Korean Guidelines for the Diagnosis and Management of Dry Eye: Development and Validation of Clinical Efficacy
- Affiliations
-
- 1Department of Ophthalmology, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Korea.
- 2Department of Ophthalmology, Korea University College of Medicine, Seoul, Korea. hyomkim@kumc.or.kr
- 3Department of Ophthalmology, Inje University College of Medicine, Goyang, Korea.
- 4Department of Ophthalmology, Sungkyunkwan University College of Medicine, Seoul, Korea.
- 5Department of Clinical Epidemiology and Biostatistics, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- KMID: 2133317
- DOI: http://doi.org/10.3341/kjo.2014.28.3.197
Abstract
- PURPOSE
To evaluate the clinical efficacy of newly developed guidelines for the diagnosis and management of dry eye.
METHODS
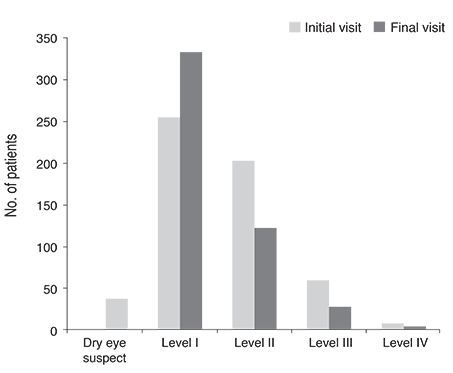
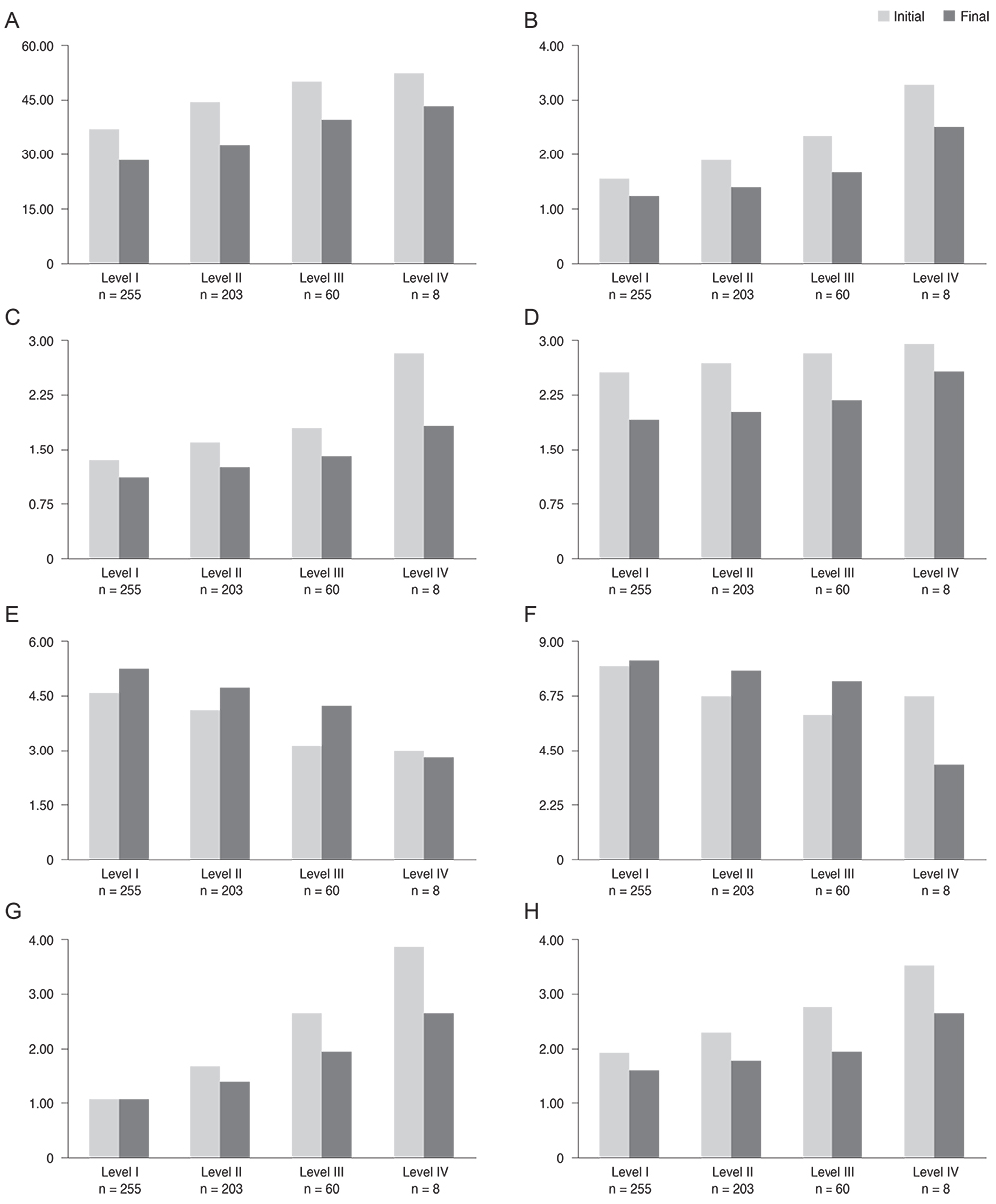
This retrospective, multi-center, non-randomized, observational study included a total of 1,612 patients with dry eye disease who initially visited the clinics from March 2010 to August 2010. Korean guidelines for the diagnosis and management of dry eye were newly developed from concise, expert-consensus recommendations. Severity levels at initial and final visits were determined using the guidelines in patients with 90 +/- 7 days of follow-up visits (n = 526). Groups with different clinical outcomes were compared with respect to clinical parameters, treatment modalities, and guideline compliance. Main outcome measures were ocular and visual symptoms, ocular surface disease index, global assessment by patient and physician, tear film break-up time, Schirmer-1 test score, ocular surface staining score at initial and final visits, clinical outcome after three months of treatment, and guideline compliance.
RESULTS
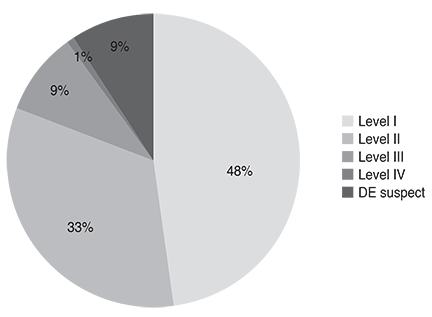
Severity level was reduced in 47.37% of patients treated as recommended by the guidelines. Younger age (odd ratio [OR], 0.984; p = 0.044), higher severity level at initial visit, compliance to treatment recommendation (OR, 1.832; p = 0.047), and use of topical cyclosporine (OR, 1.838; p = 0.011) were significantly associated with improved clinical outcomes.
CONCLUSIONS
Korean guidelines for the diagnosis and management of dry eye can be used as a valid and effective tool for the treatment of dry eye disease.
Keyword
MeSH Terms
-
Adult
Aged
Aged, 80 and over
Diagnostic Techniques, Ophthalmological/*standards
*Disease Management
Dry Eye Syndromes/*diagnosis/*drug therapy/epidemiology
Female
Follow-Up Studies
Humans
Immunosuppressive Agents/*administration & dosage
Male
Middle Aged
Morbidity/trends
Ophthalmic Solutions
*Practice Guidelines as Topic
Republic of Korea/epidemiology
Retrospective Studies
Young Adult
Immunosuppressive Agents
Ophthalmic Solutions
Figure
Cited by 11 articles
-
Effect of Intense Regulated Pulse Light on Thickness of Tear Film Lipid Layer and Dry Eye Syndrome
Tae Young Gil, Gi Hyun Bae, Joo Young Kwag, Joo Hyun, Jin Seok Choi, Kyu Hong Pak, Sung Kun Chung
J Korean Ophthalmol Soc. 2018;59(12):1103-1107. doi: 10.3341/jkos.2018.59.12.1103.Lid Wiper Epitheliopathy in Patients with Dry Eye Refractory to Conventional Medical Treatment
Jungah Huh, In Ho Woo, Youngsub Eom, Su-Yeon Kang, Hyo Myung Kim, Jong-Suk Song
J Korean Ophthalmol Soc. 2018;59(8):718-723. doi: 10.3341/jkos.2018.59.8.718.Clinical Significance of Computerized Videokeratoscopic Indices for Dry Eye
Jong-Ha Lee, Min-Hwan Kim, Byung-Yi Ko
J Korean Ophthalmol Soc. 2019;60(7):627-634. doi: 10.3341/jkos.2019.60.7.627.Evaluation of the Protective Effect of an Ophthalmic Viscosurgical Device on the Ocular Surface in Dry Eye Patients during Cataract Surgery
Do Yeh Yoon, Joo Hyun Kim, Hyun Sun Jeon, Hee Eun Jeon, Sang Beom Han, Joon Young Hyon
Korean J Ophthalmol. 2019;33(5):467-474. doi: 10.3341/kjo.2019.0060.Anterior segment eye diseases associated with rheumatic diseases
Joon Young Hyon
J Korean Med Assoc. 2016;59(1):45-51. doi: 10.5124/jkma.2016.59.1.45.The Effect of Topical Cyclosporine 0.05% on Tear Osmolarity for Dry Eye Syndrome
Hyunseung Kang, San Seong, Chul Myong Choe, Se Kyung Kim, Tae Hoon Choi
J Korean Ophthalmol Soc. 2015;56(2):174-179. doi: 10.3341/jkos.2015.56.2.174.Therapeutic Effects of 0.03% Tacrolimus Eye Drops for Chronic Ocular Graft-Versus-Host Disease
Soon Il Choi, So Hyang Chung
J Korean Ophthalmol Soc. 2015;56(10):1505-1510. doi: 10.3341/jkos.2015.56.10.1505.Tear Meniscus Evaluation Using Optical Coherence Tomography in Dry Eye Patients
Na Yeon Jung, Ji Won Baek, Seong Joo Shin, Sung Kun Chung
J Korean Ophthalmol Soc. 2015;56(3):323-330. doi: 10.3341/jkos.2015.56.3.323.Prevalence and Risk Factors of Dry Eye Disease after Refractive Surgery
Dae Yong Son, Sungsoon Hwang, Joo Hyun, Dong Hui Lim, Eui Sang Chung, Tae-Young Chung
J Korean Ophthalmol Soc. 2017;58(7):782-787. doi: 10.3341/jkos.2017.58.7.782.Diagnosis and treatment of dry eye syndrome
Eun Chul Kim
J Korean Med Assoc. 2018;61(6):352-364. doi: 10.5124/jkma.2018.61.6.352.Ophthalmologic manifestations in patients with inflammatory bowel disease
Hye Jin Lee, Hyun Joo Song, Jin Ho Jeong, Heung Up Kim, Sun-Jin Boo, Soo-Young Na
Intest Res. 2017;15(3):380-387. doi: 10.5217/ir.2017.15.3.380.
Reference
-
1. Lemp MA. Report of the National Eye Institute/Industry workshop on clinical trials in dry eyes. CLAO J. 1995; 21:221–232.2. Behrens A, Doyle JJ, Stern L, et al. Dysfunctional tear syndrome: a Delphi approach to treatment recommendations. Cornea. 2006; 25:900–907.3. The definition and classification of dry eye disease: report of the Definition and Classification Subcommittee of the International Dry Eye WorkShop (2007). Ocul Surf. 2007; 5:75–92.4. Methodologies to diagnose and monitor dry eye disease: report of the Diagnostic Methodology Subcommittee of the International Dry Eye WorkShop (2007). Ocul Surf. 2007; 5:108–152.5. Han SB, Hyon JY, Woo SJ, et al. Prevalence of dry eye disease in an elderly Korean population. Arch Ophthalmol. 2011; 129:633–638.6. The epidemiology of dry eye disease: report of the Epidemiology Subcommittee of the International Dry Eye Work-Shop (2007). Ocul Surf. 2007; 5:93–107.7. Nichols KK, Nichols JJ, Mitchell GL. The lack of association between signs and symptoms in patients with dry eye disease. Cornea. 2004; 23:762–770.8. Nichols KK, Smith JA. Association of clinical diagnostic tests and dry eye surveys: the NEI-VFQ-25 and the OSDI. Adv Exp Med Biol. 2002; 506(Pt B):1177–1181.9. Bron AJ, Evans VE, Smith JA. Grading of corneal and conjunctival staining in the context of other dry eye tests. Cornea. 2003; 22:640–650.10. Schiffman RM, Christianson MD, Jacobsen G, et al. Reliability and validity of the Ocular Surface Disease Index. Arch Ophthalmol. 2000; 118:615–621.11. Nelson JD, Shimazaki J, Benitez-del-Castillo JM, et al. The international workshop on meibomian gland dysfunction: report of the definition and classification subcommittee. Invest Ophthalmol Vis Sci. 2011; 52:1930–1937.12. Wilson SE, Stulting RD. Agreement of physician treatment practices with the international task force guidelines for diagnosis and treatment of dry eye disease. Cornea. 2007; 26:284–289.13. Perry HD, Solomon R, Donnenfeld ED, et al. Evaluation of topical cyclosporine for the treatment of dry eye disease. Arch Ophthalmol. 2008; 126:1046–1050.14. Management and therapy of dry eye disease: report of the Management and Therapy Subcommittee of the International Dry Eye WorkShop (2007). Ocul Surf. 2007; 5:163–178.15. Baiza-Duran L, Medrano-Palafox J, Hernandez-Quintela E, et al. A comparative clinical trial of the efficacy of two different aqueous solutions of cyclosporine for the treatment of moderate-to-severe dry eye syndrome. Br J Ophthalmol. 2010; 94:1312–1315.16. Barber LD, Pflugfelder SC, Tauber J, Foulks GN. Phase III safety evaluation of cyclosporine 0.1% ophthalmic emulsion administered twice daily to dry eye disease patients for up to 3 years. Ophthalmology. 2005; 112:1790–1794.17. Sall K, Stevenson OD, Mundorf TK, Reis BL. CsA Phase 3 Study Group. Two multicenter, randomized studies of the efficacy and safety of cyclosporine ophthalmic emulsion in moderate to severe dry eye disease. Ophthalmology. 2000; 107:631–639.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- New Korean Guidelines for the Diagnosis and Management of Dry Eye Disease
- Recent treatment of dry eye
- Dry Eye Assessment of Patients Undergoing Endoscopic Dacryocystorhinostomy for Nasolacrimal Duct Obstruction Combined with Dry Eye Syndrome
- Points to consider when developing drugs for dry eye syndrome
- The proportional Changes of Tear Proteins in the Dry Eye Patients