J Korean Med Sci.
2015 Aug;30(8):1136-1142. 10.3346/jkms.2015.30.8.1136.
Establishment of Reference Ranges for Prostate Volume and Annual Prostate Volume Change Rate in Korean Adult Men: Analyses of a Nationwide Screening Population
- Affiliations
-
- 1Department of Urology, Eulji University Hospital, Daejeon, Korea.
- 2Department of Urology, Kyung Hee University School of Medicine, Seoul, Korea. hllee61@hanmail.net
- 3Department of Family Medicine, Seoul National University Hospital, Seoul, Korea.
- 4Department of Urology, Boramae Medical Center, Seoul, Korea.
- 5Department of Urology, Chung-Ang University College of Medicine, Seoul, Korea.
- 6Department of Urology, Konkuk University Hospital, Seoul, Korea.
- KMID: 2164509
- DOI: http://doi.org/10.3346/jkms.2015.30.8.1136
Abstract
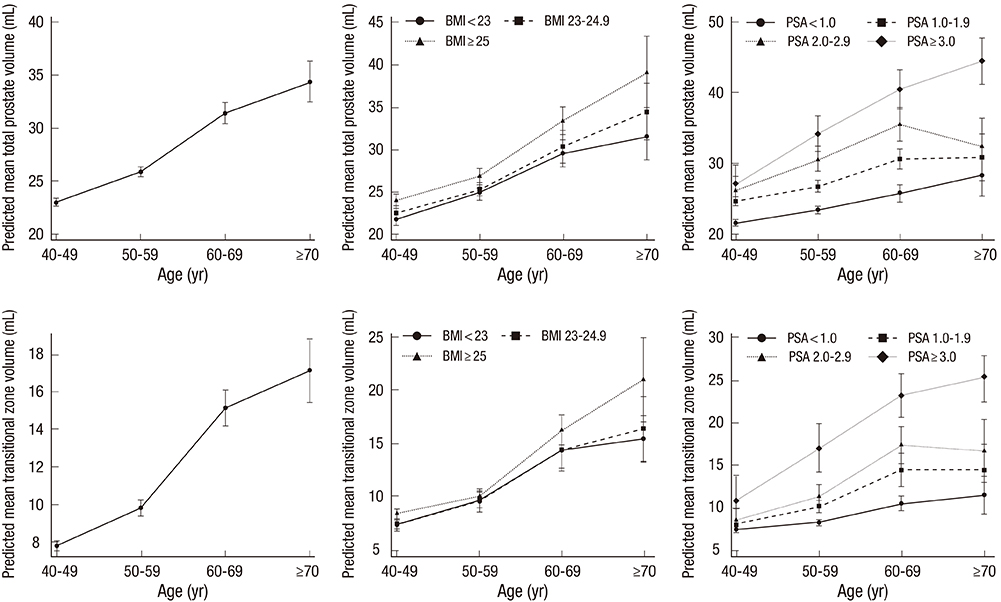
- We aimed to determine normal reference ranges for prostate volume (PV) and annual PV change rate in a Korean nationwide screening population. Data from men who underwent a routine health check-up were collected from 13 university hospitals. The cohort comprised men aged > or =40 yr who had undergone 2 or more serial transrectal ultrasonographies. Men with initial PV>100 mL; serum PSA level>10 ng/mL; PV reduction>20% compared with initial PV, or who had history of prostate cancer or prostate surgery, were excluded. Linear regression and mixed effects regression analyses were used to predict mean PV and longitudinal change in PV over time. A total of 2,967 men formed the study cohort. Age, body mass index (BMI), and serum prostate-specific antigen (PSA) level were found to be significant predictors of PV. A predicted PV table, with a 95% confidence interval (CIs), was developed after adjusting for these 3 variables. Annual PV change rate was 0.51 mL/year (95% CI, 0.47-0.55). Annual PV change rate according to age was 0.68 mL/year, 0.84 mL/year, 1.09 mL/year, and 0.50 mL/year for subjects in their 40s, 50s, 60s, and > or =70 yr, respectively. Predicted annual PV change rate differed depending on age, BMI, serum PSA level and baseline PV. From a nationwide screening database, we established age-, PSA-, and BMI-specific reference ranges for PV and annual PV change rate in Korean men. Our newly established reference ranges for PV and annual PV change rate will be valuable in interpreting PV data in Korean men.
Keyword
MeSH Terms
Figure
Reference
-
1. Walsh PC. Treatment of benign prostatic hyperplasia. N Engl J Med. 1996; 335:586–587.2. Lepor H. Alpha blockade for the treatment of benign prostatic hyperplasia. Urol Clin North Am. 1995; 22:375–386.3. McConnell JD. Benign prostatic hyperplasia. Hormonal treatment. Urol Clin North Am. 1995; 22:387–400.4. McConnell JD, Bruskewitz R, Walsh P, Andriole G, Lieber M, Holtgrewe HL, Albertsen P, Roehrborn CG, Nickel JC, Wang DZ, et al. The effect of finasteride on the risk of acute urinary retention and the need for surgical treatment among men with benign prostatic hyperplasia. Finasteride Long-Term Efficacy and Safety Study Group. N Engl J Med. 1998; 338:557–563.5. Marberger MJ, Andersen JT, Nickel JC, Malice MP, Gabriel M, Pappas F, Meehan A, Stoner E, Waldstreicher J. Prostate volume and serum prostate-specific antigen as predictors of acute urinary retention. Combined experience from three large multinational placebo-controlled trials. Eur Urol. 2000; 38:563–568.6. Roehrborn CG, McConnell J, Bonilla J, Rosenblatt S, Hudson PB, Malek GH, Schellhammer PF, Bruskewitz R, Matsumoto AM, Harrison LH, et al. Serum prostate specific antigen is a strong predictor of future prostate growth in men with benign prostatic hyperplasia. PROSCAR long-term efficacy and safety study. J Urol. 2000; 163:13–20.7. Lepor H. Pathophysiology of lower urinary tract symptoms in the aging male population. Rev Urol. 2005; 7:S3–S11.8. Vesely S, Knutson T, Damber JE, Dicuio M, Dahlstrand C. Relationship between age, prostate volume, prostate-specific antigen, symptom score and uroflowmetry in men with lower urinary tract symptoms. Scand J Urol Nephrol. 2003; 37:322–328.9. Loeb S, Kettermann A, Carter HB, Ferrucci L, Metter EJ, Walsh PC. Prostate volume changes over time: results from the Baltimore Longitudinal Study of Aging. J Urol. 2009; 182:1458–1462.10. Rhodes T, Girman CJ, Jacobsen SJ, Roberts RO, Guess HA, Lieber MM. Longitudinal prostate growth rates during 5 years in randomly selected community men 40 to 79 years old. J Urol. 1999; 161:1174–1179.11. Sarma AV, Wei JT, Jacobson DJ, Dunn RL, Roberts RO, Girman CJ, Lieber MM, Cooney KA, Schottenfeld D, Montie JE, et al. Olmsted County Study of Urinary Symptoms and Health Status. Flint Men's Health Study. Comparison of lower urinary tract symptom severity and associated bother between community-dwelling black and white men: the Olmsted County Study of Urinary Symptoms and Health Status and the Flint Men's Health Study. Urology. 2003; 61:1086–1091.12. Ochiai A, Fritsche HA, Babaian RJ. Influence of anthropometric measurements, age, and prostate volume on prostate-specific antigen levels in men with a low risk of prostate cancer. Urology. 2005; 66:819–823.13. Bosch JL, Tilling K, Bohnen AM, Bangma CH, Donovan JL. Establishing normal reference ranges for prostate volume change with age in the population-based Krimpen-study: prediction of future prostate volume in individual men. Prostate. 2007; 67:1816–1824.14. Joseph MA, Wei JT, Harlow SD, Cooney KA, Dunn RL, Jaffe CA, Montie JE, Schottenfeld D. Relationship of serum sex-steroid hormones and prostate volume in African American men. Prostate. 2002; 53:322–329.15. Bhindi B, Margel D, Trottier G, Hamilton RJ, Kulkarni GS, Hersey KM, Finelli A, Trachtenberg J, Zlotta A, Toi A, et al. Obesity is associated with larger prostate volume but not with worse urinary symptoms: analysis of a large multiethnic cohort. Urology. 2014; 83:81–87.16. Wallner LP, Morgenstern H, McGree ME, Jacobson DJ, St Sauver JL, Jacobsen SJ, Sarma AV. The effects of body mass index on changes in prostate-specific antigen levels and prostate volume over 15 years of follow-up: implications for prostate cancer detection. Cancer Epidemiol Biomarkers Prev. 2011; 20:501–508.17. Muller RL, Gerber L, Moreira DM, Andriole G Jr, Hamilton RJ, Fleshner N, Parsons JK, Freedland SJ. Obesity is associated with increased prostate growth and attenuated prostate volume reduction by dutasteride. Eur Urol. 2013; 63:1115–1121.18. Hoke G, Baker W, Barnswell C, Bennett J, Davis R, Mason T, Rayford W. Racial differences in pathogenetic mechanisms, prevalence, and progression of benign prostatic hyperplasia. Urology. 2006; 68:924–930.19. Fowler JE Jr, Bigler SA, Kilambi NK, Land SA. Relationships between prostate-specific antigen and prostate volume in black and white men with benign prostate biopsies. Urology. 1999; 53:1175–1178.20. Masumori N, Tsukamoto T, Kumamoto Y, Miyake H, Rhodes T, Girman CJ, Guess HA, Jacobsen SJ, Lieber MM. Japanese men have smaller prostate volumes but comparable urinary flow rates relative to American men: results of community based studies in 2 countries. J Urol. 1996; 155:1324–1327.21. Chung BH, Hong SJ, Cho JS, Seong DH. Relationship between serum prostate-specific antigen and prostate volume in Korean men with benign prostatic hyperplasia: a multicentre study. BJU Int. 2006; 97:742–746.22. Gupta A, Aragaki C, Gotoh M, Masumori N, Ohshima S, Tsukamoto T, Roehrborn CG. Relationship between prostate specific antigen and indexes of prostate volume in Japanese men. J Urol. 2005; 173:503–506.23. Chang YL, Lin AT, Chen KK, Chang YH, Wu HH, Kuo JY, Huang WJ, Lu SH, Hsu YS, Chung HJ, et al. Correlation between serum prostate specific antigen and prostate volume in Taiwanese men with biopsy proven benign prostatic hyperplasia. J Urol. 2006; 176:196–199.24. Terris MK, Stamey TA. Determination of prostate volume by transrectal ultrasound. J Urol. 1991; 145:984–987.25. Roehrborn CG, Siami P, Barkin J, Damião R, Major-Walker K, Nandy I, Morrill BB, Gagnier RP, Montorsi F. CombAT Study Group. The effects of combination therapy with dutasteride and tamsulosin on clinical outcomes in men with symptomatic benign prostatic hyperplasia: 4-year results from the CombAT study. Eur Urol. 2010; 57:123–131.26. Kanazawa M, Yoshiike N, Osaka T, Numba Y, Zimmet P, Inoue S. Criteria and classification of obesity in Japan and Asia-Oceania. World Rev Nutr Diet. 2005; 94:1–12.27. Laird NM, Ware JH. Random-effects models for longitudinal data. Biometrics. 1982; 38:963–974.28. Sarma AV, Jaffe CA, Schottenfeld D, Dunn R, Montie JE, Cooney KA, Wei JT. Insulin-like growth factor-1, insulin-like growth factor binding protein-3, and body mass index: clinical correlates of prostate volume among Black men. Urology. 2002; 59:362–367.29. McConnell JD, Roehrborn CG, Bautista OM, Andriole GL Jr, Dixon CM, Kusek JW, Lepor H, McVary KT, Nyberg LM Jr, Clarke HS, et al. Medical Therapy of Prostatic Symptoms (MTOPS) Research Group. The long-term effect of doxazosin, finasteride, and combination therapy on the clinical progression of benign prostatic hyperplasia. N Engl J Med. 2003; 349:2387–2398.30. Tsukamoto T, Masumori N, Rahman M, Crane MM. Change in International Prostate Symptom Score, prostrate-specific antigen and prostate volume in patients with benign prostatic hyperplasia followed longitudinally. Int J Urol. 2007; 14:321–324. discussion 5
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Age-specific Reference Ranges for Prostate Specific Antigen from a Health Center in Korea
- Reevaluation of the Reference Range of Prostate-specific Antigen in Korean Men
- Can Serum PSA Predict Prostate Volume in Men with Benign Prostatic Hyperplasia?
- Errors of Prostate Volume between Actual Models and Transrectal Ultrasonographic Measurement
- The Prostate Cancer Detection Rate on the Second Prostate Biopsy according to Prostate-Specific Antigen Trend