J Gynecol Oncol.
2008 Jun;19(2):113-116. 10.3802/jgo.2008.19.2.113.
Does pretreatment HPV viral load correlate with prognosis in patients with early stage cervical carcinoma?
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Ajou University School of Medicine, Suwon, Korea. drchang@ajou.ac.kr
- KMID: 2129910
- DOI: http://doi.org/10.3802/jgo.2008.19.2.113
Abstract
-
OBJECTIVE
Recent data suggest that pretreatment HPV (Human papillomavirus) viral load is useful to predict the severity of intraepithelial lesions of the uterine cervix and formulate a treatment plan. However, the relationship between initial HPV viral load and prognosis of cervical cancer patients has not yet been clearly defined. The objective of this study was to determine whether HPV viral load has prognostic significance in patients with early stage cervical carcinoma treated by surgery.
METHODS
A retrospective review of all patients with early stage cervical carcinoma who underwent radical hysterectomy and pelvic lymphadenectomy at our institution from August 2003 to December 2007 was conducted. Patients were included only if they had pretreatment Hybrid Capture II test for HPV DNA detection.
RESULTS
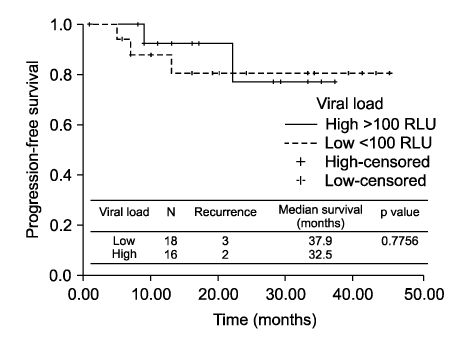
We identified 34 patients who met the inclusion criteria. Two groups were identified: patients who had low HPV viral load (< or =100 RLU) versus those who had high viral load (>100 RLU). There were no differences in age, FIGO stage, histology, pathologic risk factors - tumor size, deep stromal invasion, lymph-vascular space invasion, parametrial extensions, vaginal margin involvement, and lymph node metastasis - and adjuvant CCRT. There was no significant difference of disease-free survival regard to pretreatment HPV viral load (p=0.7756).
CONCLUSION
In our study, survival was not significantly different between early stage cervical cancer patients who had low and high pretreatment HPV viral load. It seems that pretreatment HPV viral load may not be of help to predict disease prognosis.
Keyword
MeSH Terms
Figure
Reference
-
1. Schiffman MH, Bauer HM, Hoover RN, Glass AG, Cadell DM, Rush BB, et al. Epidemiologic evidence showing that human papillomavirus infection causes most cervical intraepithelial neoplasia. J Natl Cancer Inst. 1993. 85:958–964.2. Bosch FX, Manos MM, Muñoz N, Sherman M, Jansen AM, Peto J, et al. Prevalence of human papillomavirus in cervical cancer: A worldwide perspective. International biological study on cervical cancer (IBSCC) Study Group. J Natl Cancer Inst. 1995. 87:796–802.3. Schlecht NF, Kulaga S, Robitaille J, Ferreira S, Santos M, Miyamura RA, et al. Persistent human papillomavirus infection as a predictor of cervical intraepithelial neoplasia. JAMA. 2001. 286:3106–3114.4. Londesborough P, Ho L, Terry G, Cuzick J, Wheeler C, Singer A. Human papillomavirus genotypes as a predictor of persistence and development of high-grade lesions in women with minor cervical abnormalities. Int J Cancer. 1996. 69:364–368.5. Remmink AJ, Walboomers JM, Helmerhorst TJ, Voorhorst FJ, Rozendaal L, Risse EK, et al. The presence of persistent high-risk HPV genotypes in dysplastic cervical lesions is associated with progressive disease: Natural history up to 36 months. Int J Cancer. 1995. 61:306–311.6. Ho GY, Burk RD, Klein S, Kadish AS, Chang CJ, Palan P, et al. Persistent genital human papillomavirus infection as a risk factor for persistent cervical dysplasia. J Natl Cancer Inst. 1995. 87:1365–1371.7. Ho GY, Palan PR, Basu J, Romney SL, Kadish AS, Mikhail M, et al. Viral characteristics of human papillomavirus infection and antioxidant levels as risk factors for cervical dysplasia. Int J Cancer. 1998. 78:594–599.8. Cuzick J, Terry G, Ho L, Hollingworth T, Anderson M. Human papillomavirus type 16 in cervical smears as predictor of high-grade cervical intraepithelial neoplasia. Lancet. 1992. 339:959–960.9. Bavin PJ, Giles JA, Deery A, Crow J, Griffiths PD, Emery VC, et al. Use of semi-quantitative PCR for human papillomavirus DNA type 16 to identify women with high-grade cervical disease in a population presenting with a mild dyskaryotic smear report. Br J Cancer. 1993. 67:602–605.10. Cuzick J, Terry G, Ho L, Hollingworth T, Anderson M. Type-specific human papillomavirus DNA in abnormal scrapings as a predictor of high-grade cervical intraepithelial neoplasia. Br J Cancer. 1994. 69:167–171.11. Dalstein V, Riethmuller D, Prétet JL, Le Bail Carval K, Sautière JL, Carbillet JP, et al. Persistence and load of high-risk HPV are predictors for development of high-grade cervical lesions: A longitudinal French cohort study. Int J Cancer. 2003. 106:396–403.12. Hernández-Hernández DM, Ornelas-Bernal L, Guido-Jiménez M, Apresa-Garcia T, Alvarado-Cabrero I, Salcedo-Vargas M, et al. Association between high-risk human papillomavirus DNA load and precursor lesions of cervical cancer in Mexican women. Gynecol Oncol. 2003. 90:310–317.13. Park JS, Ryu HS, Chang SJ, Kim YM, Chang KH, Lee JP. The association of the cervical intraepithelial neoplasia and human papillomavirus viral Load. Korean J Obstet Gynecol. 2005. 48:2888–2895.14. Farthing A, Masterson P, Mason WP, Vousden KH. Human papillomavirus detection by hybrid capture and its possible clinical use. J Clin Pathol. 1994. 47:649–652.15. Sun XW, Ferenczy A, Johnson D, Koulos JP, Lungu O, Richard RM, et al. Evaluation of the Hybrid Capture human papillomavirus deoxyribonucleic acid detection test. Am J Obstet Gynecol. 1995. 173:1432–1437.16. Sailors J, Gander R, Saboorian MH, Berkley P, Foster B, Ashfaq R. Stability of PreservCyt for Hybrid Capture (HC II) HPV test. Diagn Cytopathol. 2005. 32:260–263.17. Hubbard RA. Human Papillomavirus testing methods. Arch Pathol Lab Med. 2003. 127:940–945.18. Bolick DR, Bolick RE, Coates F, Daniels CM, Juretich MB, Lin KK, et al. Laboratory implementation of human papillomavirus testing. Arch Pathol Lab Med. 2003. 127:984–990.19. Castle PE, Lorincz AT, Mielzynska-Lohnas I, Scott DR, Glass AG, Sherman ME, et al. Results of human papillomavirus DNA testing with the hybrid capture 2 assay are reproducible. J Clin Microbiol. 2002. 40:1088–1090.20. Belinson J, Qiao YL, Pretorius R, Zhang WH, Elson P, Li L, et al. Shanxi province cervical cancer screening study: A cross sectional comparativetrial of multiple techniques to detect cervical neoplasia. Gynecol Oncol. 2001. 83:439–444.21. Wright TC Jr, Denny L, Kuhn L, Pollack A, Lorincz A. HPV DNA testing of self collected vaginal samples compared with cytologic screening to detect cervical cancer. JAMA. 2000. 283:81–86.22. Schiffman M, Herrero R, Hildesheim A, Sherman ME, Bratti M, Wacholder S, et al. HPV DNA testing in cervical cancer screening: Results from women in a high risk province of Costa Rica. JAMA. 2000. 283:87–93.23. Kuhn L, Denny L, Pollack A, Lorincz A, Richart RM, Wright TC, et al. Human papillomavirus DNA testing for cervical cancer screening in low resource settings. J Natl Cancer Inst. 2000. 92:818–825.24. Bozzetti M, Nonnenmacher B, Mielzinska II, Villa L, Lorincz A, Breitenbach VV, et al. Comparison between hybrid capture II and polymerase chain reaction results among women at low risk for cervical cancer. Ann Epidemiol. 2000. 10:466.25. Park JY, Lee KH, Dong SM, Kang S, Park SY, Seo SS. The association of pre-conization high-risk HPV load and the persistence of HPV infection and persistence/recurrence of cervical intraepithelial neoplasia after conization. Gynecol Oncol. 2008. 108:549–554.26. Riou G, Favre M, Jeannel D, Bourhis J, Le Doussal V, Ortho G. Association between poor prognosis in early stage invasive cervical carcinoma and non-detection of HPV DNA. Lancet. 1990. 335:1171–1174.27. Datta NR, Kumar P, Singh S, Gupta D, Srivastava A, Dhole TN. Does pretreatment human papillomavirus titers predict radiation response and survival outcomes in cancer cervix? Gynecol Oncol. 2006. 103:100–105.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Low initial human papillomavirus viral load may indicate worse prognosis in patients with cervical carcinoma treated with surgery
- Physical Status and Viral load in Women with Positive Human Papillomavirus (HPV) Infection in Uterine Cervix
- The association of the cervical intraepithelial neoplasia and human papillomavirus viral load
- A phase 1/2a, dose-escalation, safety, and preliminary efficacy study of the RKP00156 vaginal tablet in healthy women and patients with cervical intraepithelial neoplasia 2
- The utility of the human papillomavirus DNA load for the diagnosis and prediction of persistent vaginal intraepithelial neoplasia