Allergy Asthma Immunol Res.
2015 Nov;7(6):573-582. 10.4168/aair.2015.7.6.573.
Prenatal Particulate Matter/Tobacco Smoke Increases Infants' Respiratory Infections: COCOA Study
- Affiliations
-
- 1Department of Pediatrics, Hallym Sacred Heart Hospital, Hallym University College of Medicine, Anyang, Korea.
- 2Department of Environmental Health, University of Cincinnati College of Medicine, Cincinnati, Ohio, USA.
- 3Department of Pediatrics, Inje University Sanggye Paik Hospital, Seoul, Korea.
- 4Institute of Environmental and Industrial Medicine, Hanyang University, Seoul, Korea.
- 5Department of Pediatrics, Childhood Asthma Atopy Center, Environmental Health Center, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. sjhong@amc.seoul.kr
- 6Asan Institute for Life Science, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 7Department of Pediatrics, Bundang CHA Medical Center, CHA University School of Medicine, Seongnam, Korea.
- 8Department of Pediatrics, Pusan National University Yangsan Hospital, Yangsan, Korea.
- 9Department of Pediatrics, Korea Cancer Center Hospital, Seoul, Korea.
- 10Department of Pediatrics, Seoul National University Bundang Hospital, Seungnam, Korea.
- 11Department of Pediatrics, College of Medicine, Korea University, Seoul, Korea.
- 12Department of Pediatrics, National Health Insurance Corporation Ilsan Hospital, Goyang, Korea.
- 13Department of Pediatrics and the Allergy and Respiratory Research Laboratory, Inje University Seoul Paik Hospital, Seoul, Korea.
- 14Department of Pediatrics, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 15Department of Pediatrics, Ajou University School of Medicine, Suwon, Korea.
- 16Department of Pediatrics, Soonchunhyang University College of Medicine, Seoul, Korea.
- 17Department of Pediatrics, Seoul National University College of Medicine, Seoul, Korea.
- 18Department of Pediatrics, Gangnam CHA Medical Center, CHA University College of Medicine, Seoul, Korea.
- 19Department of Pediatrics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 20Department of Pediatrics, Yonsei University College of Medicine, Seoul, Korea.
- 21Division of Allergy and Chronic Respiratory diseases, Center for of Biomedical Sciences, Korea National Institute of Health, Korea Centers for Disease Control and Prevention, Osong, Korea.
- KMID: 2130264
- DOI: http://doi.org/10.4168/aair.2015.7.6.573
Abstract
- PURPOSE
To investigate whether prenatal exposure to indoor fine particulate matter (PM2.5) and environmental tobacco smoke (ETS) affects susceptibility to respiratory tract infections (RTIs) in infancy, to compare their effects between prenatal and postnatal exposure, and to determine whether genetic factors modify these environmental effects.
METHODS
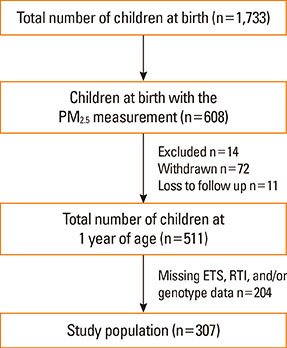
The study population consisted of 307 birth cohort infants. A diagnosis of RTIs was based on parental report of a physician's diagnosis. Indoor PM2.5 and ETS levels were measured during pregnancy and infancy. TaqMan was used for genotyping of nuclear factor erythroid 2-related factor (Nrf2) (rs6726395), glutathione-S-transferase-pi (GSTP) 1 (rs1695), and glutathione-S-transferase-mu (GSTM) 1. Microarrays were used for genome-wide methylation analysis.
RESULTS
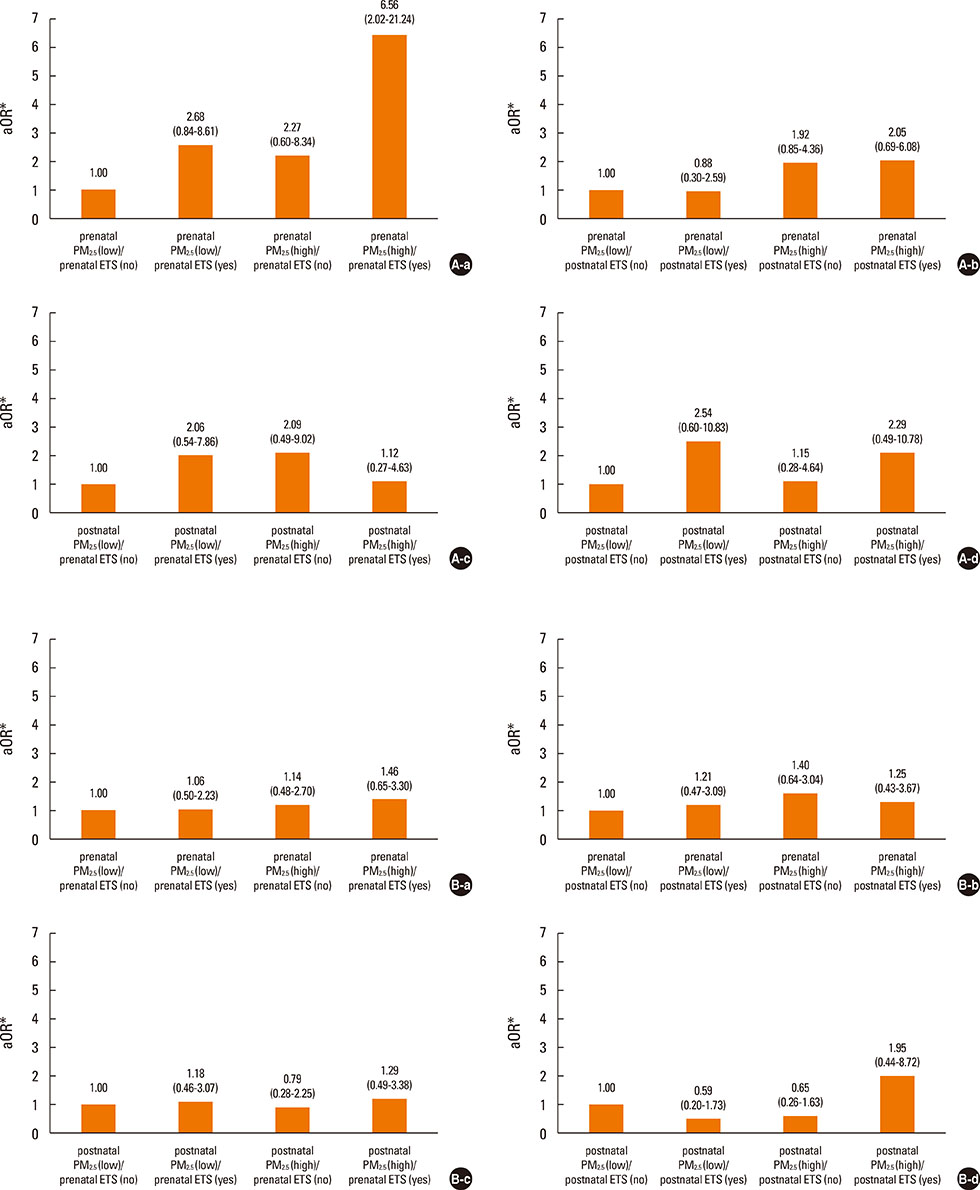
Prenatal exposure to indoor PM2.5 increased the susceptibility of lower RTIs (LRTIs) in infancy (adjusted odds ratio [aOR]=2.11). In terms of combined exposure to both indoor PM2.5 and ETS, prenatal exposure to both pollutants increased susceptibility to LRTIs (aOR=6.56); however, this association was not found for postnatal exposure. The Nrf2 GG (aOR=23.69), GSTM1 null (aOR=8.18), and GSTP1 AG or GG (aOR=7.37) genotypes increased the combined LRTIs-promoting effects of prenatal exposure to the 2 indoor pollutants. Such effects of prenatal indoor PM2.5 and ETS exposure were not found for upper RTIs.
CONCLUSIONS
Prenatal exposure to both indoor PM2.5 and ETS may increase susceptibility to LRTIs. This effect can be modified by polymorphisms in reactive oxygen species-related genes.
Keyword
MeSH Terms
Figure
Reference
-
1. Hasheminassab S, Daher N, Shafer MM, Schauer JJ, Delfino RJ, Sioutas C. Chemical characterization and source apportionment of indoor and outdoor fine particulate matter (PM(2.5)) in retirement communities of the Los Angeles Basin. Sci Total Environ. 2014; 490:528–537.2. Clougherty JE, Houseman EA, Levy JI. Source apportionment of indoor residential fine particulate matter using land use regression and constrained factor analysis. Indoor Air. 2011; 21:53–66.3. Wallace LA, Mitchell H, O'Connor GT, Neas L, Lippmann M, Kattan M, et al. Particle concentrations in inner-city homes of children with asthma: the effect of smoking, cooking, and outdoor pollution. Environ Health Perspect. 2003; 111:1265–1272.4. Guarnieri M, Balmes JR. Outdoor air pollution and asthma. Lancet. 2014; 383:1581–1592.5. Rosa MJ, Jung KH, Perzanowski MS, Kelvin EA, Darling KW, Camann DE, et al. Prenatal exposure to polycyclic aromatic hydrocarbons, environmental tobacco smoke and asthma. Respir Med. 2011; 105:869–876.6. Miller RL, Garfinkel R, Horton M, Camann D, Perera FP, Whyatt RM, et al. Polycyclic aromatic hydrocarbons, environmental tobacco smoke, and respiratory symptoms in an inner-city birth cohort. Chest. 2004; 126:1071–1078.7. Sonnenschein-van der Voort AM, de Kluizenaar Y, Jaddoe VW, Gabriele C, Raat H, Moll HA, et al. Air pollution, fetal and infant tobacco smoke exposure, and wheezing in preschool children: a population-based prospective birth cohort. Environ Health. 2012; 11:91.8. Rabinovitch N, Silveira L, Gelfand EW, Strand M. The response of children with asthma to ambient particulate is modified by tobacco smoke exposure. Am J Respir Crit Care Med. 2011; 184:1350–1357.9. Nicolai T, Carr D, Weiland SK, Duhme H, von Ehrenstein O, Wagner C, et al. Urban traffic and pollutant exposure related to respiratory outcomes and atopy in a large sample of children. Eur Respir J. 2003; 21:956–963.10. Jedrychowski W, Bendkowska I, Flak E, Penar A, Jacek R, Kaim I, et al. Estimated risk for altered fetal growth resulting from exposure to fine particles during pregnancy: an epidemiologic prospective cohort study in Poland. Environ Health Perspect. 2004; 112:1398–1402.11. Hertz-Picciotto I, Park HY, Dostal M, Kocan A, Trnovec T, Sram R. Prenatal exposures to persistent and non-persistent organic compounds and effects on immune system development. Basic Clin Pharmacol Toxicol. 2008; 102:146–154.12. Herr CE, Ghosh R, Dostal M, Skokanova V, Ashwood P, Lipsett M, et al. Exposure to air pollution in critical prenatal time windows and IgE levels in newborns. Pediatr Allergy Immunol. 2011; 22:75–84.13. Latzin P, Frey U, Armann J, Kieninger E, Fuchs O, Röösli M, et al. Exposure to moderate air pollution during late pregnancy and cord blood cytokine secretion in healthy neonates. PLoS One. 2011; 6:e23130.14. Hertz-Picciotto I, Herr CE, Yap PS, Dostál M, Shumway RH, Ashwood P, et al. Air pollution and lymphocyte phenotype proportions in cord blood. Environ Health Perspect. 2005; 113:1391–1398.15. Gern JE. Viral respiratory infection and the link to asthma. Pediatr Infect Dis J. 2008; 27:S97–S103.16. Sigurs N, Aljassim F, Kjellman B, Robinson PD, Sigurbergsson F, Bjarnason R, et al. Asthma and allergy patterns over 18 years after severe RSV bronchiolitis in the first year of life. Thorax. 2010; 65:1045–1052.17. Holt PG, Sly PD. Viral infections and atopy in asthma pathogenesis: new rationales for asthma prevention and treatment. Nat Med. 2012; 18:726–735.18. Ciencewicki J, Trivedi S, Kleeberger SR. Oxidants and the pathogenesis of lung diseases. J Allergy Clin Immunol. 2008; 122:456–468.19. Kim BJ, Lee SY, Kim HB, Lee E, Hong SJ. Environmental changes, microbiota, and allergic diseases. Allergy Asthma Immunol Res. 2014; 6:389–400.20. Gilliland FD, Li YF, Dubeau L, Berhane K, Avol E, McConnell R, et al. Effects of glutathione S-transferase M1, maternal smoking during pregnancy, and environmental tobacco smoke on asthma and wheezing in children. Am J Respir Crit Care Med. 2002; 166:457–463.21. Breton CV, Byun HM, Wenten M, Pan F, Yang A, Gilliland FD. Prenatal tobacco smoke exposure affects global and gene-specific DNA methylation. Am J Respir Crit Care Med. 2009; 180:462–467.22. Guerrero-Preston R, Goldman LR, Brebi-Mieville P, Ili-Gangas C, Lebron C, Witter FR, et al. Global DNA hypomethylation is associated with in utero exposure to cotinine and perfluorinated alkyl compounds. Epigenetics. 2010; 5:539–546.23. Flom JD, Ferris JS, Liao Y, Tehranifar P, Richards CB, Cho YH, et al. Prenatal smoke exposure and genomic DNA methylation in a multiethnic birth cohort. Cancer Epidemiol Biomarkers Prev. 2011; 20:2518–2523.24. Kim HB, Ahn KM, Kim KW, Shin YH, Yu J, Seo JH, et al. Cord blood cellular proliferative response as a predictive factor for atopic dermatitis at 12 months. J Korean Med Sci. 2012; 27:1320–1326.25. Yang HJ, Lee SY, Suh DI, Shin YH, Kim BJ, Seo JH, et al. The Cohort for Childhood Origin of Asthma and allergic diseases (COCOA) study: design, rationale and methods. BMC Pulm Med. 2014; 14:109.26. Pawankar R, Canonica GW, Holgate ST, Lockey RF. World Allergy Organization (WAO) white book on allergy. Milwaukee (WI): World Allergy Organization;2011.27. Yoon C, Ryu K, Kim J, Lee K, Park D. New approach for particulate exposure monitoring: determination of inhaled particulate mass by 24 h real-time personal exposure monitoring. J Expo Sci Environ Epidemiol. 2012; 22:344–351.28. Magnusson LL, Olesen AB, Wennborg H, Olsen J. Wheezing, asthma, hayfever, and atopic eczema in childhood following exposure to tobacco smoke in fetal life. Clin Exp Allergy. 2005; 35:1550–1556.29. Jedrychowski W, Perera FP, Maugeri U, Mrozek-Budzyn D, Mroz E, Flak E, et al. Early wheezing phenotypes and severity of respiratory illness in very early childhood: study on intrauterine exposure to fine particle matter. Environ Int. 2009; 35:877–884.30. Jedrychowski WA, Perera FP, Maugeri U, Mrozek-Budzyn D, Mroz E, Klimaszewska-Rembiasz M, et al. Intrauterine exposure to polycyclic aromatic hydrocarbons, fine particulate matter and early wheeze. Prospective birth cohort study in 4-year olds. Pediatr Allergy Immunol. 2010; 21:e723–e732.31. Jedrychowski WA, Perera FP, Spengler JD, Mroz E, Stigter L, Flak E, et al. Intrauterine exposure to fine particulate matter as a risk factor for increased susceptibility to acute broncho-pulmonary infections in early childhood. Int J Hyg Environ Health. 2013; 216:395–401.32. Raherison C, Pénard-Morand C, Moreau D, Caillaud D, Charpin D, Kopfersmitt C, et al. In utero and childhood exposure to parental tobacco smoke, and allergies in schoolchildren. Respir Med. 2007; 101:107–117.33. Singh SP, Gundavarapu S, Peña-Philippides JC, Rir-Sima-ah J, Mishra NC, Wilder JA, et al. Prenatal secondhand cigarette smoke promotes Th2 polarization and impairs goblet cell differentiation and airway mucus formation. J Immunol. 2011; 187:4542–4552.34. Jedrychowski WA, Perera FP, Majewska R, Camman D, Spengler JD, Mroz E, et al. Separate and joint effects of tranplacental and postnatal inhalatory exposure to polycyclic aromatic hydrocarbons: prospective birth cohort study on wheezing events. Pediatr Pulmonol. 2014; 49:162–172.35. Kelly FJ. Oxidative stress: its role in air pollution and adverse health effects. Occup Environ Med. 2003; 60:612–616.36. Auten RL, Potts EN, Mason SN, Fischer B, Huang Y, Foster WM. Maternal exposure to particulate matter increases postnatal ozone-induced airway hyperreactivity in juvenile mice. Am J Respir Crit Care Med. 2009; 180:1218–1226.37. Auerbach A, Hernandez ML. The effect of environmental oxidative stress on airway inflammation. Curr Opin Allergy Clin Immunol. 2012; 12:133–139.38. Wu W, Peden DB, McConnell R, Fruin S, Diaz-Sanchez D. Glutathione-S-transferase M1 regulation of diesel exhaust particle-induced pro-inflammatory mediator expression in normal human bronchial epithelial cells. Part Fibre Toxicol. 2012; 9:31.39. Messier EM, Day BJ, Bahmed K, Kleeberger SR, Tuder RM, Bowler RP, et al. N-acetylcysteine protects murine alveolar type II cells from cigarette smoke injury in a nuclear erythroid 2-related factor-2-independent manner. Am J Respir Cell Mol Biol. 2013; 48:559–567.40. Wu W, Muller R, Berhane K, Fruin S, Liu F, Jaspers I, et al. Inflammatory response of monocytes to ambient particles varies by highway proximity. Am J Respir Cell Mol Biol. 2014; 51:802–809.41. Kohli A, Garcia MA, Miller RL, Maher C, Humblet O, Hammond SK, et al. Secondhand smoke in combination with ambient air pollution exposure is associated with increasedx CpG methylation and decreased expression of IFN-gamma in T effector cells and Foxp3 in T regulatory cells in children. Clin Epigenetics. 2012; 4:17.42. Wang IJ, Hsieh WS, Wu KY, Guo YL, Hwang YH, Jee SH, et al. Effect of gestational smoke exposure on atopic dermatitis in the offspring. Pediatr Allergy Immunol. 2008; 19:580–586.43. Carlsten C, Dimich-Ward H, DyBuncio A, Becker AB, Chan-Yeung M. Cotinine versus questionnaire: early-life environmental tobacco smoke exposure and incident asthma. BMC Pediatr. 2012; 12:187.44. Yi O, Kwon HJ, Kim H, Ha M, Hong SJ, Hong YC, et al. Effect of environmental tobacco smoke on atopic dermatitis among children in Korea. Environ Res. 2012; 113:40–45.45. Masuko H, Sakamoto T, Kaneko Y, Iijima H, Naito T, Noguchi E, et al. An interaction between Nrf2 polymorphisms and smoking status affects annual decline in FEV1: a longitudinal retrospective cohort study. BMC Med Genet. 2011; 12:97.46. Lee E, Chang HY, Lee KS, Suh DI, Yu HS, Kang MJ, et al. The effect of perinatal anxiety on bronchiolitis is influenced by polymorphisms in ROS-related genes. BMC Pulm Med. 2014; 14:154.47. Kang SH, Jung YH, Kim HY, Seo JH, Lee JY, Kwon JW, et al. Effect of paracetamol use on the modification of the development of asthma by reactive oxygen species genes. Ann Allergy Asthma Immunol. 2013; 110:364–369.e1.48. Brasch-Andersen C, Christiansen L, Tan Q, Haagerup A, Vestbo J, Kruse TA. Possible gene dosage effect of glutathione-S-transferases on atopic asthma: using real-time PCR for quantification of GSTM1 and GSTT1 gene copy numbers. Hum Mutat. 2004; 24:208–214.