J Korean Med Sci.
2012 Apr;27(4):337-342. 10.3346/jkms.2012.27.4.337.
Prospective Controlled Protocol for Three Months Steroid Withdrawal with Tacrolimus, Basiliximab, and Mycophenolate Mofetil in Renal Transplant Recipients
- Affiliations
-
- 1Department of Surgery, Ajou University School of Medicine, Suwon, Korea. hartsol@ajou.ac.kr
- KMID: 2157891
- DOI: http://doi.org/10.3346/jkms.2012.27.4.337
Abstract
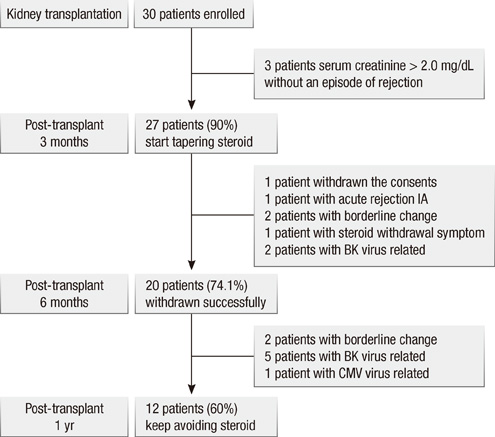
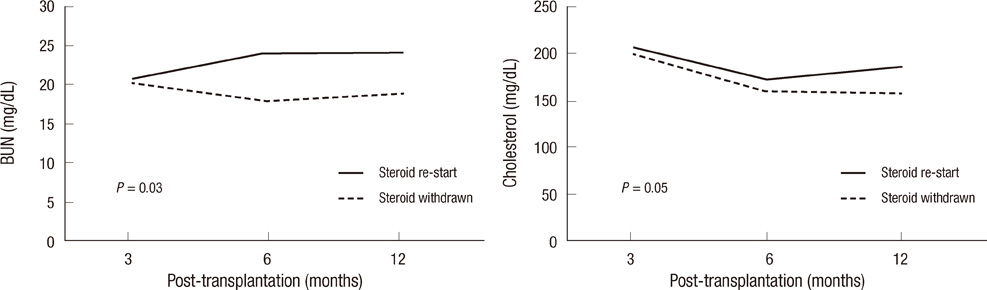
- During the past few years, new immunosuppressants, such as tacrolimus, mycophenolate mofetil (MMF) and basiliximab, have been shown to successfully decrease the incidence of acute rejection, possibly acting as potent substrates for safe steroid withdrawal. Therefore, clinical outcome of 3 months steroid withdrawal, while using the above immunosuppressants, was analyzed. Clinical trial registry No. was NCT 01550445. Thirty de novo renal transplant recipients were enrolled, and prednisolone was slowly withdrawn 3 months post-transplantation by 2.5 mg at every two weeks, until 8 weeks. During steroid withdrawal, 10 patients (30.0%) discontinued the protocol and they were maintained on steroid treatment. Among 20 steroid free patients, 8 patients (40.0%) re-started the steroid within 12 months post-transplantation. By the study endpoint, 12 (40%) recipients did not take steroid and survival of patients and grafts was 100%. In conclusion, in kidney transplant patients, 3 months steroid withdrawal while taking tacrolimus, basiliximab and mycophenolate mofetil was not associated with increased mortality or graft loss. Despite various causes of failure of steroid withdrawal during the follow-up period, it is a strategy well advised for kidney transplant recipients with regard to long-term steroid-related complications.
Keyword
MeSH Terms
-
Adult
Anti-Inflammatory Agents/*therapeutic use
Antibodies, Monoclonal/therapeutic use
Blood Urea Nitrogen
Cholesterol/blood
Creatinine/blood
Female
Graft Rejection/mortality/*prevention & control
Humans
Immunosuppressive Agents/*therapeutic use
*Kidney Transplantation
Male
Middle Aged
Mycophenolic Acid/analogs & derivatives/therapeutic use
Prednisolone/*therapeutic use
Prospective Studies
Recombinant Fusion Proteins/therapeutic use
Tacrolimus/therapeutic use
Figure
Reference
-
1. Veenstra DL, Best JH, Hornberger J, Sullivan SD, Hricik DE. Incidence and long-term cost of steroid-related side effects after renal transplantation. Am J Kidney Dis. 1999. 33:829–839.2. Ojo AO, Hanson JA, Wolfe RA, Leichtman AB, Agodoa LY, Port FK. Long-term survival in renal transplant recipients with graft function. Kidney Int. 2000. 57:307–313.3. Matas AJ. Steroid elimination-who, when, how? Transplant Proc. 2008. 40:S52–S56.4. Knight SR, Morris PJ. Steroid avoidance or withdrawal after renal transplantation increases the risk of acute rejection but decreases cardiovascular risk. A meta-analysis. Transplantation. 2010. 89:1–14.5. Pascual J, Galeano C, Royuela A, Zamora J. A systematic review on steroid withdrawal between 3 and 6 months after kidney transplantation. Transplantation. 2010. 90:343–349.6. Hricik DE. Steroid withdrawal in renal transplant recipients: pro point of view. Transplant Proc. 1998. 30:1380–1382.7. Wlodarczyk Z, Walaszewski J, Perner F, Vitko S, Ostrowski M, Bachleda P, Kokot F, Klinger M, Szenohradszky P, Studenik P, Navratil P, Asztalos L, Rutkowski B, Kalmar KN, Hickey D. Steroid withdrawal at 3 months after kidney transplantation: a comparison of two tacrolimus-based regimens. Transpl Int. 2005. 18:157–162.8. Kato Y, Tojimbara T, Iwadoh K, Koyama I, Nanmoku K, Kai K, Sannomiya A, Nakajima I, Fuchinoue S, Teraoka S. Early steroid withdrawal protocol with basiliximab, cyclosporine and mycophenolate mofetil in renal-transplant recipients. Int Immunopharmacol. 2006. 6:1984–1992.9. Lee YJ, Kim B, Lee JE, Kim YG, Kim DJ, Kim SJ, Joh JW, Oh HY, Huh W. Randomized trial of cyclosporine and tacrolimus therapy with steroid withdrawal in living-donor renal transplantation: 5-year follow-up. Transpl Int. 2010. 23:147–154.10. Prima V, Depoix C, Masselot B, Formstecher P, Lefebvre P. Alteration of the glucocorticoid receptor subcellular localization by non steroidal compounds. J Steroid Biochem Mol Biol. 2000. 72:1–12.11. Bemelman FJ, de Maar EF, Press RR, van Kan HJ, ten Berge IJ, Homan van der Heide JJ, de Fijter HW. Minimization of maintenance immunosuppression early after renal transplantation: an interim analysis. Transplantation. 2009. 88:421–428.12. Vera-Sempere FJ, Rubio L, Moreno-Baylach MJ, García A, Prieto M, Camañas A, Mayordomo F, Sánchez-Plumed J, Beneyto I, Ramos D, Zamora I, Simón J. Polymerase chain reaction detection of BK virus and monitoring of BK nephropathy in renal transplant recipients at the University Hospital La Fe. Transplant Proc. 2005. 37:3770–3773.13. Hollander AA, Hene RJ, Hermans J, van Es LA, van der Woude FJ. Late prednisone withdrawal in cyclosporine-treated kidney transplant patients: a randomized study. J Am Soc Nephrol. 1997. 8:294–301.14. Matl I, Lácha J, Lodererová A, Símová M, Teplan V, Lánská V, Vítko S. Withdrawal of steroids from triple-drug therapy in kidney transplant patients. Nephrol Dial Transplant. 2000. 15:1041–1045.15. Kupin W, Venkat KK, Goggins M, Douzdjian V, Escobar F 3rd, Mozes M, Abouljoud M. Improved outcome of steroid withdrawal in mycophenolate mofetil-treated primary cadaveric renal transplant recipients. Transplant Proc. 1999. 31:1131–1132.16. Feinfeld DA, Bargouthi H, Niaz Q, Carvounis CP. Massive and disproportionate elevation of blood urea nitrogen in acute azotemia. Int Urol Nephrol. 2002. 34:143–145.17. ter Meulen CG, van Riemsdijk I, Hené RJ, Christiaans MH, Borm GF, van Gelder T, Hilbrands LB, Weimar W, Hoitsma AJ. Steroid-withdrawal at 3 days after renal transplantation with anti-IL-2 receptor alpha therapy: a prospective, randomized, multicenter study. Am J Transplant. 2004. 4:803–810.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Efficacy and Outcome of Reduced Dose of Tacrolimus in Renal Transplantation
- Randomized Trial of Tacrolimus Versus Cyclosporine in Steroid Withdrawal in Living Donor Renal Transplant Recipients
- Safety and Efficacy of Early Steroid Withdrawal with Thymoglobuline Induction in Kidney Transplantation
- Safety and metabolic advantages of steroid withdrawal after 6-months post-transplant in de novo kidney transplantation: 1-year prospective cohort study
- Efficacy of early steroid withdrawal using everolimus in de novo kidney transplantation: a single center cohort study