus combined chemotherapy and radiotherapy in nonresectable non-small-cell lung cancer: first analysis of a randomized trial in 353 patients. J Natl Cancer Inst. 1991; 83:417–423. PMID: 1847977.
Article3. Perez C, Stanley K, Rubin P, Kramer S, Brady L, Perez-Tamayo R, et al. A prospective randomized study of various irradiation doses and fractionation schedules in the treatment of inoperable non-oat-cell carcinoma of the lung: preliminary report by the Radiation Therapy Oncology Group. Cancer. 1980; 45:2744–2753. PMID: 6991092.
Article4. Armstrong J, Raben A, Zelefsky M, Burt M, Leibel S, Burman C, et al. Promising survival with three-dimensional conformal radiation therapy for non small cell lung cancer. Radiother Oncol. 1997; 44:17–22. PMID: 9288852.5. Robertson JM, Ten Haken RK, Hazuka MB, Turrisi AT, Martel MK, Pu AT, et al. Dose escalation for non-small cell lung cancer using conformal radiation therapy. Int J Radiat Oncol Biol Phys. 1997; 37:1079–1085. PMID: 9169816.
Article6. Non-small Cell Lung Cancer Collaborative Group. Chemotherapy in non-small cell lung cancer: a meta-analysis using updated data on individual patients from 52 randomised clinical trials. BMJ. 1995; 311:899–909. PMID: 7580546.7. World Health Organization Offset Publication No.48. WHO handbook for reporting results of cancer treatments. 1979. Geneva (Switzerland):8. Cox JD, Azarnia N, Byhardt RW, Shin KH, Emami B, Pajak TF. A randomized phase I/II trial of hyperfractionated radiation therapy with total doses of 60.0 Gy to 79.2 Gy: possible survival benefit with 69.6 Gy in favorable patients with Radiation Therapy Oncology Group stage III non small cell lung carcinoma: report of Radiation Therapy Oncology Group 83-11. J Clin Oncol. 1990; 8:1543–1555. PMID: 2167952.9. Cox JD, Pajak TF, Asbell S, Russell AH, Pederson J, Byhardt RW, et al. Interruptions of high-dose radiation therapy decrease long-term survival of favorable patients with unresectable non-small cell carcinoma of the lung: Analysis of 1244 cases from 3 Radiation Therapy Oncology Group (RTOG) trials. Int J Radiat Oncol Biol Phys. 1993; 27:493–498. PMID: 8226140.
Article10. Vijayakumar S, Low N, Chen GT, Myrianthopoulos L, Culbert H, Chiru P, et al. Beam's Eye View-based photon radiotherapy I. Int Radiat Oncol Biol Phys. 1991; 21:1575–1586.
Article11. Myrianthopoulos LC, Chen GT, Vijayakumar S, Halpern HJ, Spelbring DR, Pelizzari CA. Beam's eye View volumetrics: an aid in rapid treatment plan development and evaluation. Int J Radiat Oncol Biol Phys. 1992; 23:367–375. PMID: 1587758.
Article12. Bradley JD, Ieumwananothachai N, Purdy JA, Wasserman TH, Lockett MA, Graham MV, et al. Gross tumor volume, critical prognostic factor in patients treated with three-dimensional conformal radiation therapy for non-small cell lung carcinoma. Int J Radiat Oncol Biol Phys. 2002; 52:49–57. PMID: 11777621.13. Hazuka MB, Turrisi AT 3rd, Lutz ST, Martel MK, Ten Haken RK, Strawderman M, et al. Results of high-dose thoracic irradiation incorporating beam's eye view display in non-small cell lung cancer: a retrospective multivariate analysis. Int J Radiat Oncol Biol Phys. 1993; 27:273–284. PMID: 8407401.
Article14. Dillman RO, Seagren SL, Propert KJ, Guerra J, Eaton WL, Perry MC, et al. A randomized trial of induction chemotherapy plus high-dose radiation versus radiation alone in stage III non-small-cell lung cancer. N Engl J Med. 1990; 323:940–945. PMID: 2169587.
Article15. Dillman RO, Herndon J, Seagren SL, Eaton WL Jr, Green MR. Improved survival in stage III non-small cell lung cancer: seven-year follow-up of Cancer and Leukemia Group B (CALGB) 8433 trial. J Natl Cancer Inst. 1996; 88:1210–1215. PMID: 8780630.16. Sim S, Rosenzweig KE, Schindelheim R, Ng KK, Leibel SA. Induction chemotherapy plus three-dimensional conformal radiation therapy in the definitive treatment of locally advanced non-small cell lung cancer. Int J Radiat Oncol Biol Phys. 2001; 51:660–665. PMID: 11597806.
Article17. Paek CW, Yun SY, Seo JH, Choi CW, Kim BS, Shin SW, et al. Concurrent chemoradiation therapy with cisplatin and oral etoposide for locally advanced non-small cell lung cancer. J Korean Cancer Assoc. 2000; 32:682–689.18. Curran WJ, Scott C, Langer C, Komaki R, Lee JS, Hauser S, et al. Phase III comparison of sequential vs. concurrent chemoradiation for patients with unresected stage III non-small cell lung cancer (NSCLC): initial report of Radiation Therapy Oncology Group (RTOG) 9410. Proc Am Soc Clin Oncol. 2000; 19:1891a.19. Rosenzweig KE, Sim SE, Mychalczak B, Braban LE, Schindelheim R, Leibel SA. Elective nodal irradiation in the treatment of non-small cell lung cancer with three-dimensional conformal radiation therapy. Int J Radiat Oncol Biol Phys. 2001; 50:681–685. PMID: 11395236.20. Senan S, Burgers S, Samson MJ, van Klaveren RJ, Oei SS, van Sornsen de Koste J, et al. Can elective nodal irradiation be omitted in stage III non-small-cell lung cancer? Analysis of recurrences in a phase II study of induction chemotherapy and involved-field radiotherapy. Int J Radiat Oncol Biol Phys. 2002; 54:999–1006. PMID: 12419425.
Article21. Sibley GS, Mundt AJ, Shapiro C, Jacobs R, Chen G, Weichselbaum R, et al. The treatment of stage III non small cell lung cancer using high dose conformal radiotherapy. Int J Radiat Oncol Biol Phys. 1995; 33:1001–1007. PMID: 7493826.22. Socinski MA, Morris DE, Halle JS, Moore DT, Hensing TA, Limentani SA, et al. Induction and concurrent chemotherapy with high-dose thoracic conformal radiation therapy in unresectable stage IIIA and IIIB non-small-cell lung cancer: a dose-escalation phase I trial. J Clin Oncol. 2004; 22:4341–4350. PMID: 15514375.
Article23. Graham MV, Purdy JA, Emami B, Harms W, Bosch W, Lockett MA, et al. Clinical dose-volume histogram analysis for pneumonitis after 3D treatment for non-small cell lung cancer (NSCLC). Int J Radiat Oncol Biol Phys. 1999; 45:323–329. PMID: 10487552.
Article