Detection of Allergen Specific Antibodies From Nasal Secretion of Allergic Rhinitis Patients
- Affiliations
-
- 1Department of Allergy and Clinical Immunology, Ajou University School of Medicine, Suwon, Korea. hspark@ajou.ac.kr
- 2Division of Allergy, Choongmoo Hospital, Cheonan, Korea.
- KMID: 2165916
- DOI: http://doi.org/10.4168/aair.2016.8.4.329
Abstract
- PURPOSE
Allergic rhinitis (AR) is a common and increasing disease in which Dermatophagoides (D.) farinae is one of the most common causative allergens. The aims of this study were to confirm the presence of locally produced antibodies to D. farinae in nasal secretions between nasal provocation test (NPT)-positive and -negative groups of AR patients, to evaluate their relationships with the levels of inflammatory mediators, and to determine adaptive and innate immune responses in nasal mucosa.
METHODS
Sixty AR patients sensitive to house dust mites confirmed by skin prick test or serum specific IgE to D. farinae underwent NPT for D. farinae. Nasal packs were placed in both nasal cavities of the patients for 5 minutes to obtain nasal secretions after NPT. The levels of total IgE, specific IgE to D. farinae, eosinophil cationic protein (ECP), and tryptase in nasal secretions were detected by using ImmunoCAP. The levels of specific IgE, IgA, and secretory IgA antibodies to D. farinae in nasal secretions were measured by using ELISA. The levels of IL-8, VEGF, IL-25, and IL-33 were also measured by using ELISA.
RESULTS
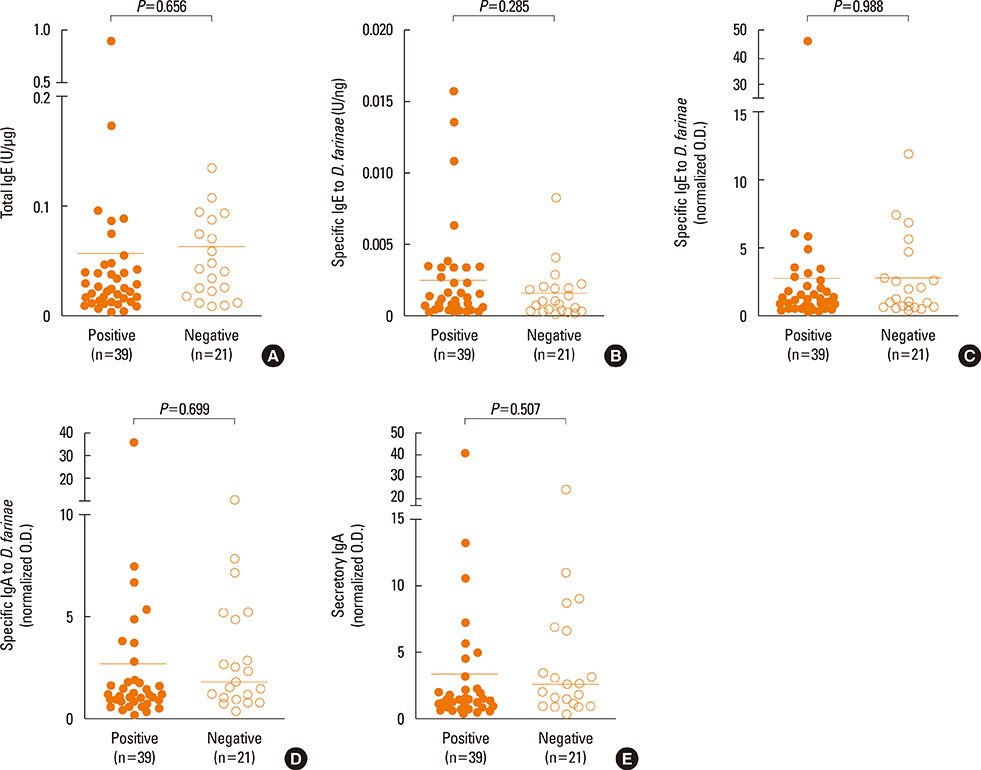
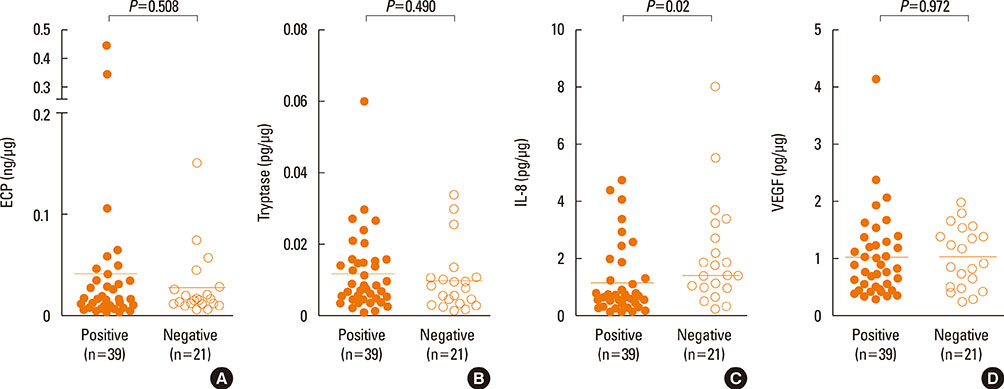
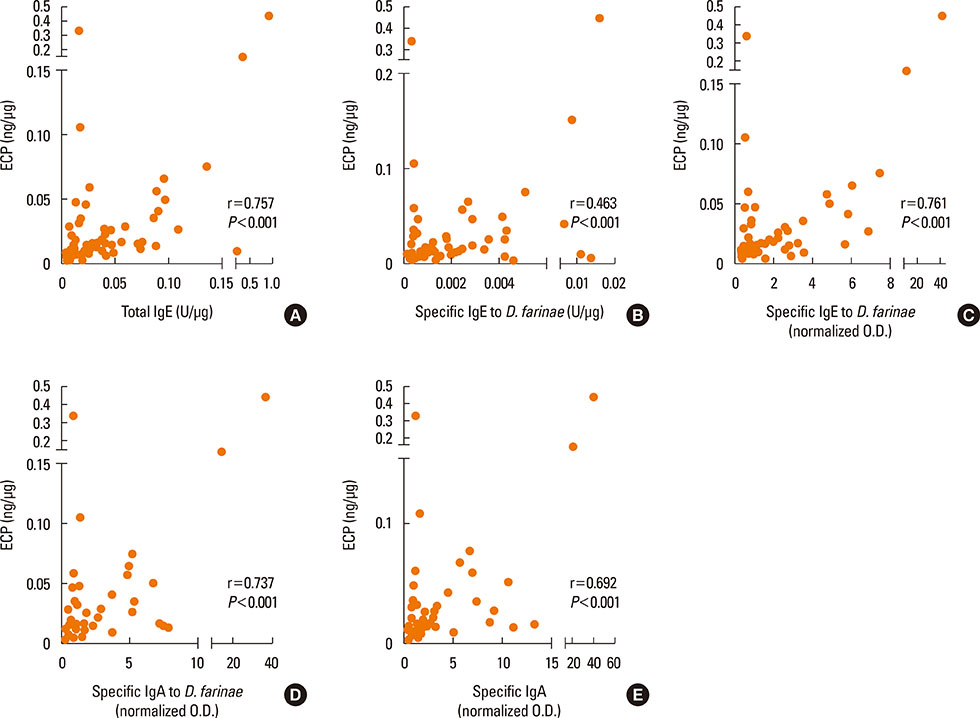
High levels of total IgE, specific IgE, specific IgA, and secretory IgA to D. farinae, as well as inflammatory mediators, such as ECP, IL-8, VEGF and tryptase, were detected in nasal secretions, although the differences were not statistically significant between the NPT-positive and NPT-negative groups. Levels of all immunoglobulins measured in this study significantly correlated with ECP, IL-8, and VEGF (P<0.05), but not with tryptase (P>0.05). IL-33 and IL-25 were also detected, and IL-25 level significantly correlated with IL-8 (r=0.625, P<0.001).
CONCLUSIONS
These findings confirmed the presence of locally produced specific antibodies, including D. farinae-specific IgE and IgA, in nasal secretions collected from D. farinae-sensitive AR patients in both the NPT-positive and NPT-negative groups, and close correlations were noted between antibodies and nasal inflammatory mediators, including such as ECP, IL-8 and VEGF, indicating that locally produced antibodies may be involved in the nasal inflammation of AR.
MeSH Terms
-
Allergens
Antibodies*
Enzyme-Linked Immunosorbent Assay
Eosinophil Cationic Protein
Humans
Immunity, Innate
Immunoglobulin A
Immunoglobulin A, Secretory
Immunoglobulin E
Immunoglobulins
Inflammation
Interleukin-8
Nasal Cavity
Nasal Mucosa
Nasal Provocation Tests
Pyroglyphidae
Rhinitis*
Skin
Tryptases
Vascular Endothelial Growth Factor A
Allergens
Antibodies
Eosinophil Cationic Protein
Immunoglobulin A
Immunoglobulin A, Secretory
Immunoglobulin E
Immunoglobulins
Interleukin-8
Tryptases
Vascular Endothelial Growth Factor A
Figure
Cited by 3 articles
-
Efficacy and Safety of Sublingual Immunotherapy in Elderly Rhinitis Patients Sensitized to House Dust Mites
Ji Hye Kim, Ji Ho Lee, Young-Min Ye, Jae-Hyun Lee, Jung Won Park, Gyu-Young Hur, Joo-Hee Kim, Hyn-Young Lee, Yoo Seob Shin, Eun-Mi Yang, Hae-Sim Park
Allergy Asthma Immunol Res. 2018;10(6):675-685. doi: 10.4168/aair.2018.10.6.675.Prevalence and Clinical Characteristics of Local Allergic Rhinitis to House Dust Mites
Chang-Gyu Jung, Ji-Ho Lee, Ga-Young Ban, Hae-Sim Park, Yoo Seob Shin
Yonsei Med J. 2017;58(5):1047-1050. doi: 10.3349/ymj.2017.58.5.1047.Usefulness of Allerkin House Dust Mite Extract for Nasal Provocation Testing
Kyu-Sung Kim, Tae Young Jang, Young Hyo Kim
Clin Exp Otorhinolaryngol. 2017;10(3):254-258. doi: 10.21053/ceo.2016.01137.
Reference
-
1. Bousquet J, Khaltaev N, Cruz AA, Denburg J, Fokkens WJ, Togias A, et al. Allergic Rhinitis and its Impact on Asthma (ARIA) 2008 update (in collaboration with the World Health Organization, GA(2)LEN and AllerGen). Allergy. 2008; 63:Suppl 86. 8–160.2. Greiner AN, Hellings PW, Rotiroti G, Scadding GK. Allergic rhinitis. Lancet. 2011; 378:2112–2122.3. Cibella F, Ferrante G, Cuttitta G, Bucchieri S, Melis MR, La Grutta S, et al. The burden of rhinitis and rhinoconjunctivitis in adolescents. Allergy Asthma Immunol Res. 2015; 7:44–50.4. Jo EJ, Kim MY, Lee SE, Lee SY, Kim MH, Song WJ, et al. Eosinophilic airway inflammation and airway hyperresponsiveness according to aeroallergen sensitization pattern in patients with lower airway symptoms. Allergy Asthma Immunol Res. 2014; 6:39–46.5. Ferrara N. VEGF: an update on biological and therapeutic aspects. Curr Opin Biotechnol. 2000; 11:617–624.6. Rydell-Törmänen K, Johnson JR, Fattouh R, Jordana M, Erjefält JS. Induction of vascular remodeling in the lung by chronic house dust mite exposure. Am J Respir Cell Mol Biol. 2008; 39:61–67.7. Shin SY, Choi SJ, Hur GY, Lee KH, Kim SW, Cho JS, et al. Local production of total IgE and specific antibodies to the house dust mite in adenoid tissue. Pediatr Allergy Immunol. 2009; 20:134–141.8. Gregory LG, Lloyd CM. Orchestrating house dust mite-associated allergy in the lung. Trends Immunol. 2011; 32:402–411.9. Reynolds JM, Angkasekwinai P, Dong C. IL-17 family member cytokines: regulation and function in innate immunity. Cytokine Growth Factor Rev. 2010; 21:413–423.10. Cayrol C, Girard JP. IL-33: an alarmin cytokine with crucial roles in innate immunity, inflammation and allergy. Curr Opin Immunol. 2014; 31:31–37.11. Saenz SA, Taylor BC, Artis D. Welcome to the neighborhood: epithelial cell-derived cytokines license innate and adaptive immune responses at mucosal sites. Immunol Rev. 2008; 226:172–190.12. Kariyawasam HH, Rotiroti G. Allergic rhinitis, chronic rhinosinusitis and asthma: unravelling a complex relationship. Curr Opin Otolaryngol Head Neck Surg. 2013; 21:79–86.13. Jankowski R, Persoons M, Foliguet B, Coffinet L, Thomas C, Verient-Montaut B. Eosinophil count in nasal secretions of subjects with and without nasal symptoms. Rhinology. 2000; 38:23–32.14. Riechelmann H, Deutschle T, Friemel E, Gross HJ, Bachem M. Biological markers in nasal secretions. Eur Respir J. 2003; 21:600–605.15. Lü FX, Esch RE. Novel nasal secretion collection method for the analysis of allergen specific antibodies and inflammatory biomarkers. J Immunol Methods. 2010; 356:6–17.16. Oh JH, Hur GY, Ye YM, Kim JE, Park K, Park HS. Correlation between specific IgA and eosinophil numbers in the lavage fluid of patients with perennial allergic rhinitis. Allergy Asthma Proc. 2008; 29:152–160.17. Kim YH, Jang TY. Proposed diagnostic standard using visual analogue scale and acoustic rhinometry in nasal provocation test in allergic patients. Auris Nasus Larynx. 2011; 38:340–346.18. Jang TY, Kim YH. Nasal provocation test is useful for discriminating allergic, nonallergic, and local allergic rhinitis. Am J Rhinol Allergy. 2015; 29:e100–e104.19. Watelet JB, Gevaert P, Holtappels G, Van Cauwenberge P, Bachert C. Collection of nasal secretions for immunological analysis. Eur Arch Otorhinolaryngol. 2004; 261:242–246.20. Jeong KY, Choi SY, Lee JH, Lee IY, Yong TS, Lee JS, et al. Standardization of house dust mite extracts in Korea. Allergy Asthma Immunol Res. 2012; 4:346–350.21. De Schryver E, Devuyst L, Derycke L, Dullaers M, Van Zele T, Bachert C, et al. Local immunoglobulin e in the nasal mucosa: clinical implications. Allergy Asthma Immunol Res. 2015; 7:321–331.22. Fagarasan S, Honjo T. Intestinal IgA synthesis: regulation of front-line body defences. Nat Rev Immunol. 2003; 3:63–72.23. Corthésy B. Role of secretory IgA in infection and maintenance of homeostasis. Autoimmun Rev. 2013; 12:661–665.24. Vazquez-Ortiz M, Pascal M, Juan M, Alsina L, Martín-Mateos MA, Plaza AM. Serum allergen-specific IgA is not associated with natural or induced tolerance to egg in children. Allergy. 2013; 68:1327–1332.25. Hupin C, Rombaux P, Bowen H, Gould H, Lecocq M, Pilette C. Downregulation of polymeric immunoglobulin receptor and secretory IgA antibodies in eosinophilic upper airway diseases. Allergy. 2013; 68:1589–1597.26. Bousquet J, Van Cauwenberge P, Bachert C, Canonica GW, Demoly P, Durham SR, et al. Requirements for medications commonly used in the treatment of allergic rhinitis. European Academy of Allergy and Clinical Immunology (EAACI), Allergic Rhinitis and its Impact on Asthma (ARIA). Allergy. 2003; 58:192–197.27. Wijburg OL, Uren TK, Simpfendorfer K, Johansen FE, Brandtzaeg P, Strugnell RA. Innate secretory antibodies protect against natural Salmonella typhimurium infection. J Exp Med. 2006; 203:21–26.28. Koyasu S, Moro K. Type 2 innate immune responses and the natural helper cell. Immunology. 2011; 132:475–481.29. Suh KS, Park HS, Nahm DH, Kim YK, Lee YM, Park K. Role of IgG, IgA, and IgE antibodies in nasal polyp tissue: their relationships with eosinophilic infiltration and degranulation. J Korean Med Sci. 2002; 17:375–380.30. Bystrom J, Patel SY, Amin K, Bishop-Bailey D. Dissecting the role of eosinophil cationic protein in upper airway disease. Curr Opin Allergy Clin Immunol. 2012; 12:18–23.31. Wedemeyer J, Tsai M, Galli SJ. Roles of mast cells and basophils in innate and acquired immunity. Curr Opin Immunol. 2000; 12:624–631.32. Williams CM, Galli SJ. The diverse potential effector and immunoregulatory roles of mast cells in allergic disease. J Allergy Clin Immunol. 2000; 105:847–859.33. Ohkubo K, Ikeda M, Pawankar R, Gotoh M, Yagi T, Okuda M. Mechanisms of IL-6, IL-8, and GM-CSF release in nasal secretions of allergic patients after nasal challenge. Rhinology. 1998; 36:156–161.34. Zuyderduyn S, Ninaber DK, Hiemstra PS, Rabe KF. The antimicrobial peptide LL-37 enhances IL-8 release by human airway smooth muscle cells. J Allergy Clin Immunol. 2006; 117:1328–1335.35. Simpson JL, Grissell TV, Douwes J, Scott RJ, Boyle MJ, Gibson PG. Innate immune activation in neutrophilic asthma and bronchiectasis. Thorax. 2007; 62:211–218.36. Dvorak HF, Brown LF, Detmar M, Dvorak AM. Vascular permeability factor/vascular endothelial growth factor, microvascular hyperpermeability, and angiogenesis. Am J Pathol. 1995; 146:1029–1039.37. Choi GS, Park HJ, Hur GY, Choi SJ, Shin SY, Ye YM, et al. Vascular endothelial growth factor in allergen-induced nasal inflammation. Clin Exp Allergy. 2009; 39:655–661.38. Fort MM, Cheung J, Yen D, Li J, Zurawski SM, Lo S, et al. IL-25 induces IL-4, IL-5, and IL-13 and Th2-associated pathologies in vivo. Immunity. 2001; 15:985–995.39. Hurst SD, Muchamuel T, Gorman DM, Gilbert JM, Clifford T, Kwan S, et al. New IL-17 family members promote Th1 or Th2 responses in the lung: in vivo function of the novel cytokine IL-25. J Immunol. 2002; 169:443–453.40. Calderón MA, Casale TB, Togias A, Bousquet J, Durham SR, Demoly P. Allergen-specific immunotherapy for respiratory allergies: from meta-analysis to registration and beyond. J Allergy Clin Immunol. 2011; 127:30–38.41. Peters AT, Spector S, Hsu J, Hamilos DL, Baroody FM, Chandra RK, et al. Diagnosis and management of rhinosinusitis: a practice parameter update. Ann Allergy Asthma Immunol. 2014; 113:347–385.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Diagnosis of Allergic Rhinitis
- Detection of Specific lgE Antibody from Nasal Secretion in Allergic Rhinitis with Negative Results of Skin Test and RAST on D.P
- Current Update of Local Allergic Rhinitis
- Acoustic Rhinometry in Nasal Provocation Test
- Diagnostic Value of Eosinophil Cationic Protein (ECP) in Nasal Secretion of Allergic Rhinitis