Yonsei Med J.
2011 Mar;52(2):211-219. 10.3349/ymj.2011.52.2.211.
The Role of the Calcium and the Voltage Clocks in Sinoatrial Node Dysfunction
- Affiliations
-
- 1Division of Cardiology, Department of Medicine, Yonsei University College of Medicine, Seoul, Korea.
- 2The Krannert Institute of Cardiology and Division of Cardiology, Department of Medicine, Indiana University School of Medicine, Indianapolis, Indiana, USA. linsf@iupui.edu
- KMID: 1779654
- DOI: http://doi.org/10.3349/ymj.2011.52.2.211
Abstract
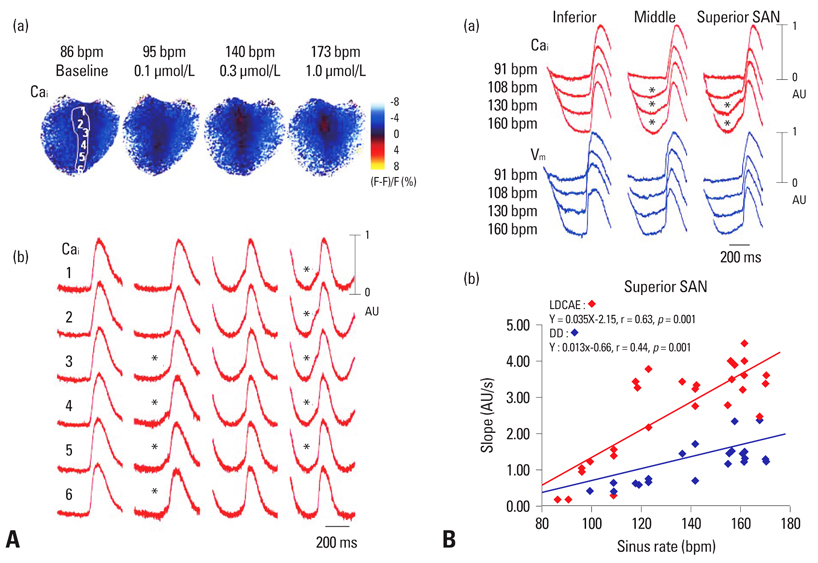
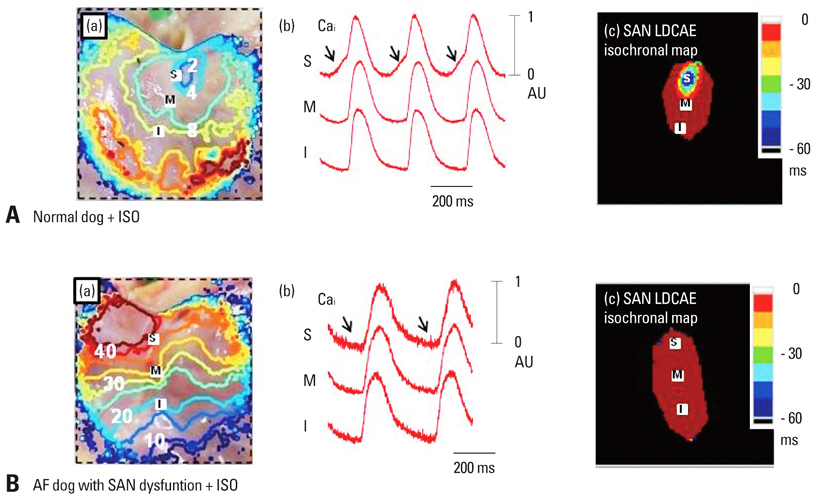
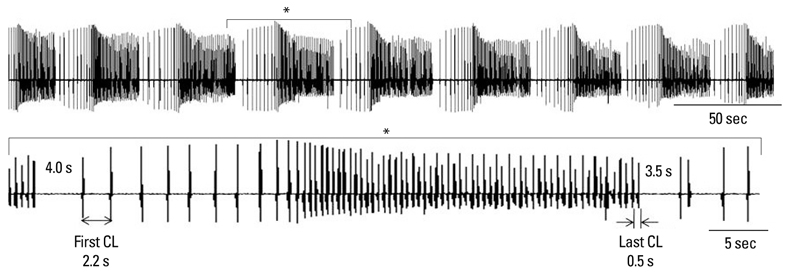
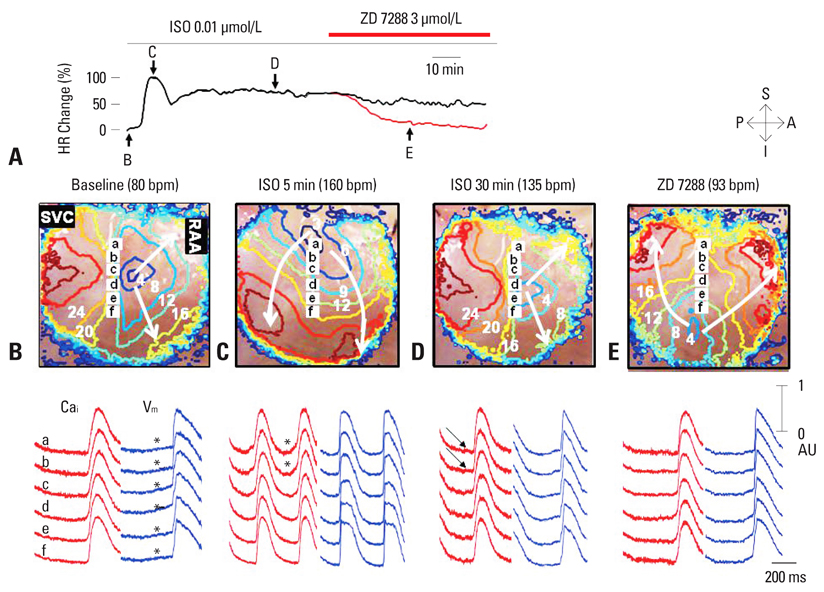
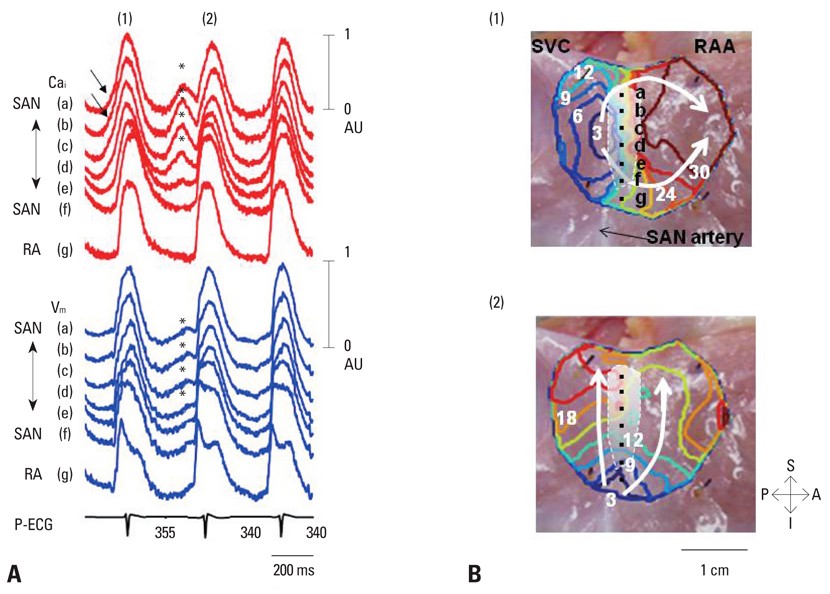
- Recent evidence indicates that the voltage clock (cyclic activation and deactivation of membrane ion channels) and Ca2+ clocks (rhythmic spontaneous sarcoplasmic reticulum Ca2+ release) jointly regulate sinoatrial node (SAN) automaticity. However, the relative importance of the voltage clock and Ca2+ clock for pacemaking was not revealed in sick sinus syndrome. Previously, we mapped the intracellular calcium (Cai) and membrane potentials of the normal intact SAN simultaneously using optical mapping in Langendorff-perfused canine right atrium. We demonstrated that the sinus rate increased and the leading pacemaker shifted to the superior SAN with robust late diastolic Cai elevation (LDCAE) during beta-adrenergic stimulation. We also showed that the LDCAE was caused by spontaneous diastolic sarcoplasmic reticulum (SR) Ca2+ release and was closely related to heart rate changes. In contrast, in pacing induced canine atrial fibrillation and SAN dysfunction models, Ca2+ clock of SAN was unresponsiveness to beta-adrenergic stimulation and caffeine. Ryanodine receptor 2 (RyR2) in SAN was down-regulated. Using the prolonged low dose isoproterenol together with funny current block, we produced a tachybradycardia model. In this model, chronically elevated sympathetic tone results in abnormal pacemaking hierarchy in the right atrium, including suppression of the superior SAN and enhanced pacemaking from ectopic sites. Finally, if the LDCAE was too small to trigger an action potential, then it induced only delayed afterdepolarization (DAD)-like diastolic depolarization (DD). The failure of DAD-like DD to consistently trigger a sinus beat is a novel mechanism of atrial arrhythmogenesis. We conclude that dysfunction of both the Ca2+ clock and the voltage clock are important in sick sinus syndrome.
MeSH Terms
Figure
Cited by 1 articles
-
Early Experience Using a Left Atrial Appendage Occlusion Device in Patients with Atrial Fibrillation
Yung Ly Kim, Boyoung Joung, Young Keun On, Chi Young Shim, Moon Hyoung Lee, Young-Hoon Kim, Hui-Nam Pak
Yonsei Med J. 2012;53(1):83-90. doi: 10.3349/ymj.2012.53.1.83.
Reference
-
1. Adán V, Crown LA. Diagnosis and treatment of sick sinus syndrome. Am Fam Physician. 2003. 67:1725–1732.2. Brown HF, Difrancesco D, Noble SJ. How does adrenaline accelerate the heart? Nature. 1979. 280:235–236.
Article3. Baruscotti M, Bucchi A, DiFrancesco D. Physiology and pharmacology of the cardiac pacemaker ("funny") current. Pharmacol Ther. 2005. 107:59–79.
Article4. DiFrancesco D. Pacemaker mechanisms in cardiac tissue. Annu Rev Physiol. 1993. 55:455–472.
Article5. DiFrancesco D. The pacemaker current (I(f)) plays an important role in regulating Sa node pacemaker activity. Cardiovasc Res. 1995. 30:307–308.
Article6. Milanesi R, Baruscotti M, Gnecchi-Ruscone T, DiFrancesco D. Familial sinus bradycardia associated with a mutation in the cardiac pacemaker channel. N Engl J Med. 2006. 354:151–157.
Article7. Bucchi A, Barbuti A, Baruscotti M, DiFrancesco D. Heart rate reduction via selective 'funny' channel blockers. Curr Opin Pharmacol. 2007. 7:208–213.
Article8. Nof E, Luria D, Brass D, Marek D, Lahat H, Reznik-Wolf H, et al. Point mutation in the HCN4 cardiac ion channel pore affecting synthesis, trafficking, and functional expression is associated with familial asymptomatic sinus bradycardia. Circulation. 2007. 116:463–470.
Article9. Rubenstein DS, Lipsius SL. Mechanisms of automaticity in subsidiary pacemakers from cat right atrium. Circ Res. 1989. 64:648–657.
Article10. Li J, Qu J, Nathan RD. Ionic basis of ryanodine's negative chronotropic effect on pacemaker cells isolated from the sinoatrial node. Am J Physiol. 1997. 273:H2481–H2489.11. Ju YK, Allen DG. Intracellular calcium and Na+-Ca2+ exchange current in isolated toad pacemaker cells. J Physiol. 1998. 508:153–166.
Article12. Hüser J, Blatter LA, Lipsius SL. Intracellular Ca2+ release contributes to automaticity in cat atrial pacemaker cells. J Physiol. 2000. 524(Pt 2):415–422.
Article13. Vinogradova TM, Bogdanov KY, Lakatta EG. Novel perspectives on the beating rate of the heart. Circ Res. 2002. 91:e3.
Article14. Maltsev VA, Vinogradova TM, Lakatta EG. The emergence of a general theory of the initiation and strength of the heartbeat. J Pharmacol Sci. 2006. 100:338–369.
Article15. Vinogradova TM, Lyashkov AE, Zhu W, Ruknudin AM, Sirenko S, Yang D, et al. High basal protein kinase A-dependent phosphorylation drives rhythmic internal Ca2+ store oscillations and spontaneous beating of cardiac pacemaker cells. Circ Res. 2006. 98:505–514.
Article16. Bhuiyan ZA, van den Berg MP, van Tintelen JP, Bink-Boelkens MT, Wiesfeld AC, Alders M, et al. Expanding spectrum of human TYR2-related disease: new electrocardiographic, structural, and genetic features. Circulation. 2007. 116:1569–1576.17. Verheijck EE, van Kempen MJ, Veereschild M, Lurvink J, Jongsma HJ, Bouman LN. Electrophysiological features of the mouse sinoatrial node in relation to connexin distribution. Cardiovasc Res. 2001. 52:40–50.
Article18. Lancaster MK, Jones SA, Harrison SM, Boyett MR. Intracellular Ca2+ and pacemaking within the rabbit sinoatrial node: Heterogeneity of role and control. J Physiol. 2004. 556:481–494.
Article19. Tellez JO, Dobrzynski H, Greener ID, Graham GM, Laing E, Honjo H, et al. Differential expression of ion channel transcripts in atrial muscle and sinoatrial node in rabbit. Circ Res. 2006. 99:1384–1393.
Article20. Boineau JP, Miller CB, Schuessler RB, Roeske WR, Autry LJ, Wylds AC, et al. Activation sequence and potential distribution maps demonstrating multicentric atrial impulse origin in dogs. Circ Res. 1984. 54:332–347.
Article21. Boineau JP, Schuessler RB, Mooney CR, Wylds AC, Miller CB, Hudson RD, et al. Multicentric origin of the atrial depolarization wave: the pacemaker complex. Relation to dynamics of atrial conduction, P-wave changes and heart rate control. Circulation. 1978. 58:1036–1048.
Article22. Schuessler RB, Boineau JP, Wylds AC, Hill DA, Miller CB, Roeske WR. Effect of canine cardiac nerves on heart rate, rhythm, and pacemaker location. Am J Physiol. 1986. 250:H630–H644.
Article23. Joung B, Tang L, Maruyama M, Han S, Chen Z, Stucky M, et al. Intracellular calcium dynamics and acceleration of sinus rhythm by beta-adrenergic stimulation. Circulation. 2009. 119:788–796.
Article24. Vinogradova TM, Bogdanov KY, Lakatta EG. beta-Adrenergic stimulation modulates ryanodine recepr Ca(2+) release during diastolic depolarization to accelerate pacemaker activity in rabbit sinoatrial nodal cells. Circ Res. 2002. 90:73–79.
Article25. Dobrzynski H, Boyett MR, Anderson RH. New insights into pacemaker activity: promoting understanding of sick sinus syndrome. Circulation. 2007. 115:1921–1932.26. Verkerk AO, Wilders R, Coronel R, Ravesloot JH, Verheijck EE. Ionic remodeling of sinoatrial node cells by heart failure. Circulation. 2003. 108:760–766.
Article27. Nattel S, Maguy A, Le Bouter S, Yeh YH. Arrhythmogenic ion-channel remodeling in the heart: heart failure, myocardial infarction, and atrial fibrillation. Physiol Rev. 2007. 87:425–456.
Article28. Van Wagoner DR, Pond AL, Lamorgese M, Rossie SS, McCarthy PM, Nerbonne JM. Atrial L-type Ca2+ currents and human atrial fibrillation. Circ Res. 1999. 85:428–436.29. Workman AJ, Kane KA, Rankin AC. The contribution of ionic currents to changes in refractoriness of human atrial myocytes associated with chronic atrial fibrillation. Cardiovasc Res. 2001. 52:226–235.
Article30. Van Wagoner DR, Pond AL, McCarthy PM, Trimmer JS, Nerbonne JM. Outward K+ current densities and Kv1.5 expression are reduced in chronic human atrial fibrillation. Circ Res. 1997. 80:772–781.
Article31. Yeh YH, Burstein B, Qi XY, Sakabe M, Chartier D, Comtois P, et al. Funny current downregulation and sinus node dysfunction associated with atrial tachyarrhythmia: a molecular basis for tachycardia-bradycardia syndrome. Circulation. 2009. 119:1576–1585.
Article32. Efimov IR, Fedorov VV, Joung B, Lin SF. Mapping cardiac pacemaker circuits: methodological puzzles of the sinoatrial node optical mapping. Circ Res. 2010. 106:255–271.33. Joung B, Lin SF, Chen Z, Antoun PS, Maruyama M, Han S, et al. Mechanisms of sinoatrial node dysfunction in a canine model of pacing-induced atrial fibrillation. Heart Rhythm. 2010. 7:88–95.
Article34. Sanders P, Kistler PM, Morton JB, Spence SJ, Kalman JM. Remodeling of sinus node function in patients with congestive heart failure: reduction in sinus node reserve. Circulation. 2004. 110:897–903.
Article35. Ogawa M, Zhou S, Tan AY, Song J, Gholmieh G, Fishbein MC, et al. Left stellate ganglion and vagal nerve activity and cardiac arrhythmias in ambulatory dogs with pacing-induced congestive heart failure. J Am Coll Cardiol. 2007. 50:335–343.
Article36. Zicha S, Fernández-Velasco M, Lonardo G, L'Heureux N, Nattel S. Sinus node dysfunction and hyperpolarization-activated (HCN) channel subunit remodeling in a canine heart failure model. Cardiovasc Res. 2005. 66:472–481.
Article37. Rubart M, Zipes DP. Mechanisms of sudden cardiac death. J Clin Invest. 2005. 115:2305–2315.
Article38. Sanders P, Berenfeld O, Hocini M, Jaïs P, Vaidyanathan R, Hsu LF, et al. Spectral analysis identifies sites of high-frequency activity maintaining atrial fibrillation in humans. Circulation. 2005. 112:789–797.
Article39. Joung B, Shinohara T, Zhang H, Kim D, Choi EK, On YK, et al. Tachybradycardia in the isolated canine right atrium induced by chronic sympathetic stimulation and pacemaker current inhibition. Am J Physiol Heart Circ Physiol. 2010. 299:H634–H642.
Article40. Bers DM. Calcium cycling and signaling in cardiac myocytes. Annu Rev Physiol. 2008. 70:23–49.
Article41. ter Keurs HE, Zhang YM, Miura M. Damage-induced arrhythmias: reversal of excitation-contraction coupling. Cardiovasc Res. 1998. 40:444–455.
Article42. Boyden PA, ter Keurs HE. Reverse excitation-contraction coupling: Ca2+ ions as initiators of arrhythmias. J Cardiovasc Electrophysiol. 2001. 12:382–385.
Article43. Lakatta EG, DiFrancesco D. What keeps us ticking: a funny current, a calcium clock, or both? J Mol Cell Cardiol. 2009. 47:157–170.
Article44. Maltsev VA, Lakatta EG. Synergism of coupled subsarcolemmal Ca2+ clocks and sarcolemmal voltage clocks confers robust and flexible pacemaker function in a novel pacemaker cell model. Am J Physiol Heart Circ Physiol. 2009. 296:H594–H615.45. Lakatta EG, Maltsev VA, Vinogradova TM. A coupled SYSTEM of intracellular Ca2+ clocks and surface membrane voltage clocks controls the timekeeping mechanism of the heart's pacemaker. Circ Res. 2010. 106:659–673.
Article46. Zhou Z, Lipsius SL. Na(+)-Ca2+ exchange current in latent pacemaker cells isolated from cat right atrium. J Physiol. 1993. 466:263–285.47. Hata T, Noda T, Nishimura M, Watanabe Y. The role of Ca2+ release from sarcoplasmic reticulum in the regulation of sinoatrial node automaticity. Heart Vessels. 1996. 11:234–241.
Article48. Rigg L, Terrar DA. Possible role of calcium release from the sarcoplasmic reticulum in pacemaking in guinea-pig sino-atrial node. Exp Physiol. 1996. 81:877–880.
Article49. Bogdanov KY, Vinogradova TM, Lakatta EG. Sinoatrial nodal cell ryanodine receptor and Na(+)-Ca(2+) exchanger: molecular partners in pacemaker regulation. Circ Res. 2001. 88:1254–1258.50. Shinohara T, Joung B, Kim D, Maruyama M, Luk HN, Chen PS, et al. Induction of atrial ectopic beats with calcium release inhibition: Local hierarchy of automaticity in the right atrium. Heart Rhythm. 2010. 7:110–116.
Article51. Joung B, Zhang H, Shinohara T, Maruyama M, Han S, Kim D, et al. Delayed Afterdepolarization in intact Canine Sinoatrial Node as a Novel Mechanism for Atrial Arrhythmia. J Cardiovasc Electrophysiol. 2010. (In press).
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Calcium and Voltage Clocks in Sinoatrial Node Automaticity
- Function and Dysfunction of Human Sinoatrial Node
- The Gradient Model of the Rabbit Sinoatrial Node
- RE: An Unusual Course of Right Coronary Artery Originating from Sinoatrial Node Artery
- Heart Rate Acceleration of a Subsidiary Pacemaker by beta-Adrenergic Stimulation