J Gynecol Oncol.
2014 Jan;25(1):43-50. 10.3802/jgo.2014.25.1.43.
Survival benefit of taxane plus platinum in recurrent ovarian cancer with non-clear cell, non-mucinous histology
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Nagoya University Graduate School of Medicine, Nagoya, Japan. kajiyama@med.nagoya-u.ac.jp
- 2Department of Obstetrics and Gynecology, Toyohashi Municipal Hospital, Toyohashi, Japan.
- 3Department of Medical Laboratory Sciences, Nagoya University Graduate School of Medicine, School of Health Science, Nagoya, Japan.
- KMID: 1811187
- DOI: http://doi.org/10.3802/jgo.2014.25.1.43
Abstract
OBJECTIVE
This study was conducted to examine the effects of front-line chemotherapy on overall survival (OS) and postrecurrence survival (PRS) of patients with recurrent ovarian cancer, when stratifying the histologic type.
METHODS
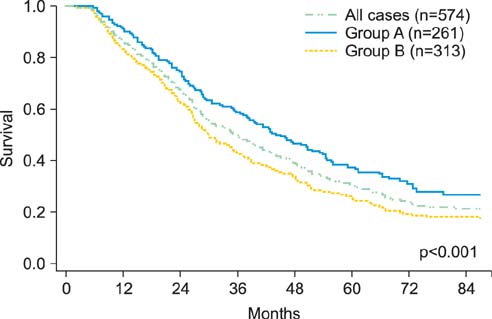
Five hundred and seventy-four patients with recurrent ovarian cancer with sufficient clinical information, including front-line chemotherapy, were analyzed. The pathologic slides were evaluated by central pathologic review. The patients were divided into two groups: group A (n=261), who underwent taxane plus platinum, and group B (n=313), who underwent conventional platinum-based chemotherapy without taxanes.
RESULTS
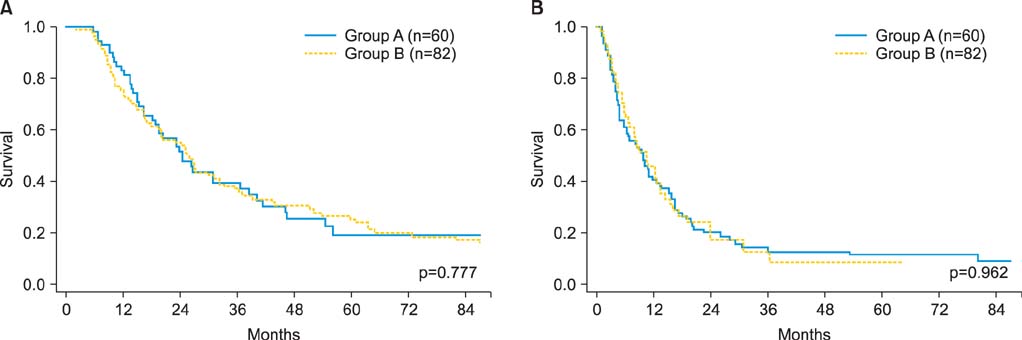
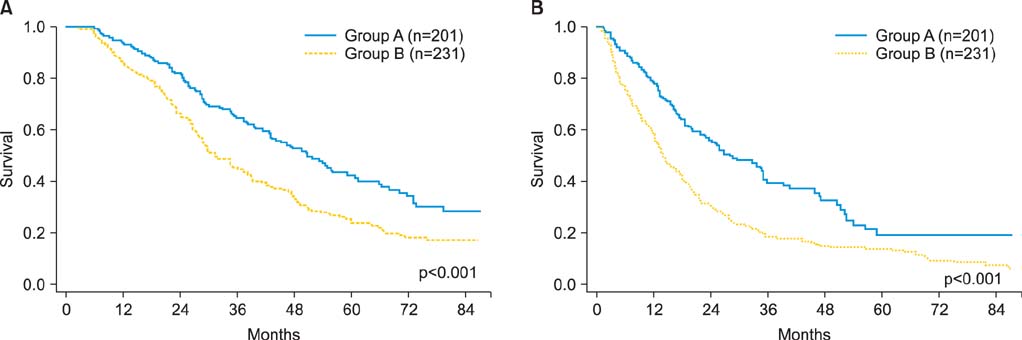
The median age was 54 years (range, 14 to 89 years). Group A had significantly better median OS (45.0 months vs. 30.3 months, p<0.001) and PRS (23.0 months vs. 13.0 months, p<0.001) compared to group B. The OS and PRS were similar between the groups in patients with clear cell or mucinous histology. In contrast, among patients with non-clear cell, non-mucinous histologies, the OS and PRS of group A were significantly better than those of group B (OS, p<0.001; PRS, p<0.001). Multivariable analyses revealed that, among patients with non-clear cell, non-mucinous histologies, chemotherapy including taxane and platinum was an independent predictor of favorable survival outcomes. Conversely, in patients with clear cell or mucinous histology, taxane-including platinum-based combination chemotherapy did not improve the OS and PRS compared to a conventional platinum-based regimen which did not include taxanes.
CONCLUSION
Since the emergence of taxane plus platinum, the prognosis of patients with recurrent ovarian cancer has improved. However, we here demonstrate that this improvement is limited to patients with non-clear cell, non-mucinous histologies.
Keyword
MeSH Terms
Figure
Reference
-
1. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011; 61:69–90.2. Eisenkop SM, Friedman RL, Wang HJ. Complete cytoreductive surgery is feasible and maximizes survival in patients with advanced epithelial ovarian cancer: a prospective study. Gynecol Oncol. 1998; 69:103–108.3. Friedlander M, Butow P, Stockler M, Gainford C, Martyn J, Oza A, et al. Symptom control in patients with recurrent ovarian cancer: measuring the benefit of palliative chemotherapy in women with platinum refractory/resistant ovarian cancer. Int J Gynecol Cancer. 2009; 19:Suppl 2. S44–S48.4. Meier W, du Bois A, Reuss A, Kuhn W, Olbricht S, Gropp M, et al. Topotecan versus treosulfan, an alkylating agent, in patients with epithelial ovarian cancer and relapse within 12 months following 1st-line platinum/paclitaxel chemotherapy: a prospectively randomized phase III trial by the Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Cancer Study Group (AGO-OVAR). Gynecol Oncol. 2009; 114:199–205.5. Hızlı D, Boran N, Yılmaz S, Turan T, Altınbaş SK, Celik B, et al. Best predictors of survival outcome after tertiary cytoreduction in patients with recurrent platinum-sensitive epithelial ovarian cancer. Eur J Obstet Gynecol Reprod Biol. 2012; 163:71–75.6. Oksefjell H, Sandstad B, Trope C. The role of secondary cytoreduction in the management of the first relapse in epithelial ovarian cancer. Ann Oncol. 2009; 20:286–293.7. Zang RY, Zhang ZY, Li ZT, Chen J, Tang MQ, Liu Q, et al. Effect of cytoreductive surgery on survival of patients with recurrent epithelial ovarian cancer. J Surg Oncol. 2000; 75:24–30.8. Zang RY, Li ZT, Tang J, Cheng X, Cai SM, Zhang ZY, et al. Secondary cytoreductive surgery for patients with relapsed epithelial ovarian carcinoma: who benefits? Cancer. 2004; 100:1152–1161.9. Benedetti Panici P, De Vivo A, Bellati F, Manci N, Perniola G, Basile S, et al. Secondary cytoreductive surgery in patients with platinum-sensitive recurrent ovarian cancer. Ann Surg Oncol. 2007; 14:1136–1142.10. Salani R, Santillan A, Zahurak ML, Giuntoli RL 2nd, Gardner GJ, Armstrong DK, et al. Secondary cytoreductive surgery for localized, recurrent epithelial ovarian cancer: analysis of prognostic factors and survival outcome. Cancer. 2007; 109:685–691.11. Dizon DS, Hensley ML, Poynor EA, Sabbatini P, Aghajanian C, Hummer A, et al. Retrospective analysis of carboplatin and paclitaxel as initial second-line therapy for recurrent epithelial ovarian carcinoma: application toward a dynamic disease state model of ovarian cancer. J Clin Oncol. 2002; 20:1238–1247.12. Christian J, Thomas H. Ovarian cancer chemotherapy. Cancer Treat Rev. 2001; 27:99–109.13. Kajiyama H, Shibata K, Mizuno M, Umezu T, Suzuki S, Yamamoto E, et al. Long-term clinical outcome of patients with recurrent epithelial ovarian carcinoma: is it the same for each histological type? Int J Gynecol Cancer. 2012; 22:394–399.14. Goff BA, Sainz de la Cuesta R, Muntz HG, Fleischhacker D, Ek M, Rice LW, et al. Clear cell carcinoma of the ovary: a distinct histologic type with poor prognosis and resistance to platinum-based chemotherapy in stage III disease. Gynecol Oncol. 1996; 60:412–417.15. Pectasides D, Fountzilas G, Aravantinos G, Kalofonos C, Efstathiou H, Farmakis D, et al. Advanced stage clear-cell epithelial ovarian cancer: the Hellenic Cooperative Oncology Group experience. Gynecol Oncol. 2006; 102:285–291.16. Hess V, A'Hern R, Nasiri N, King DM, Blake PR, Barton DP, et al. Mucinous epithelial ovarian cancer: a separate entity requiring specific treatment. J Clin Oncol. 2004; 22:1040–1044.17. Suzuki S, Kajiyama H, Shibata K, Ino K, Nawa A, Sakakibara K, et al. Is there any association between retroperitoneal lymphadenectomy and survival benefit in ovarian clear cell carcinoma patients? Ann Oncol. 2008; 19:1284–1287.18. Rustin GJ, Vergote I, Eisenhauer E, Pujade-Lauraine E, Quinn M, Thigpen T, et al. Definitions for response and progression in ovarian cancer clinical trials incorporating RECIST 1.1 and CA 125 agreed by the Gynecological Cancer Intergroup (GCIG). Int J Gynecol Cancer. 2011; 21:419–423.19. Parmar MK, Ledermann JA, Colombo N, du Bois A, Delaloye JF, Kristensen GB, et al. Paclitaxel plus platinum-based chemotherapy versus conventional platinum-based chemotherapy in women with relapsed ovarian cancer: the ICON4/AGO-OVAR-2.2 trial. Lancet. 2003; 361:2099–2106.20. Kommoss F, Kommoss S, Schmidt D, Trunk MJ, Pfisterer J, du Bois A, et al. Survival benefit for patients with advanced-stage transitional cell carcinomas vs. other subtypes of ovarian carcinoma after chemotherapy with platinum and paclitaxel. Gynecol Oncol. 2005; 97:195–199.21. Omura GA, Brady MF, Homesley HD, Yordan E, Major FJ, Buchsbaum HJ, et al. Long-term follow-up and prognostic factor analysis in advanced ovarian carcinoma: the Gynecologic Oncology Group experience. J Clin Oncol. 1991; 9:1138–1150.22. Winter WE 3rd, Maxwell GL, Tian C, Carlson JW, Ozols RF, Rose PG, et al. Prognostic factors for stage III epithelial ovarian cancer: a Gynecologic Oncology Group Study. J Clin Oncol. 2007; 25:3621–3627.23. Chaitin BA, Gershenson DM, Evans HL. Mucinous tumors of the ovary: a clinicopathologic study of 70 cases. Cancer. 1985; 55:1958–1962.24. Takano M, Sugiyama T, Yaegashi N, Sakuma M, Suzuki M, Saga Y, et al. Low response rate of second-line chemotherapy for recurrent or refractory clear cell carcinoma of the ovary: a retrospective Japan Clear Cell Carcinoma Study. Int J Gynecol Cancer. 2008; 18:937–942.25. Levy MH, Back A, Benedetti C, Billings JA, Block S, Boston B, et al. NCCN clinical practice guidelines in oncology: palliative care. J Natl Compr Canc Netw. 2009; 7:436–473.26. McCartney CF, Larson DB. Quality of life in patients with gynecologic cancer. Cancer. 1987; 60:8 Suppl. 2129–2136.27. Zang RY, Harter P, Chi DS, Sehouli J, Jiang R, Trope CG, et al. Predictors of survival in patients with recurrent ovarian cancer undergoing secondary cytoreductive surgery based on the pooled analysis of an international collaborative cohort. Br J Cancer. 2011; 105:890–896.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Impact on survival with adjuvant radiotherapy for clear cell, mucinous, and endometriod ovarian cancer: the SEER experience from 2004 to 2011
- Evaluation of secondary cytoreduction surgery in platinum-resistant ovarian cancer patients within three-line recurrent: a multicenter, randomized controlled study
- Outcomes of Non-High Grade Serous Carcinoma after Neoadjuvant Chemotherapy for Advanced-Stage Ovarian Cancer: Single-Institution Experience
- The role of topotecan as second-line chemotherapy in patients with recurrent epithelial ovarian cancer
- A case of advanced ovarian cancer which was treated with topotecan after taxol-cisplatin treatment failed