Clin Endosc.
2013 Jan;46(1):7-23. 10.5946/ce.2013.46.1.7.
Advance in Photosensitizers and Light Delivery for Photodynamic Therapy
- Affiliations
-
- 1PDT Research Institute, Inje University School of Nano System Engineering, Gimhae, Korea. ykshim@inje.ac.kr
- KMID: 1792635
- DOI: http://doi.org/10.5946/ce.2013.46.1.7
Abstract
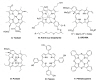
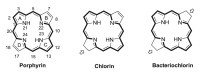
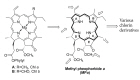
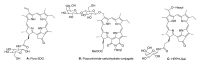
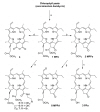
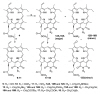
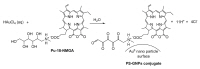
- The brief history of photodynamic therapy (PDT) research has been focused on photosensitizers (PSs) and light delivery was introduced recently. The appropriate PSs were developed from the first generation PS Photofrin (QLT) to the second (chlorins or bacteriochlorins derivatives) and third (conjugated PSs on carrier) generations PSs to overcome undesired disadvantages, and to increase selective tumor accumulation and excellent targeting. For the synthesis of new chlorin PSs chlorophyll a is isolated from natural plants or algae, and converted to methyl pheophorbide a (MPa) as an important starting material for further synthesis. MPa has various active functional groups easily modified for the preparation of different kinds of PSs, such as methyl pyropheophorbide a, purpurin-18, purpurinimide, and chlorin e6 derivatives. Combination therapy, such as chemotherapy and photothermal therapy with PDT, is shortly described here. Advanced light delivery system is shown to establish successful clinical applications of PDT. Phtodynamic efficiency of the PSs with light delivery was investigated in vitro and/or in vivo.
Keyword
MeSH Terms
Figure
Reference
-
1. Szaciłowski K, Macyk W, Drzewiecka-Matuszek A, Brindell M, Stochel G. Bioinorganic photochemistry: frontiers and mechanisms. Chem Rev. 2005; 105:2647–2694. PMID: 15941225.
Article2. Pandey RK, Zheng G. Kadish KM, Smith KM, Guilard R, editors. Porphyrins as photosensitizers in photodynamic therapy. The Porphyrin Handbook. 2000. Boston: Academic Press;p. 157–230.3. Zuluaga MF, Lange N. Combination of photodynamic therapy with anti-cancer agents. Curr Med Chem. 2008; 15:1655–1673. PMID: 18673216.
Article4. Juarranz A, Jaén P, Sanz-Rodríguez F, Cuevas J, González S. Photodynamic therapy of cancer. Basic principles and applications. Clin Transl Oncol. 2008; 10:148–154. PMID: 18321817.
Article5. Dougherty TJ, Henderson BW, Schwartz S, Winkelman JW, Lipson RL. Henderson BW, Dougherty TJ, editors. Historical perspective. Photodynamic Therapy: Basic Principles and Clinical Applications. 1992. New York: M. Dekker;p. 1–15.
Article6. Dougherty TJ, Grindey GB, Fiel R, Weishaupt KR, Boyle DG. Photoradiation therapy. II. Cure of animal tumors with hematoporphyrin and light. J Natl Cancer Inst. 1975; 55:115–121. PMID: 1159805.7. Orenstein A, Kostenich G, Roitman L, et al. A comparative study of tissue distribution and photodynamic therapy selectivity of chlorin e6, photofrin II and ALA-induced protoporphyrin IX in a colon carcinoma model. Br J Cancer. 1996; 73:937–944. PMID: 8611429.
Article8. Spikes JD. Chlorins as photosensitizers in biology and medicine. J Photochem Photobiol B. 1990; 6:259–274. PMID: 2120404.9. Baas P, van Mansom I, van Tinteren H, Stewart FA, van Zandwijk N. Effect of N-acetylcysteine on photofrin-induced skin photosensitivity in patients. Lasers Surg Med. 1995; 16:359–367. PMID: 7651057.10. Kessel D, Thompson P. Purification and analysis of hematoporphyrin and hematoporphyrin derivative by gel exclusion and reverse-phase chromatography. Photochem Photobiol. 1987; 46:1023–1025. PMID: 2964039.
Article11. Castanoa AP, Demidova TN, Hamblin MR. Mechanisms in photodynamic therapy: part one: photosensitizers, photochemistry and cellular localization. Photodiagnosis Photodyn Ther. 2004; 1:279–293.12. Sternberg ED, Dolphin D, Brückner C. Porphyrin-based photosensitizers for use in photodynamic therapy. Tetrahedron. 1998; 54:4151–4202.
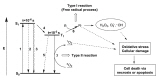
Article13. Foote CS. Definition of type I and type II photosensitized oxidation. Photochem Photobiol. 1991; 54:659. PMID: 1798741.
Article14. Gál D. Effect of photosensitizers in chemical and biological processes: the MTO mechanism in photodynamic therapy. Biochem Biophys Res Commun. 1992; 186:1032–1036. PMID: 1497636.
Article15. Vidóczy T, Elzemzam S, Gál D. Physico-chemical modeling of the role of free radicals in photo-dynamic therapy. I. utilization of quantum yield data of singlet oxygen formation for the study of the interaction between excited photosensitizer and stable free radicals. Biochem Biophys Res Commun. 1992; 189:1548–1552. PMID: 1482365.
Article16. Gál D, Kriska T, Maltseva E. In vivo experimental studies on the role of free radicals in photodynamic therapy. III. photodynamic effect on free radicals generated in cell cultures. Biochem Biophys Res Commun. 1997; 233:173–176. PMID: 9144417.17. Figge FH, Weiland GS, Manganiello LO. Cancer detection and therapy: affinity of neoplastic, embryonic, and traumatized tissues for porphyrins and metalloporphyrins. Proc Soc Exp Biol Med. 1948; 68:640. PMID: 18884315.
Article18. Hamblin MR, Newman EL. On the mechanism of the tumour-localising effect in photodynamic therapy. J Photochem Photobiol B. 1994; 23:3–8. PMID: 8021748.19. Boyle RW, Dolphin D. Structure and biodistribution relationships of photodynamic sensitizers. Photochem Photobiol. 1996; 64:469–485. PMID: 8806226.
Article20. Castano AP, Demidova TN, Hamblin MR. Mechanisms in photodynamic therapy: part three: photosensitizer pharmacokinetics, biodistribution, tumor localization and modes of tumor destruction. Photodiagnosis Photodyn Ther. 2005; 2:91–106.21. Yuan F, Leunig M, Berk DA, Jain RK. Microvascular permeability of albumin, vascular surface area, and vascular volume measured in human adenocarcinoma LS174T using dorsal chamber in SCID mice. Microvasc Res. 1993; 45:269–289. PMID: 8321142.
Article22. Pottier R, Kennedy JC. The possible role of ionic species in selective biodistribution of photochemotherapeutic agents toward neoplastic tissue. J Photochem Photobiol B. 1990; 8:1–16. PMID: 2127428.23. Allison BA, Pritchard PH, Levy JG. Evidence for low-density lipoprotein receptor-mediated uptake of benzoporphyrin derivative. Br J Cancer. 1994; 69:833–839. PMID: 8180011.
Article24. Roberts WG, Hasan T. Role of neovasculature and vascular permeability on the tumor retention of photodynamic agents. Cancer Res. 1992; 52:924–930. PMID: 1371089.25. Korbelik M, Krosl G. Photofrin accumulation in malignant and host cell populations of a murine fibrosarcoma. Photochem Photobiol. 1995; 62:162–168. PMID: 7638261.
Article26. Wendler G, Lindemann P, Lacapère JJ, Papadopoulos V. Protoporphyrin IX binding and transport by recombinant mouse PBR. Biochem Biophys Res Commun. 2003; 311:847–852. PMID: 14623258.
Article27. Wilson BC, Jeeves WP, Lowe DM. In vivo and post mortem measurements of the attenuation spectra of light in mammalian tissues. Photochem Photobiol. 1985; 42:153–162. PMID: 4048297.
Article28. Szacilowski K, Macyk W, Drzewiecka-Matuszek A, Brindell M, Stochel G. Bioinorganic photochemistry: frontiers and mechanisms. Chem Rev. 2005; 105:2647–2694. PMID: 15941225.
Article29. O'Connor AE, Gallagher WM, Byrne AT. Porphyrin and nonporphyrin photosensitizers in oncology: preclinical and clinical advances in photodynamic therapy. Photochem Photobiol. 2009; 85:1053–1074. PMID: 19682322.30. Allison RR, Sibata CH. Oncologic photodynamic therapy photosensitizers: a clinical review. Photodiagnosis Photodyn Ther. 2010; 7:61–75. PMID: 20510301.
Article31. Brandis AS, Salomon Y, Scherz A. Grimm B, Porra RJ, Rüdiger W, Scheer H, editors. Chlorophyll sensitizers in photodynamic therapy. Chlorophylls and Bacteriochlorophylls: Biochemistry, Biophysics, Functions and Applications. 2006. Dordrecht: Springer;p. 461–483.
Article32. Pandey RK, Shiau FY, Sumlin AB, Dougherty TJ, Smith KM. Structure/activity relationships among photosensitizers related to pheophorbides and bacteriopheophorbides. Bioorg Med Chem Lett. 1992; 2:491–496.
Article33. Bellnier DA, Henderson BW, Pandey RK, Potter WR, Dougherty TJ. Murine pharmacokinetics and antitumor efficacy of the photodynamic sensitizer 2-[1-hexyloxyethyl]-2-devinyl pyropheophorbide-a. J Photochem Photobiol B. 1993; 20:55–61. PMID: 8229470.
Article34. Pandey RK, Sumlin AB, Constantine S, et al. Alkyl ether analogs of chlorophyll-a derivatives: part 1. synthesis, photophysical properties and photodynamic efficacy. Photochem Photobiol. 1996; 64:194–204. PMID: 8787014.
Article35. Lobel J, MacDonald IJ, Ciesielski MJ, et al. 2-[1-hexyloxyethyl]-2-devinyl pyropheophorbide-a (HPPH) in a nude rat glioma model: implications for photodynamic therapy. Lasers Surg Med. 2001; 29:397–405. PMID: 11891727.
Article36. Pandey RK, Herman CK. Shedding some light on tumours. Chem Ind. 1998; 18:739–743.37. Clinicaltrials.gov. Photodynamic Therapy Using HPPH in Treating Patients With Advanced Non-Small Cell Lung Cancer That Blocks the Air Passages [Internet]. 2013. updated 2013 Jan 10; cited 2013 Jan 21. Bethesda: The U.S. National Institutes of Health;Available from: http://clinicaltrials.gov/ct2/show/NCT00528775.38. Matsumura H, Akimoto J, Haraoka J, Aizawa K. Uptake and retention of the photosensitizer mono-L-asparthyl chlorine e6 in experimental malignant glioma. Lasers Med Sci. 2008; 23:237–245. PMID: 17703335.
Article39. Kennedy JC, Nadeau P, Petryka ZJ, Pottier RH, Weagle G. Clearance times of porphyrin derivatives from mice as measured by in vivo fluorescence spectroscopy. Photochem Photobiol. 1992; 55:729–734. PMID: 1528987.
Article40. Ando T, Irie K, Koshimizu K, et al. Synthesis, physicochemical properties and photocytotoxicity of five new δ-substituted chlorin e6 derivatives. Tetrahedron. 1990; 46:5921–5930.
Article41. Kato H, Furukawa K, Sato M, et al. Phase II clinical study of photodynamic therapy using mono-L-aspartyl chlorin e6 and diode laser for early superficial squamous cell carcinoma of the lung. Lung Cancer. 2003; 42:103–111. PMID: 14512194.
Article42. Kujundzić M, Vogl TJ, Stimac D, et al. A Phase II safety and effect on time to tumor progression study of intratumoral light infusion technology using talaporfin sodium in patients with metastatic colorectal cancer. J Surg Oncol. 2007; 96:518–524. PMID: 17671969.43. Hoober JK, Sery TW, Yamamoto N. Photodynamic sensitizers from chlorophyll: purpurin-18 and chlorin p6. Photochem Photobiol. 1988; 48:579–582. PMID: 3241831.44. Zenkevich E, Sagun E, Knyukshto V, et al. Photophysical and photochemical properties of potential porphyrin and chlorin photosensitizers for PDT. J Photochem Photobiol B. 1996; 33:171–180.
Article45. Zheng G, Aoudia M, Lee D, et al. Chlorin-based symmetrical and unsymmetrical dimers with amide linkages: effect of the substituents on photodynamic and photophysical properties. J Chem Soc Perkin Trans 1. 2000; (24):3113–3121.
Article46. Smith KM, Lee SJ, Shiau FY, Pandey RK, Jagerovic N. Spinelli P, Dal Fante M, Marchesini R, editors. Syntheses of chlorin and bacteriochlorin-type photosensitizers for photodynamic therapy. Photodynamic Therapy and Biomedical Lasers. 1992. Amsterdam: Elsevier Science Publishers B.V.;p. 769–773.47. Pandey RK, Goswami LN, Chen Y, et al. Nature: a rich source for developing multifunctional agents. Tumor-imaging and photodynamic therapy. Lasers Surg Med. 2006; 38:445–467. PMID: 16788930.
Article48. Zheng G, Potter WR, Sumlin A, Dougherty TJ, Pandey RK. Photosensitizers related to purpurin-18-N-alkylimides: a comparative in vivo tumoricidal ability of ester versus amide functionalities. Bioorg Med Chem Lett. 2000; 10:123–127. PMID: 10673094.49. Pandey RK, James NS, Chen Y, Missert J, Sajjad M. Bifunctional agents for imaging and therapy. Methods Mol Biol. 2010; 635:223–259. PMID: 20552351.
Article50. Pandey RK, Constantine S, Tsuchida T, et al. Synthesis, photophysical properties, in vivo photosensitizing efficacy, and human serum albumin binding properties of some novel bacteriochlorins. J Med Chem. 1997; 40:2770–2779. PMID: 9276023.
Article51. Mettath S, Shibata M, Alderfer JL, et al. Synthesis and spectroscopic properties of novel benzochlorins derived from chlorophyll a. J Org Chem. 1998; 63:1646–1656.
Article52. Li G, Graham A, Chen Y, et al. Synthesis, comparative photosensitizing efficacy, human serum albumin (site II) binding ability, and intracellular localization characteristics of novel benzobacteriochlorins derived from vic-dihydroxybacteriochlorins. J Med Chem. 2003; 46:5349–5359. PMID: 14640543.
Article53. Zheng X, Pandey RK. Porphyrin-carbohydrate conjugates: impact of carbohydrate moieties in photodynamic therapy (PDT). Anticancer Agents Med Chem. 2008; 8:241–268. PMID: 18393785.
Article54. Shadidi M, Sioud M. Selective targeting of cancer cells using synthetic peptides. Drug Resist Updat. 2003; 6:363–371. PMID: 14744500.
Article55. Polo L, Valduga G, Jori G, Reddi E. Low-density lipoprotein receptors in the uptake of tumour photosensitizers by human and rat transformed fibroblasts. Int J Biochem Cell Biol. 2002; 34:10–23. PMID: 11733181.
Article56. Vrouenraets MB, Visser GW, Loup C, et al. Targeting of a hydrophilic photosensitizer by use of internalizing monoclonal antibodies: a new possibility for use in photodynamic therapy. Int J Cancer. 2000; 88:108–114. PMID: 10962447.
Article57. Chatterjee DK, Fong LS, Zhang Y. Nanoparticles in photodynamic therapy: an emerging paradigm. Adv Drug Deliv Rev. 2008; 60:1627–1637. PMID: 18930086.
Article58. Greco F, Vicent MJ. Polymer-drug conjugates: current status and future trends. Front Biosci. 2008; 13:2744–2756. PMID: 17981750.
Article59. Zhang M, Zhang Z, Blessington D, et al. Pyropheophorbide 2-deoxyglucosamide: a new photosensitizer targeting glucose transporters. Bioconjug Chem. 2003; 14:709–714. PMID: 12862422.
Article60. Pandey SK, Zheng X, Morgan J, et al. Purpurinimide carbohydrate conjugates: effect of the position of the carbohydrate moiety in photosensitizing efficacy. Mol Pharm. 2007; 4:448–464. PMID: 17373821.
Article61. Zheng X, Morgan J, Pandey SK, et al. Conjugation of 2-(1'-hexyloxyethyl)-2-devinylpyropheophorbide-a (HPPH) to carbohydrates changes its subcellular distribution and enhances photodynamic activity in vivo. J Med Chem. 2009; 52:4306–4318. PMID: 19507863.
Article62. Liu FT, Rabinovich GA. Galectins as modulators of tumour progression. Nat Rev Cancer. 2005; 5:29–41. PMID: 15630413.
Article63. Stefflova K, Li H, Chen J, Zheng G. Peptide-based pharmacomodulation of a cancer-targeted optical imaging and photodynamic therapy agent. Bioconjug Chem. 2007; 18:379–388. PMID: 17298029.
Article64. Chen J, Jarvi M, Lo PC, Stefflova K, Wilson BC, Zheng G. Using the singlet oxygen scavenging property of carotenoid in photodynamic molecular beacons to minimize photodamage to non-targeted cells. Photochem Photobiol Sci. 2007; 6:1311–1317. PMID: 18046487.
Article65. Ogura S, Yazaki K, Yamaguchi K, Kamachi T, Okura I. Localization of poly-L-lysine-photosensitizer conjugate in nucleus. J Control Release. 2005; 103:1–6. PMID: 15773061.
Article66. Tijerina M, Kopecková P, Kopecek J. Correlation of subcellular compartmentalization of HPMA copolymer-Mce6 conjugates with chemotherapeutic activity in human ovarian carcinoma cells. Pharm Res. 2003; 20:728–737. PMID: 12751627.67. Gariépy J. The use of Shiga-like toxin 1 in cancer therapy. Crit Rev Oncol Hematol. 2001; 39:99–106. PMID: 11418306.
Article68. Tarragó-Trani MT, Jiang S, Harich KC, Storrie B. Shiga-like toxin subunit B (SLTB)-enhanced delivery of chlorin e6 (Ce6) improves cell killing. Photochem Photobiol. 2006; 82:527–537. PMID: 16613509.
Article69. Zheng G, Li H, Zhang M, Lund-Katz S, Chance B, Glickson JD. Lowdensity lipoprotein reconstituted by pyropheophorbide cholesteryl oleate as target-specific photosensitizer. Bioconjug Chem. 2002; 13:392–396. PMID: 12009925.
Article70. Nikolaeva IA, Misharin AY, Ponomarev GV, Timofeev VP, Tkachev YV. Chlorin e6-cholesterol conjugate and its copper complex. Simple synthesis and entrapping in phospholipid vesicles. Bioorg Med Chem Lett. 2010; 20:2872–2875. PMID: 20356740.
Article71. Roy I, Ohulchanskyy TY, Pudavar HE, et al. Ceramic-based nanoparticles entrapping water-insoluble photosensitizing anticancer drugs: a novel drug-carrier system for photodynamic therapy. J Am Chem Soc. 2003; 125:7860–7865. PMID: 12823004.
Article72. Ohulchanskyy TY, Roy I, Goswami LN, et al. Organically modified silica nanoparticles with covalently incorporated photosensitizer for photodynamic therapy of cancer. Nano Lett. 2007; 7:2835–2842. PMID: 17718587.
Article73. Cinteza LO, Ohulchanskyy TY, Sahoo Y, Bergey EJ, Pandey RK, Prasad PN. Diacyllipid micelle-based nanocarrier for magnetically guided delivery of drugs in photodynamic therapy. Mol Pharm. 2006; 3:415–423. PMID: 16889435.
Article74. Hynninen PH. Scheer H, editor. Chemistry of chlorophylls: Modifications. Chlorophylls. 1991. Boca Raton: CRC Press;p. 145–209.75. Smith KM, Goff DA, Simpson DJ. The meso substitution of chlorophyll derivatives: direct route for transformation of bacteriopheophorbides d into bacteriopheophorbides c. J Am Chem Soc. 1985; 107:4946–4954.
Article76. Falk JE, Smith KM. Porphyrins and Metalloporphyrins. 1975. Amsterdam: Elsevier.77. Tamiaki H, Amakawa M, Shimono Y, Tanikaga R, Holzwarth AR, Schaffner K. Synthetic zinc and magnesium chlorin aggregates as models for supramolecular antenna complexes in chlorosomes of green photosynthetic bacteria. Photochem Photobiol. 1996; 63:92–99.
Article78. Kozyrev AN, Zheng G, Lazarou E, Dougherty TJ, Smith KM, Pandey RK. Syntheses of emeraldin and purpurin-18 analogs as target-specific photosensitizers for photodynamic therapy. Tetrahedron Lett. 1997; 38:3335–3338.
Article79. Rungta A, Zheng G, Missert JR, Potter WR, Dougherty TJ, Pandey RK. Purpurinimides as photosensitizers: effect of the presence and position of the substituents in the in vivo photodynamic efficacy. Bioorg Med Chem Lett. 2000; 10:1463–1466. PMID: 10888333.
Article80. Spikes JD, Bommer JC. Photobleaching of mono-L-aspartyl chlorin e6 (NPe6): a candidate sensitizer for the photodynamic therapy of tumors. Photochem Photobiol. 1993; 58:346–350. PMID: 8234467.81. Li G, Slansky A, Dobhal MP, et al. Chlorophyll-a analogues conjugated with aminobenzyl-DTPA as potential bifunctional agents for magnetic resonance imaging and photodynamic therapy. Bioconjug Chem. 2005; 16:32–42. PMID: 15656573.
Article82. Weiss C. Dolphin D, editor. Electronic absorption spectra of chlorophylls. The Porphyrins. 1978. New York: Academic Press;p. 211–223.
Article83. Hutter E, Fendler JH. Exploitation of localized surface plasmon resonance. Adv Mater. 2004; 16:1685–1706.
Article84. Torchilin VP. Multifunctional nanocarriers. Adv Drug Deliv Rev. 2006; 58:1532–1555. PMID: 17092599.
Article85. Jain PK, Huang X, El-Sayed IH, El-Sayed MA. Noble metals on the nanoscale: optical and photothermal properties and some applications in imaging, sensing, biology, and medicine. Acc Chem Res. 2008; 41:1578–1586. PMID: 18447366.
Article86. Cheng Y, Meyers JD, Broome AM, Kenney ME, Basilion JP, Burda C. Deep penetration of a PDT drug into tumors by noncovalent drug-gold nanoparticle conjugates. J Am Chem Soc. 2011; 133:2583–2591. PMID: 21294543.
Article87. Giernoth R. Task-specific ionic liquids. Angew Chem Int Ed Engl. 2010; 49:2834–2839. PMID: 20229544.
Article88. Wasserscheid P, Welton T. Ionic Liquids in Synthesis. 2003. Weinheim: Wiley-VCH.89. Itoh H, Naka K, Chujo Y. Synthesis of gold nanoparticles modified with ionic liquid based on the imidazolium cation. J Am Chem Soc. 2004; 126:3026–3027. PMID: 15012113.
Article90. Kim KS, Choi S, Cha JH, Yeon SH, Lee H. Facile one-pot synthesis of gold nanoparticles using alcohol ionic liquids. J Mater Chem. 2006; 16:1315–1317.
Article91. Pegaz B, Debefve E, Borle F, et al. Preclinical evaluation of a novel water-soluble chlorin E6 derivative (BLC 1010) as photosensitizer for the closure of the neovessels. Photochem Photobiol. 2005; 81:1505–1510. PMID: 15960590.
Article92. Sengee GI, Badraa N, Shim YK. Synthesis and biological evaluation of new imidazolium and piperazinium salts of pyropheophorbide-a for photodynamic cancer therapy. Int J Mol Sci. 2008; 9:1407–1415. PMID: 19325811.
Article93. Pissuwan D, Niidome T, Cortie MB. The forthcoming applications of gold nanoparticles in drug and gene delivery systems. J Control Release. 2011; 149:65–71. PMID: 20004222.
Article94. Demberelnyamba D, Ariunaa M, Shim YK. Newly synthesized water soluble cholinium-purpurin photosensitizers and their stabilized gold nanoparticles as promising anticancer agents. Int J Mol Sci. 2008; 9:864–871. PMID: 19325790.
Article95. Lkhagvadulam B, Kim JH, Yoon I, Shim YK. Synthesis and photodynamic activities of novel water soluble purpurin-18-N-methyl-D-glucamine photosensitizer and its gold nanoparticles conjugate. J Porphyr Phthalocyanines. 2012; 16:331–340.
Article96. Lkhagvadulam B, Kim JH, Yoon I, Shim YK. Size-dependent photodynamic activity of gold nanoparticles conjugate of water soluble purpurin-18-N-methyl-D-glucamine. BioMed Res Int. 2013; 2013:720579.
Article97. Gamaleia NF, Shishko ED, Dolinsky GA, Shcherbakov AB, Usatenko AV, Kholin VV. Photodynamic activity of hematoporphyrin conjugates with gold nanoparticles: experiments in vitro. Exp Oncol. 2010; 32:44–47. PMID: 20332758.98. Lottner C, Bart KC, Bernhardt G, Brunner H. Soluble tetraarylporphyrin-platinum conjugates as cytotoxic and phototoxic antitumor agents. J Med Chem. 2002; 45:2079–2089. PMID: 11985475.
Article99. Lottner C, Bart KC, Bernhardt G, Brunner H. Hematoporphyrin-derived soluble porphyrin-platinum conjugates with combined cytotoxic and phototoxic antitumor activity. J Med Chem. 2002; 45:2064–2078. PMID: 11985474.
Article100. Lottner C, Knuechel R, Bernhardt G, Brunner H. Combined chemotherapeutic and photodynamic treatment on human bladder cells by hematoporphyrin-platinum(II) conjugates. Cancer Lett. 2004; 203:171–180. PMID: 14732225.
Article101. Hongrapipat J, Kopecková P, Liu J, Prakongpan S, Kopecek J. Combination chemotherapy and photodynamic therapy with fab' fragment targeted HPMA copolymer conjugates in human ovarian carcinoma cells. Mol Pharm. 2008; 5:696–709. PMID: 18729468.
Article102. Frank J, Flaccus A, Schwarz C, Lambert C, Biesalski HK. Ascorbic acid suppresses cell death in rat DS-sarcoma cancer cells induced by 5-aminolevulinic acid-based photodynamic therapy. Free Radic Biol Med. 2006; 40:827–836. PMID: 16520235.
Article103. Jakus J, Farkas O. Photosensitizers and antioxidants: a way to new drugs? Photochem Photobiol Sci. 2005; 4:694–698. PMID: 16121279.
Article104. Kaliya OL, Lukyanets EA, Vorozhtsov GN. Catalysis and photocatalysis by phthalocyanines for technology, ecology and medicine. J Porphyr Phthalocyanines. 1999; 3:592–610.
Article105. Shevchuk I, Chekulayev V, Chekulayeva L. Enhancement of the efficiency of photodynamic therapy of tumours by t-butyl-4-hydroxyanisole. J Photochem Photobiol B. 1998; 45:136–143. PMID: 9868804.
Article106. Raish M, Husain SZ, Bae SM, et al. Photodynamic therapy in combination with green tea polyphenol EGCG enhances antitumor efficacy in human papillomavirus 16 (E6/E7) immortalized tumor cells. J Appl Res. 2010; 10:58–67.107. Melnikova V, Bezdetnaya L, Belitchenko I, Potapenko A, Merlin JL, Guillemin F. Meta-tetra(hydroxyphenyl)chlorin-sensitized photodynamic damage of cultured tumor and normal cells in the presence of high concentrations of alpha-tocopherol. Cancer Lett. 1999; 139:89–95. PMID: 10408914.108. Melnikova VO, Bezdetnaya LN, Brault D, Potapenko AY, Guillemin F. Enhancement of meta-tetrahydroxyphenylchlorin-sensitized photodynamic treatment on human tumor xenografts using a water-soluble vitamin E analogue, Trolox. Int J Cancer. 2000; 88:798–803. PMID: 11072251.
Article109. Kriska T, Jakus J, Keszler A, Vanyur R, Nemeth A, Gal D. Ehrenberg B, Berg K, editors. Type III photosensitization: attempt for quantification and a way toward new sensitizers. Photochemotherapy of Cancer and Other Diseases. 1999. Vol. 3563. [place unknown]: SPIE Proceedings;p. 11–17.110. Ashkenazi H, Nitzan Y, Gál D. Photodynamic effects of antioxidant substituted porphyrin photosensitizers on gram-positive and -negative bacterial. Photochem Photobiol. 2003; 77:186–191. PMID: 12785058.111. Pankhurst QA, Connolly J, Jones SK, Dobson J. Applications of magnetic nanoparticles in biomedicine. J Phys D Appl Phys. 2003; 36:R167–R181.
Article112. Park JH, von Maltzahn G, Ong LL, et al. Cooperative nanoparticles for tumor detection and photothermally triggered drug delivery. Adv Mater. 2010; 22:880–885. PMID: 20217810.
Article113. Weissleder R. A clearer vision for in vivo imaging. Nat Biotechnol. 2001; 19:316–317. PMID: 11283581.
Article114. Lee J, Yang J, Ko H, et al. Multifunctional magnetic gold nanocomposites: human epithelial cancer detection via magnetic resonance imaging and localized synchronous therapy. Adv Funct Mater. 2008; 18:258–264.
Article115. Durr NJ, Larson T, Smith DK, Korgel BA, Sokolov K, Ben-Yakar A. Two-photon luminescence imaging of cancer cells using molecularly targeted gold nanorods. Nano Lett. 2007; 7:941–945. PMID: 17335272.
Article116. Moon HK, Lee SH, Choi HC. In vivo near-infrared mediated tumor destruction by photothermal effect of carbon nanotubes. ACS Nano. 2009; 3:3707–3713. PMID: 19877694.
Article117. Melancon MP, Elliott AM, Shetty A, Huang Q, Stafford RJ, Li C. Nearinfrared light modulated photothermal effect increases vascular perfusion and enhances polymeric drug delivery. J Control Release. 2011; 156:265–272. PMID: 21763373.
Article118. Ke H, Wang J, Dai Z, et al. Gold-nanoshelled microcapsules: a theranostic agent for ultrasound contrast imaging and photothermal therapy. Angew Chem Int Ed Engl. 2011; 50:3017–3021. PMID: 21404389.
Article119. Yang J, Choi J, Bang D, et al. Convertible organic nanoparticles for near-infrared photothermal ablation of cancer cells. Angew Chem Int Ed Engl. 2011; 50:441–444. PMID: 21132823.
Article120. Passos SK, Tedesco AC, Eid DR, Lacava ZG. Bowen disease treated with PDT using ALA in nanostructured vehicle and two light deliveries: a case report. J Am Acad Dermatol. 2011; 64(2 Suppl 1):AB141.121. Kruijt B, van der, Sterenborg HJ, Amelink A, Robinson DJ. A dedicated applicator for light delivery and monitoring of PDT of intra-anal intraepithelial neoplasia. Photodiagnosis Photodyn Ther. 2010; 7:3–9. PMID: 20230986.
Article122. Mang TS. Light sources and delivery devices for photodynamic therapy. Photodiagnosis Photodyn Ther. 2009; 6:147.
Article123. Mang TS. Lasers and light sources for PDT: past, present and future. Photodiagnosis Photodyn Ther. 2004; 1:43–48.
Article124. Panjehpour M, Overholt BF, Haydek JM. Light sources and delivery devices for photodynamic therapy in the gastrointestinal tract. Gastrointest Endosc Clin N Am. 2000; 10:513–532. PMID: 10899261.
Article125. Floyd TL. . Electric Circuit Fundamentals. 1991. 2nd ed. New York: Merrill.126. Star WM, Wilson BC, Patterson MS. Henderson BW, Dougherty TJ, editors. Light delivery and optical dosimetry in photodynamic therapy of solid tumors. Photodynamic Therapy: Basic Principles and Clinical Applications. 1992. New York: M. Dekker;p. 335–368.
Article127. Grossweiner LI. PDT light dosimetry revisited. J Photochem Photobiol B. 1997; 38:258–268. PMID: 9203389.
Article128. Tudge SH, Kaye AH, Hill JS. Modulation of light delivery in photodynamic therapy of brain tumours. J Clin Neurosci. 1999; 6:227–232. PMID: 18639156.
Article129. Madsen SJ, Sun CH, Tromberg BJ, Hirschberg H. Development of a novel indwelling balloon applicator for optimizing light delivery in photodynamic therapy. Lasers Surg Med. 2001; 29:406–412. PMID: 11891728.
Article130. Nyst HJ, van Veen RL, Tan IB, et al. Performance of a dedicated light delivery and dosimetry device for photodynamic therapy of nasopharyngeal carcinoma: phantom and volunteer experiments. Lasers Surg Med. 2007; 39:647–653. PMID: 17886277.
Article131. Kostenich G, Orenstein A, Roitman L, Malik Z, Ehrenberg B. In vivo photodynamic therapy with the new near-IR absorbing water soluble photosensitizer lutetium texaphyrin and a high intensity pulsed light delivery system. J Photochem Photobiol B. 1997; 39:36–42. PMID: 9210320.
Article132. Lapchak PA, Salgado KF, Chao CH, Zivin JA. Transcranial near-infrared light therapy improves motor function following embolic strokes in rabbits: an extended therapeutic window study using continuous and pulse frequency delivery modes. Neuroscience. 2007; 148:907–914. PMID: 17693028.
Article133. Usuda J, Ichinose S, Ishizumi T, et al. Outcome of photodynamic therapy using NPe6 for bronchogenic carcinomas in central airways >1.0 cm in diameter. Clin Cancer Res. 2010; 16:2198–2204. PMID: 20332318.134. Cassidy CM, Tunney MM, Magee ND, et al. Drug and light delivery strategies for photodynamic antimicrobial chemotherapy (PACT) of pulmonary pathogens: a pilot study. Photodiagnosis Photodyn Ther. 2011; 8:1–6. PMID: 21333929.
Article135. Friedberg JS, Skema C, Burdick J, Yodh AG, Carr SR, Culver JP. A novel technique for light delivery through branched or bent anatomic structures. J Thorac Cardiovasc Surg. 2003; 126:1963–1967. PMID: 14688713.
Article136. Wang LW, Li LB, Li ZS, Chen YK, Hetzel FW, Huang Z. Self-expandable metal stents and trans-stent light delivery: are metal stents and photodynamic therapy compatible. Lasers Surg Med. 2008; 40:651–659. PMID: 18951422.
Article137. Yang L, Wei Y, Xing D, Chen Q. Increasing the efficiency of photodynamic therapy by improved light delivery and oxygen supply using an anticoagulant in a solid tumor model. Lasers Surg Med. 2010; 42:671–679. PMID: 20740620.
Article138. Mitra S, Foster TH. Carbogen breathing significantly enhances the penetration of red light in murine tumours in vivo. Phys Med Biol. 2004; 49:1891–1904. PMID: 15214531.139. Xiao Z, Dickey D, Owen RJ, Tulip J, Moore R. Interstitial photodynamic therapy of the canine prostate using intra-arterial administration of photosensitizer and computerized pulsed light delivery. J Urol. 2007; 178:308–313. PMID: 17499802.
Article140. Dickey D, Barajas O, Brown K, Tulip J, Moore RB. Radiance modelling using the P3 approximation. Phys Med Biol. 1998; 43:3559–3570. PMID: 9869032.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Delivery of Photosensitizers for Photodynamic Therapy
- Photodynamic Therapy in Gastroenterology
- Photodynamic Diagnosis and Therapy for Peritoneal Carcinomatosis from Gastrointestinal Cancers: Status, Opportunities, and Challenges
- Photodynamic Therapy (PDT): PDT Mechanisms
- Topical Photodynamic Therapy for Treatment of Actinic Keratosis Using Light-Emitting Diode (LED) Device