Korean J Obstet Gynecol.
2012 Jan;55(1):1-7. 10.5468/KJOG.2012.55.1.1.
Pregnancy outcomes after exposure to phendimetrazine in first trimester of pregnancy
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Cheil General Hospital and Women's Healthcare Center, Kwandong University College of Medicine, Seoul, Korea. juneobgy@yahoo.co.kr
- 2The Korean Motherisk Program, Seoul, Korea.
- KMID: 1836756
- DOI: http://doi.org/10.5468/KJOG.2012.55.1.1
Abstract
OBJECTIVE
Phendimetrazine is an amphetamine analogue that acts as a sympathomimetic with anorectic properties, used for weight loss. There is scarce information on the safety of phendimetrazine associated with fetal anomaly when used early in pregnancy. Some studies suggested that use of phendimetrazine during pregnancy may increase the risk of cleft lip and palate, and congenital diaphragmatic hernia. The aim of the study was to evaluate fetal outcomes among pregnant women who were inadvertently exposed to phendimetrazine in early pregnancy.
METHODS
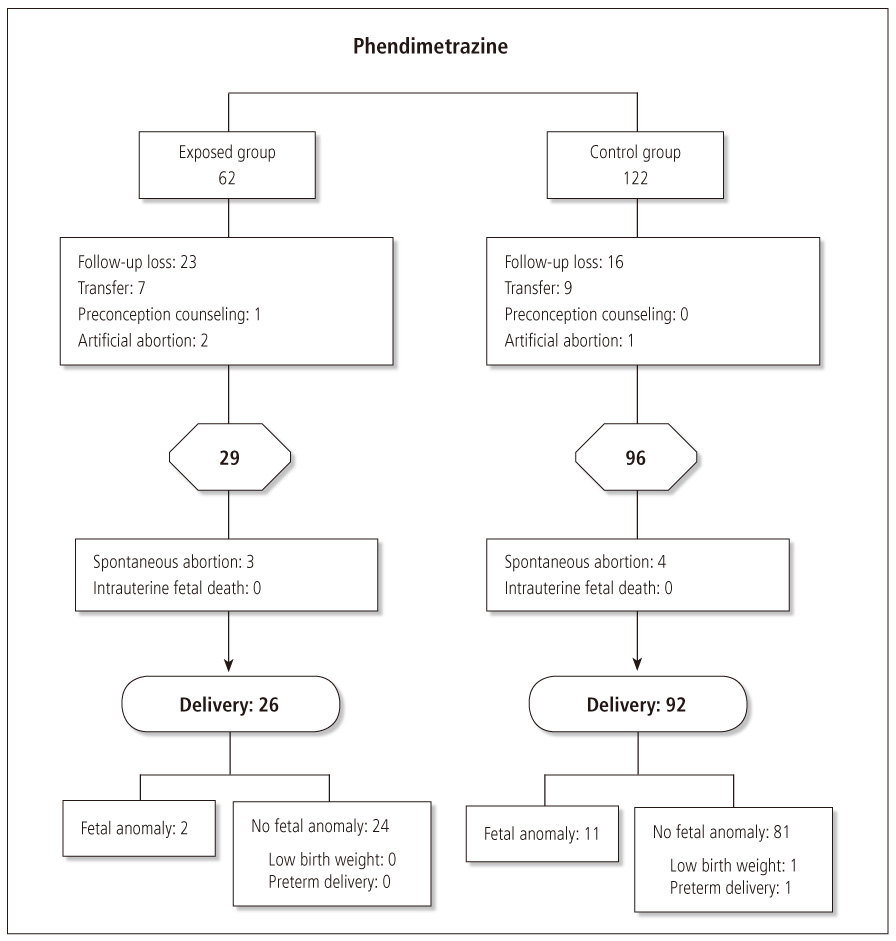
Total 62 singleton pregnant women who were inadvertently exposed to phendimetrazine during early pregnancy were prospectively followed up (twin pregnancy was excluded). In addition, 122 age and gravity matched pregnant women were not exposed to any potential teratogenic agent during pregnancy were recruited as controls.
RESULTS
Mean age of exposed women was 33.2 +/- 4.9 years, with mean gravity was 2.2 +/- 1.1 and mean gestational weeks at exposure was 4.2 +/- 2.0. All gestations were confirmed by ultrasonography. Of exposed women, 3 (1.0%) had spontaneous abortions, 2 had artificial abortion, 23 cases were lost to follow-up and 7 cases were transferred to other hospital. Therefore, 26 pregnancies inadvertently exposed to phendimetrazine were evaluated. Two babies had abnormal outcome: one had choroid plexus cyst, another baby had bilateral equinovarus. In control group, 11 babies were born with abnormal outcome (7.7% vs. 12.0%; P = 0.54; odds ratio, 0.61; 95% confidence interval, 0.12 to 2.96).
CONCLUSION
These preliminary results suggest that phendimetrazine may not pose substantial fetal risks. Although more information is needed, there is a critical need for further research on medication used during pregnancy.
Keyword
MeSH Terms
Figure
Reference
-
1. Kim BS, Kim YS, Ahn ES, Roh YJ, Jung SG, Kim JM, et al. Postmarketing surveillance study on the efficacy and safety of phendimetrazine in patients with obesity. Korean J Health Promot. 2010. 10:97–103.2. Milkovich L, van der Berg BJ. Effects of antenatal exposure to anorectic drugs. Am J Obstet Gynecol. 1977. 129:637–642.3. Powell PD, Johnstone JM. Phenmetrazine and foetal abnormalities. Br Med J. 1962. 2:1327.4. Moss PD. Phenmetrazine and foetal abnormalities. Br Med J. 1962. 2:1610.5. Fogh-Anderson P. Genetic and non-genetic factors in the etiology of facial clefts. Scand J Plast Reconstr Surg. 1967. 1:22–29.6. Choi JS, Han JY, Ahn HK, Lee SW, Kim MH, Chung JH, et al. Pregnancy outcomes after peri-conceptional medication exposure: 10 years experience. Study for application of reproductive toxicity information. Korean J Perinatol. 2010. 21:48–58.7. Cunningham F, Leveno K, Bloom S, Hauth J, Rouse D, Spong C. Williams obstetrics. 2009. 23rd ed. New York (NY): McGraw-Hill.8. Park KH. Update on anti-obesity medications. J Korean Med Assoc. 2005. 48:896–903.9. Lee JM, Lee KU, Jhoo JH, Park JI. Two cases of psychotic disorder following phendimetrazine use. Korean J Psychopharmacol. 2010. 21:95–98.10. Phendimetrazine [Internet]. Drugs.com. c2000-2011. cited 2011 Oct 25. Drugs.com;Available from: http://www.drugs.com/pro/phendimetrazine.html.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Impetigo Herpetiformis in Pregnancy
- A study of the factors associated with the pattern of gestational weight gain
- Torsion of Benign Cystic Teratoma of Ovary at 37 Weeks Gestation
- Autologous blood donation in the third trimester of pregnancy
- Cevix Cancer Associated with Pregnancy: A Case Report and Review of the Literature