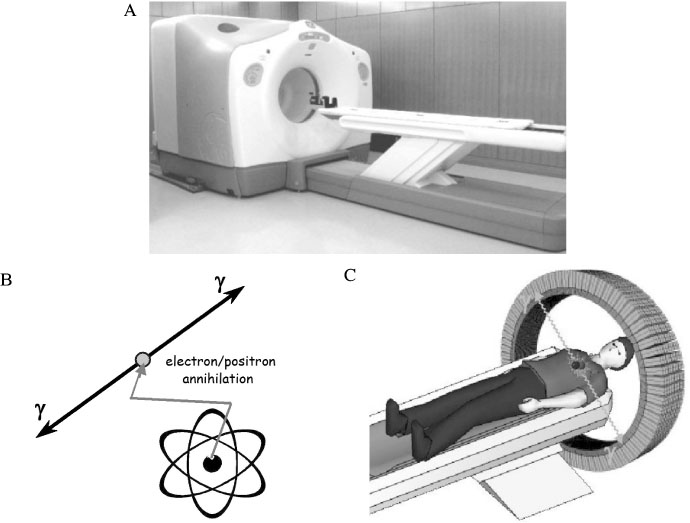
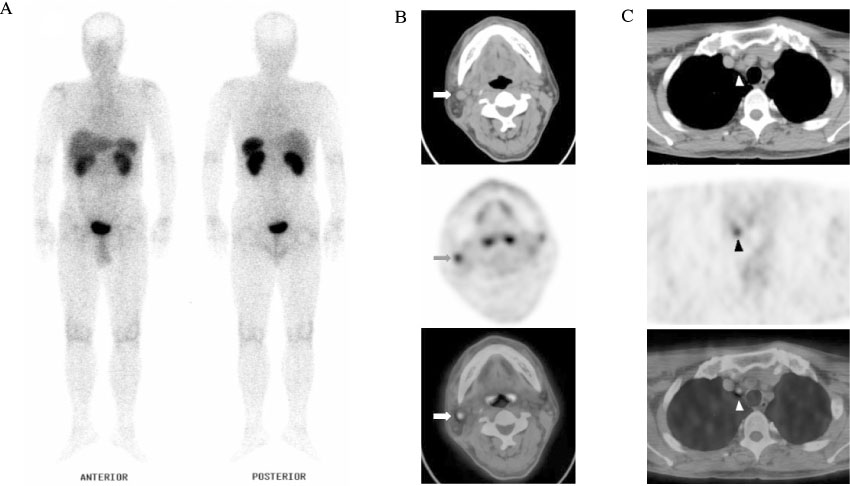
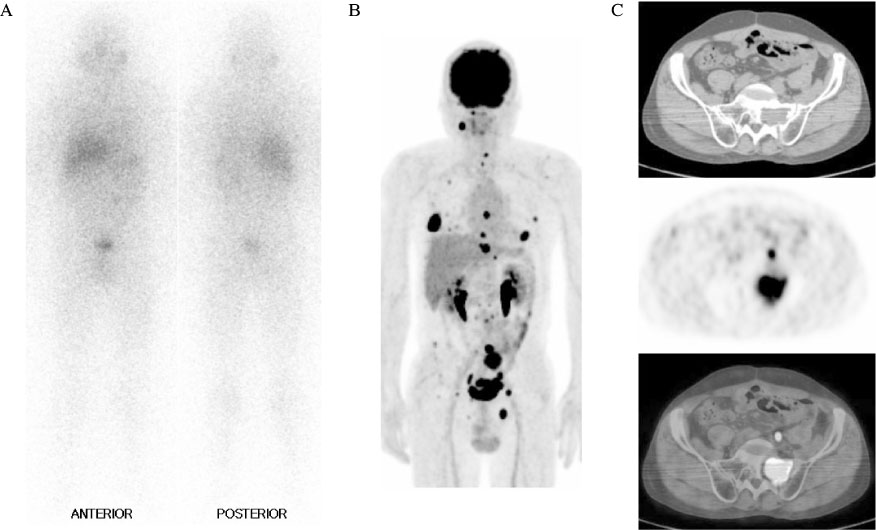
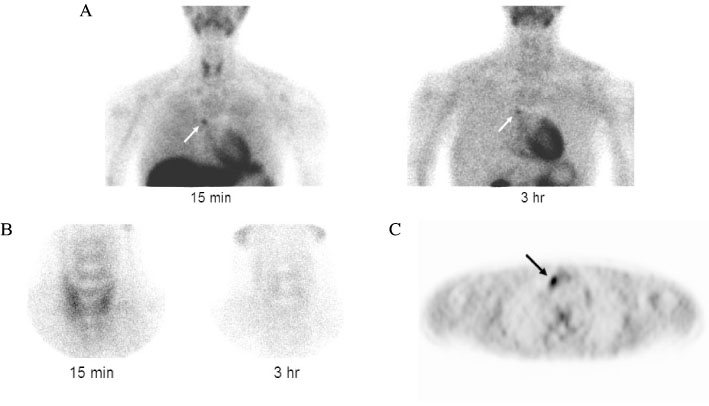
J Korean Endocr Soc.
2007 Dec;22(6):397-406. 10.3803/jkes.2007.22.6.397.
PET Application in Neuroendocrine Tumors
- Affiliations
-
- 1Department of Nuclear Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Korea.
- KMID: 1958903
- DOI: http://doi.org/10.3803/jkes.2007.22.6.397
Abstract
- No abstract available.
MeSH Terms
Figure
Reference
-
1. Jeong JG. PET application in thyroid cancer. J Kor Endocrinol Soc. 2004. 19:1–9.2. Gambhir SS. Molecular imaging of cancer with positron emission tomography. Nat Rev Cancer. 2002. 2:683–693.3. Rufini V, Salvatori M, Garganese MC, Di Giuda D, Lodovica Maussier M, Troncone L. Role of nuclear medicine in the diagnosis and therapy of medullary thyroid carcinoma. Rays. 2000. 25:273–282.4. Baulieu JL, Guilloteau D, Delisle MJ, Perdrisot R, Gardet P, Delpine N, Baulieu F, Dupont JL, Talbot JN, Coutris G. Radioiodinated meta-iodobenzylguanidine uptake in medullary thyroid cancer. A French cooperative study. Cancer. 1987. 60:2189–2194.5. Baudin E, Lumbroso J, Schlumberger M, Leclere J, Giammarile F, Gardet P, Roche A, Travagli JP, Parmentier C. Comparison of octreotide scintigraphy and conventional imaging in medullary thyroid carcinoma. J Nucl Med. 1996. 37:912–916.6. Drr U, Sautter-Bihl ML, Bihl H. The contribution of somatostatin receptor scintigraphy to the diagnosis of recurrent medullary carcinoma of the thyroid. Semin Oncol. 1994. 21:Suppl 13. 42–45.7. Diehl M, Risse JH, Brandt-Mainz K, Dietlein M, Bohuslavizki KH, Matheja P, Lange H, Bredow J, Körber C, Grünwald F. Fluorine-18 fluorodeoxyglucose positron emission tomography in medullary thyroid cancer: results of a multicentre study. Eur J Nucl Med. 2001. 28:1671–1676.8. Szakáll S Jr, Esik O, Bajzik G, Repa I, Dabasi G, Sinkovics I, Agoston P, Trn L. 18F-FDG PET detection of lymph node metastases in medullary thyroid carcinoma. J Nucl Med. 2002. 43:66–71.9. de Groot JW, Links TP, Jager PL, Kahraman T, Plukker JT. Impact of 18F-fluoro-2-deoxy-D-glucose positron emission tomography (FDG-PET) in patients with biochemical evidence of recurrent or residual medullary thyroid cancer. Ann Surg Oncol. 2004. 11:786–794.10. Iagaru A, Masamed R, Singer PA, Conti PS. Detection of occult medullary thyroid cancer recurrence with 2-deoxy-2-[F-18]fluoro-D-glucose-PET and PET/CT. Mol Imaging Biol. 2007. 9:72–77.11. Adams S, Baum RP, Hertel A, Schumm-Dräger PM, Usadel KH, Hör G. Metabolic (PET) and receptor (SPET) imaging of well- and less well-differentiated tumours: comparison with the expression of the Ki-67 antigen. Nucl Med Commun. 1998. 19:641–647.12. Ong SC, Schöder H, Patel SG, Tabangay-Lim IM, Doddamane I, Gnen M, Shaha AR, Tuttle RM, Shah JP, Larson SM. Diagnostic accuracy of 18F-FDG PET in restaging patients with medullary thyroid carcinoma and elevated calcitonin levels. J Nucl Med. 2007. 48:501–507.13. Gotthardt M, Battmann A, Höffken H, Schurrat T, Pollum H, Beuter D, Gratz S, Béhé M, Bauhofer A, Klose KJ, Behr TM. 18F-FDG PET, somatostatin receptor scintigraphy, and CT in metastatic medullary thyroid carcinoma: a clinical study and an analysis of the literature. Nucl Med Commun. 2004. 25:439–443.14. Brandt-Mainz K, Müller SP, Görges R, Saller B, Bockisch A. The value of fluorine-18 fluorodeoxyglucose PET in patients with medullary thyroid cancer. Eur J Nucl Med. 2000. 27:490–496.15. Maurea S, Cuocolo A, Reynolds JC, Tumeh SS, Begley MG, Linehan WM, Norton JA, Walther MM, Keiser HR, Neumann RD. Iodine-131-metaiodobenzylguanidine scintigraphy in preoperative and post-operative evaluation of paragangliomas: comparison with CT and MRI. J Nucl Med. 1993. 34:173–179.16. Jalil ND, Pattou FN, Combemale F, Chapuis Y, Henry JF, Peix JL, Proye CA. Effectiveness and limits of preoperative imaging studies for the localisation of pheochromocytomas and paragangliomas: a review of 282 cases. French Association of Surgery (AFC), and the French Association of Endocrine Surgeons (AFCE). Eur J Surg. 1998. 164:23–28.17. Berglund AS, Hulthen UL, Manhem P, Thorsson O, Wollmer P, Tornquist C. Metaiodobenzylguanidine (MIBG) scintigraphy and computed tomography (CT) in clinical practice. Primary and secondary evaluation for localization of pheochromocytomas. J Intern Med. 2001. 249:247–251.18. Guller U, Turek J, Eubanks S, Delong ER, Oertli D, Feldman JM. Detecting pheochromocytoma: defining the most sensitive test. Ann Surg. 2006. 243:102–107.19. Furuta N, Kiyota H, Yoshigoe F, Hasegawa N, Ohishi Y. Diagnosis of pheochromocytoma using [123I]-compared with [131]-metaiodobenzylguanidine scintigraphy. Int J Urol. 1999. 6:119–124.20. van der Harst E, De Herder WW, Bruining HA, Bonjer HJ, de Krijger RR, Lamberts SW, van de Meiracker AH, Boomsma F, Stijnen T, Krenning EP, Bosman FT, Kwekkeboom DJ. [123I] Metaiodobenzylguanidine and [111In]Octreotide uptake in benign and malignant pheochromocytomas. J Clin Endocrinol Metab. 2001. 86:685–693.21. Pacak K, Eisenhofer G, Carrasquillo JA, Chen CC, Li ST, Goldstein DS. 6-[18F]Fluorodopamine positron emission tomographic (PET) scanning for diagnostic localization of pheochromocytoma. Hypertension. 2001. 38:6–8.22. Shulkin BL, Wieland DM, Schwaiger M, Thompson NW, Francis IR, Haka MS, Rosenspire KC, Shapiro B, Sisson JC, Kuhl DE. PET scanning with hydroxyephedrine: an approach to the localization of pheochromocytoma. J Nucl Med. 1992. 33:1125–1131.23. Shapiro B, Gross MD, Shulkin B. Radioisotope diagnosis andtherapy of malignant pheochromocytoma. Trends Endocrinol Metab. 2001. 12:469–475.24. Eisenhofer G. The role of neuronal and extraneuronal plasma membrane transporters in the inactivation of peripheral catecholamines. Pharmacol Ther. 2001. 91:35–62.25. Pacak K, Linehan WM, Eisenhofer G, Walther MM, Goldstein DS. Recent advances in genetics, diagnosis, localization, and treatment of pheochromocytoma. Ann Intern Med. 2001. 134:315–329.26. Hoegerle S, Nitzsche E, Altehoefer C, Ghanem N, Manz T, Brink I, Reincke M, Moser E, Neumann HP. Pheochromocytomas: detection with 18F DOPA whole-body PET-initial results. Radiology. 2002. 222:507–512.27. Trampal C, Engler H, Juhlin C, Bergstrom M, Langstrom B. Pheochromocytomas: detection with 11C hydroxyephedrine PET. Radiology. 2004. 230:423–428.28. Shulkin BL, Thompson NW, Shapiro B, Francis IR, Sisson JC. Pheochromocytomas: imaging with 2-[fluorine-18]fluoro-2-deoxy-D-glucose PET. Radiology. 1999. 212:35–41.29. Mamede M, Carrasquillo JA, Chen CC, Del Corral P, Whatley M, Ilias I, Ayala A, Pacak K. Discordant localization of 2-[18F]-fluoro-2-deoxy-D-glucose in 6-[18F]-fluorodopamine- and [123I]-metaiodobenzylguanidine-negative metastatic pheochromocytoma sites. Nucl Med Commun. 2006. 27:31–36.30. Timmers HJ, Kozupa A, Chen CC, Carrasquillo JA, Ling A, Eisenhofer G, Adams KT, Solis D, Lenders JW, Pacak K. Superiority of fluorodeoxyglucose positron emission tomography to other functional imaging techniques in the evaluation of metastatic SDHB-associated pheochromocytoma and paraganglioma. J Clin Oncol. 2007. 25:2262–2269.31. Pacak K, Eisenhofer G, Goldstein DS. Functional imaging of endocrine tumors: role of positron emission tomography. Endocr Rev. 2004. 25:568–580.32. Rufini V, Calcagni ML, Baum RP. Imaging of neuroendocrine tumors. Semin Nucl Med. 2006. 36:228–247.33. Krenning EP, Kwekkeboom DJ, Bakker WH, Breeman WA, Kooij PP, Oei HY, van Hagen M, Postema PT, de Jong M, Reubi JC. Somatostatin receptor scintigraphy with [111In-DTPA-D-Phe1]- and [123I-Tyr3]-octreotide: the Rotterdam experience with more than 1000 patients. Eur J Nucl Med. 1993. 20:716–731.34. Janson ET, Westlin JE, Eriksson B, Ahlstrom H, Nilsson S, Oberg K. [111In-DTPA-D-Phe1]octreotide scintigraphy in patients with carcinoid tumours: the predictive value for somatostatin analogue treatment. Eur J Endocrinol. 1994. 131:577–581.35. Eriksson B, Bergström M, Orlefrs H, Sundin A, Oberg K, Langstrm B. Use of PET in neuroendocrine tumors. In vivo applications and in vitro studies. Q J Nucl Med. 2000. 44:6876.36. Bombardieri E, Maccauro M, De Deckere E, Savelli G, Chiti A. Nuclear medicine imaging of neuroendocrine tumours. Ann Oncol. 2001. 12:S51–S61.37. Orlefors H, Sundin A, Garske U, Juhlin C, Oberg K, Skogseid B, Langstrom B, Bergstrom M, Eriksson B. Whole-body 11C-5-hydroxytryptophan positron emission tomography as a universal imaging technique for neuroendocrine tumors: comparison with somatostatin receptor scintigraphy and computed tomography. J Clin Endocrinol Metab. 2005. 90:3392–3400.38. Hoegerle S, Altehoefer C, Ghanem N, Koehler G, Waller CF, Scheruebl H, Moser E, Nitzsche E. Whole-body 18F-dopa PET for detection of gastrointestinal carcinoid tumors. Radiology. 2001. 220:373–380.39. Hofmann M, Maecke H, Borner R, Weckesser E, Schoffski P, Oei L, Schumacher J, Henze M, Heppeler A, Meyer J, Knapp H. Biokinetics and imaging with the somatostatin receptor PET radioligand 68Ga-DOTATOC: preliminary data. Eur J Nucl Med. 2001. 28:1751–1757.40. Kowalski J, Henze M, Schuhmacher J, Mcke HR, Hofmann M, Haberkorn U. Evaluation of positron emission tomography imaging using [68Ga]-DOTA-D Phe(1)-Tyr(3)-Octreotide in comparison to [111In]-DTPAOC SPECT. First results in patients with neuroendocrine tumors. Mol Imaging Biol. 2003. 5:42–48.41. Adams S, Baum R, Rink T, Schumm-Drager PM, Usadel KH, Hör G. Limited value of fluorine-18 fluorodeoxyglucose positron emission tomography for the imaging of neuroendocrine tumours. Eur J Nucl Med. 1998. 25:79–83.42. Nakamoto Y, Higashi T, Sakahara H, Tamaki N, Itoh K, Imamura M, Konishi J. Evaluation of pancreatic islet cell tumors by fluorine-18 fluorodeoxyglucose positron emission tomography: comparison with other modalities. Clin Nucl Med. 2000. 25:115–119.43. Dwamena BA, Kloos RT, Fendrick AM, Gross MD, Francis IR, Korobkin MT, Shapiro B. Diagnostic evaluation of the adrenal incidentaloma: decision and cost-effectiveness analyses. J Nucl Med. 1998. 39:707–712.44. Gross MD, Shapiro B, Francis IR, Glazer GM, Bree RL, Arcomano MA, Schteingart DE, McLeod MK, Sanfield JA, Thompson NW. Scintigraphic evaluation of clinically silent adrenal masses. J Nucl Med. 1994. 35:1145–1152.45. Weber MM, Lang J, Abedinpour F, Zeilberger K, Adelmann B, Engelhardt D. Different inhibitory effect of etomidate and ketoconazole on the human adrenal steroid biosynthesis. Clin Investig. 1993. 71:933–938.46. Bergström M, Juhlin C, Bonasera TA, Sundin A, Rastad J, Akerström G, Långström B. PET imaging of adrenal cortical tumors with the 11β-hydroxylase tracer 11C-metomidate. J Nucl Med. 2000. 41:275–282.47. Bergström M, Bonasera TA, Lu L, Bergström E, Backlin C, Juhlin C, Långström B. In vitro and in vivo primate evaluation of carbon-11-etomidate and carbon-11-metomidate as potential tracers for PET imaging of the adrenal cortex and its tumors. J Nucl Med. 1998. 39:982–989.48. Boland GW, Goldberg MA, Lee MJ, Mayo-Smith WW, Dixon J, McNicholas MM, Mueller PR. Indeterminate adrenal mass in patients with cancer: evaluation at PET with 2-[F-18]-fluoro-2-deoxy-D-glucose. Radiology. 1995. 194:131–134.49. Maurea S, Klain M, Mainolfi C, Ziviello M, Salvatore M. The diagnostic role of radionuclide imaging in evaluation of patients with nonhypersecreting adrenal masses. J Nucl Med. 2001. 42:884–892.50. Yun M, Kim W, Alnafisi N, Lacorte L, Jang S, Alavi A. 18F-FDG PET in characterizing adrenal lesions detected on CT or MRI. J Nucl Med. 2001. 42:1795–1799.51. van Heerden JA, Grant CS. Surgical treatment of primary hyperparathyroidism: an institutional perspective. World J Surg. 1991. 15:688–692.52. Perrier ND, Ituarte PH, Morita E, Hamill T, Gielow R, Duh QY, Clark OH. Parathyroid surgery: separating promise from reality. J Clin Endocrinol Metab. 2002. 87:1024–1029.53. O'Doherty MJ, Kettle AG, Wells P, Collins RE, Coakley AJ. Parathyroid imaging with technetium-99m-sestamibi: preoperative localization and tissue uptake studies. J Nucl Med. 1992. 33:313–318.54. Neumann DR, Esselstyn CB, Maclntyre WJ, Go RT, Obuchowski NA, Chen EQ, Licata AA. Comparison of FDG-PET and sestamibi-SPECT in primary hyperparathyroidism. J Nucl Med. 1996. 37:1809–1815.55. Melon P, Luxen A, Hamoir E, Meurisse M. Fluorine-18-fluorodeoxyglucose positron emission tomography for preoperative parathyroid imaging in primary hyperparathyroidism. Eur J Nucl Med. 1995. 22:556–558.56. Sundin A, Johansson C, Hellman P, Bergström M, Ahlström H, Jacobson GB, Långström B, Rastad J. PET and parathyroid L-[carbon-11]methionine accumulation in hyperparathyroidism. J Nucl Med. 1996. 37:1766–1770.57. Hellman P, Ahlström H, Bergstrm M, Sundin A, Långström B, Westerberg G, Juhlin C, Akerström G, Rastad J. Positron emission tomography with 11C-methionine in hyperparathyroidism. Surgery. 1994. 116:974–981.58. Cook GJ, Wong JC, Smellie WJ, Young AE, Maisey MN, Fogelman I. [11C]Methionine positron emission tomography for patients with persistent or recurrent hyperparathyroidism after surgery. Eur J Endocrinol. 1998. 139:195–197.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Neuroendocrine Tumors of the Female Reproductive Tract: A Literature Review
- Fine Needle Aspiration Cytologic Findings of Pulmonary Neuroendocrine Tumors
- A Case of Glomus Tumor Mimicking Neuroendocrine Tumor on 68 Ga-DOTATOC PET/CT
- Splenosis Mimicking Relapse of a Neuroendocrine Tumor at Gallium-68-DOTATOC PET/CT
- A Case of Neuroendocrine Carcinoma of the Thymus