Cancer Res Treat.
2006 Dec;38(4):229-233.
The Effect of Pentoxifylline on Radiobiological Parameters in the Rat Radiation Myelopathy
- Affiliations
-
- 1Department of Radiation Oncology, Chungbuk National University College of Medicine, Cheongju, Korea. wdkim3@chungbuk.ac.kr
Abstract
- PURPOSE
There is great recent interest in the potential value of using pentoxifylline (3,7-dimethyl-1(5-oxyhexyl)- xanthine, PTX) as an inhibitor of radiation-induced late normal tissue damage. The effects of PTX on the radiobiological parameters (alpha/beta ratio, repair half time T1/2) of radiation myelopathy were studied in a rat model.
MATERIALS AND METHODS
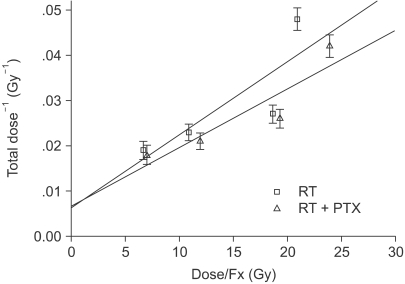
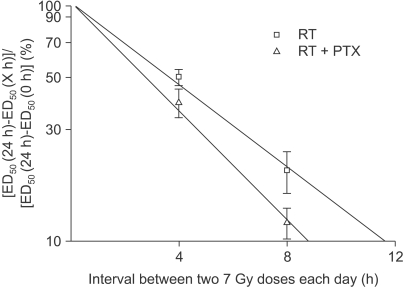
Anesthetized Sprague-Dawley rats received irradiation to 2 cm of their cervical spines with using a 6MV LINAC (dose rate: 3 Gy/min). Radiation was administered in single, two, four and eight fractions with a fraction interval of 24 h with or without PTX. PTX was added to the rats' distilled drinking water at a concentration of 2 g/L; the water was consumed ad libitum. After tabulation of the ED(50) (the estimated dose needed to produce 50% paralysis in a group of irradiated animals), alpha/beta could be estimated from the ratio of the slope to the intercept of the reciprocal-dose plot. Subsequently, the repair half time T(1/2) was obtained from the data of the experimental group that received a pair of 7 Gy fractions on each day, separated by intervals of 4 and 8 h.
RESULTS
The alpha values calculated for RT alone and RT+PTX were almost the same. We noticed that the beta value for the RT+PTX was lower than that for RT alone. So, the alpha/beta ratio for the RT+PTX was higher. The T(1/2) obtained from monoexponential model was 3.27 and 2.58 h for RT alone and RT+PTX, respectively.
CONCLUSION
PTX increased the alpha/beta ratio and it decreased the T(1/2) of radiation myelopathy, suggesting that a decreasing fractionation sensitivity occurred. This implies that PTX, which distinctly acts upon the bending region of the high dose, may be expected to protect the spinal cord with a larger fraction size.
Keyword
MeSH Terms
Figure
Reference
-
1. Schultheiss TE, Kun LE, Ang KK, Stephens LC. Radiation response of the central nervous system. Int J Radiat Oncol Biol Phys. 1995; 31:1093–1112. PMID: 7677836.
Article2. Hopewell JW, Campling D, Calvo W, Reinhold H, Wilkinson J, Yeung T. Vascular irradiation damage: its cellular basis and likely consequences. Br J Cancer. 1986; 53:181–191. PMID: 3485441.3. Dion MW, Hussey DH, Osborne JW. The effect of pentoxifylline on early and late radiation injury following fractionated irradiation in C3H mice. Int J Radiat Oncol Biol Phys. 1989; 17:101–107. PMID: 2745184.
Article4. Koh WJ, Stelzer KJ, Peterson LM, Staker BL, Ward WF, Russell KJ, et al. Effects of pentoxifylline on radiation-induced lung and skin toxicity in rats. Int J Radiat Oncol Biol Phys. 1995; 31:71–77. PMID: 7995770.5. Kim WD. The effect of pentoxifylline on rat spinal cord damage to fractionated irradiation. J Korean Cancer Assoc. 1998; 30:178–187.6. Douglas BG, Fowler JF. The effect of multiple small doses of x-rays on skin reactions in the mouse and a basic interpretation. Radiat Res. 1976; 66:401–426. PMID: 1265229.7. Litchfield JT, Wilcoxon F. A simplified method of evaluating dose-effect experiment. J Pharmacol Exp Ther. 1949; 96:99–113. PMID: 18152921.8. Landuyt W, Fowler J, Ruifrok A, Stuben G, Van der Kogel AJ, Van der Schuerena E. Kinetics of repair in the spinal cord of the rat. Radiother Oncol. 1997; 45:55–62. PMID: 9364632.
Article9. Leith JT, DeWyngaert JK, Glicksman AS. Radiation myelopathy in the rat: an interpretation of dose effect relationships. Int J Radiat Oncol Biol Phys. 1981; 7:1673–1677. PMID: 7333909.
Article10. Bentzen SM, Turesson I, Thames HD. Fractionation sensitivity and latency of telangiectasia after postmastectomy radiotherapy: a graded-response analysis. Radiother Oncol. 1990; 18:95–106. PMID: 2367694.
Article11. Ang KK, Van der Kogel AJ, Van Dam J, Van der Schueren E. The kinetics of repair of sublethal damage in the rat cervical spinal cord during fractionated irradiations. Radiother Oncol. 1984; 1:247–253. PMID: 6505261.12. Hopewell JW, Van den Aardweg GJ. Current concepts of dose fractionation in radiotherapy: normal tissue tolerance. Br J Radiol Suppl. 1988; 22:88–94. PMID: 3152506.13. Ang KK, Jiang GL, Guttenberger R, Thames HD, Stephens LC, Smith CD, et al. Impact of spinal cord repair kinetics on the practice of altered fractionation schedules. Radiother Oncol. 1992; 25:287–294. PMID: 1480775.
Article14. Rube CE, Wilfert F, Uthe D, Schmid KW, Knoop R, Willich N, et al. Modulation of radiation-induced tumour necrosis fac tor alpha (TNF-alpha) expression in the lung tissue by pentoxifylline. Radiother Oncol. 2002; 64:177–187. PMID: 12242128.15. Turesson I, Thames HD. Repair capacity and kinetics of human skin during fractionated radiotherapy: erythema, desquamation, and telangiectasia after 3 and 5 year's follow-up. Radiother Oncol. 1989; 15:169–188. PMID: 2762590.
Article16. Thompson MK, Tuma RF, Young WF. The effects of pentoxifylline on spinal cord blood flow after experimental spinal cord injury. J Assoc Acad Minor Phys. 1999; 10:23–26. PMID: 10826005.17. Laurent C, Pouget JP, Voisin P. Modulation of DNA damage by pentoxifylline and alpha-tocopherol in skin fibroblasts exposed to Gamma rays. Radiat Res. 2005; 164:63–72. PMID: 15966766.18. Danielsson A, Karlsson E, Delle U, Helou K, Mercke C. The biological effect of pentoxifylline on the survival of human head and neck cancer cells treated with continuous low and high dose-rate irradiation. J Cancer Res Clin Oncol. 2005; 131:459–467. PMID: 15776273.
Article19. Eley KW, Benedict SH, Chung TD, Kavanagh BD, Broaddus WC, Schmidt-Ullrich RK, et al. The effects of pentoxifylline on the survival of human glioma cells with continuous and intermittent stereotactic radiosurgery irradiation. Int J Radiat Oncol Biol Phys. 2002; 54:542–550. PMID: 12243834.
Article20. Theron T, Binder A, Verheye-Dua F, Bohm L. The role of G2-block abrogation, DNA double-strand break repair and apoptosis in the radiosensitization of melanoma and squamous cell carcinoma cell lines by pentoxifylline. Int J Radiat Biol. 2000; 76:1197–1208. PMID: 10993631.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effect of Pentoxifylline on Rat Spinal Cord Damage to Fractionated Irradiation
- Modulation of the action of proinflammatory cytokines on neutrophil function by pentoxifylline
- Protective Effect of Pentoxifylline and Ciprofloxacin on Dimethylnitrosamine-induced Hepatic Fibrosis in Rats
- Role of pentoxifylline in neonatal hypoxic ischaemic encephalopathy: a systematic review of animal studies
- The Effect of Pentoxifylline on Radiation-Induced Cardiac Injury in ICR Mice