J Bacteriol Virol.
2014 Dec;44(4):342-351. 10.4167/jbv.2014.44.4.342.
The Management of Laboratory Biological Safety in Korea
- Affiliations
-
- 1Department of Microbiology, College of Medicine, Konkuk University, Seoul, Korea. wjjang@kku.ac.kr
- 2Institute of Glocal Disease Control, Konkuk University, Seoul, Korea.
- 3Internationl Vaccine Institute, Seoul, Korea.
- KMID: 1737323
- DOI: http://doi.org/10.4167/jbv.2014.44.4.342
Abstract
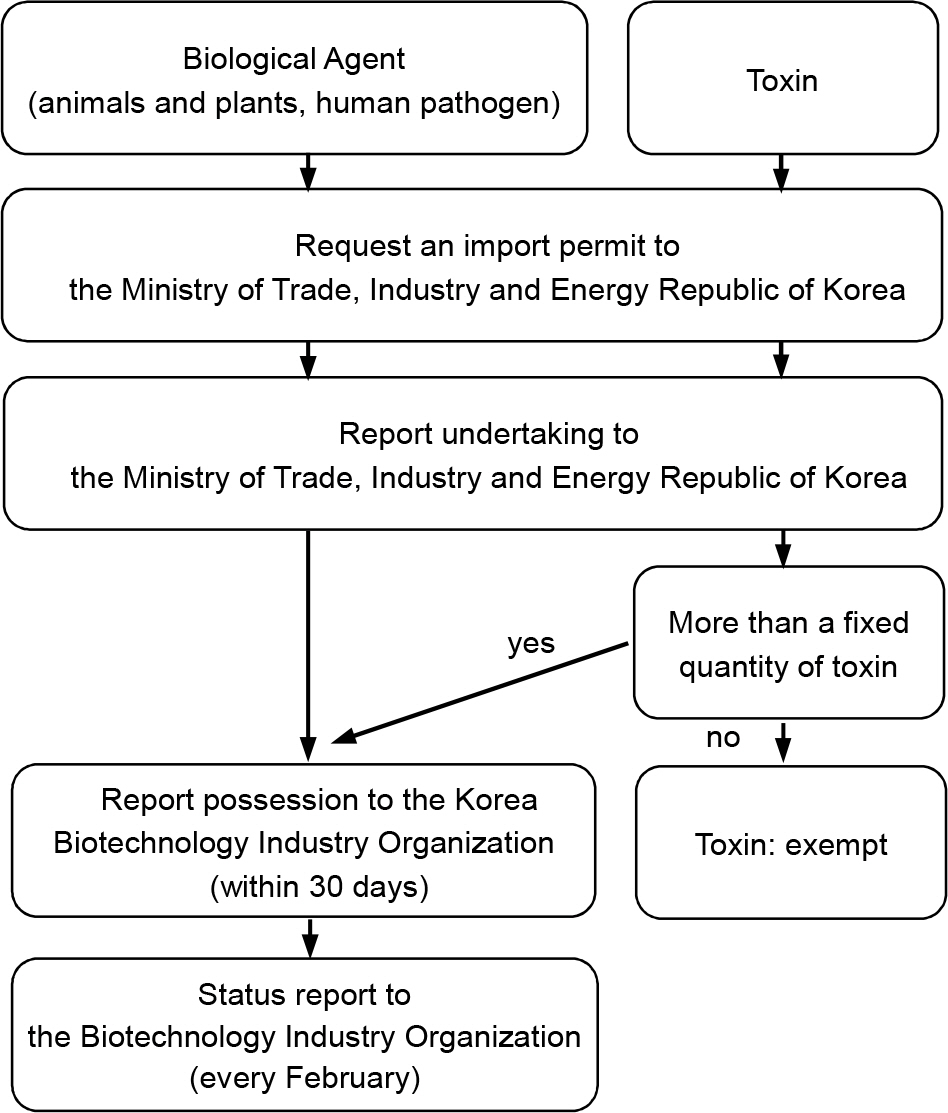
- The Laboratories engaging with microorganisms have some potential risks, depending on handling of pathogens. The safety management system to extend recognition of biosafety and biosecurity needs to be established and practiced in order to prevent hazard possibility and secure safety of researchers who deal with pathogen. As the importance and regulation of global biosafety and biosecurity tended to be intensified, various laws and institutions for the security of biosafety were established when the Act on implementation of Living Modified Organisms, an implantation act of Cartagena was enforced in Korea in 2008. In particular, the system of permission report for Living Modified Organism (LMO), Highly Dangerous Pathogens (HDPs) and Biological Weapons Convention (BWC), in order to prevent intended or non-intended leakage and occurrence of biohazard derived from pathogen and infectious substance and safely use them. The bioterrorism can be controlled and autonomous management of biosafety by agencies that handle pathogens can be also secured by practicing the various laws and institutions.
Keyword
MeSH Terms
Figure
Cited by 2 articles
-
Survey on the Status of Biosafety Awareness Among Domestic Biology Researchers
Kyung-Min Lee, Dong-Wook Kim, Yeon-Joo Choi, Kyung-Hee Park, Won-Jong Jang
J Bacteriol Virol. 2017;47(2):96-101. doi: 10.4167/jbv.2017.47.2.96.Emerging Infectious Diseases Require Biosafety Awareness and Procedures
Kyung-Min Lee, Won-Jong Jang, Yeon-Joo Choi, Kyung-Hee Park
J Bacteriol Virol. 2016;46(2):104-107. doi: 10.4167/jbv.2016.46.2.104.
Reference
-
1). Martini GA, Schmidt HA. Spermatogenic transmission of the “Marburg virus”. (Causes of “Marburg simian disease”). Klin Wochenschr. 1968; 46:398–400.2). Oliphant JW, Parker RR. Q fever; Three cases of laboratory infection. Public Health Rep. 1948; 63:1364–70.
Article3). Beeman EA. Q fever; An epidemiological note. Public Health Rep. 1950; 65:88–92.
Article4). Holmes GP, Hilliard JK, Klontz KC, Rupert AH, Schindler CM, Parrish E, et al. B virus (Herpesvirus simiae) infection in humans: epidemiologic investigation of a cluster. Ann Intern Med. 1990; 112:833–9.
Article5). Barton Behravesh C, Mody RK, Jungk J, Gaul L, Redd JT, Chen S, et al. 2008 outbreak of Salmonella Saintpaul infections associated with raw produce. N Engl J Med. 2011; 364:918–27.
Article6). Chong PY, Chui P, Ling AE, Franks TJ, Tai DY, Leo YS, et al. Analysis of deaths during the severe acute respiratory syndrome (SARS) epidemic in Singapore: challenges in determining a SARS diagnosis. Arch Pathol Lab Med. 2004; 128:195–204.
Article7). Koh D, Sng J. Lessons from the past: perspectives on severe acute respiratory syndrome. Asia Pac J Public Health. 2010; 22:32S–6S.
Article8). Jiang TJ, Zhou XZ, Zhao M, Zhou ZP, Jiang SC, Ye WH, et al. Analysis of severe acute respiratory syndrome in Beijing. Zhonghua Nei Ke Za Zhi. 2003; 42:369–72.9). Orellana C. Laboratory-acquired SARS raises worries on biosafety. Lancet Infect Dis. 2004; 4:64.
Article10). Lee KM, Nam HY, Shin SH, Choi MK, Choi YJ, Kwon SH, et al. A Survey on Laboratory Biosafety Status of Public Healthcare Centers in Korea. J Bacteriol Virol. 2013; 43:217–28.
Article11). U.S. Department of Health & Human Services, Center for Disease Control and Prevention, National Institutes of Health. Biosafety in Microbiological and Biomedical Laboratories (BMBL). 5th ed.Washington D.C.: CDC/NIH;2009. Available from: URL:. http://www.cdc.gov/biosafety/publications/bmbl5/BMBL.pdf.12). World Health Organization. Laboratory biosafety manual. 3rd ed.Geneva: World Health Organization;2004. Available from: URL:. http://www.who.int/csr/resources/publications/biosafety/Biosafety7.pdf.13). Cho A, Seok SH. Ethical Guidelines for Use of Experimental Animals in Biomedical Research. J Bacteriol Virol. 2013; 43:18–26.
Article14). Convention on Biological Diversity. http://www.cbd.int.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Nurses' Patient Safety Management Importance, Patient Safety Culture and Nursing Service Quality on Patient Safety Management Activities in Tertiary Hospitals
- Associations of perceptions of patient safety culture, job crafting, and perceptions of patient rounding with patient safety management activities among tertiary hospital nurses
- Are Your Laboratory Data Reproducible? The Critical Role of Imprecision from Replicate Measurements to Clinical Decision-making
- Effects of perceptions of the importance of patient safety management and patient safety competency on patient safety management activities among nurses at unaccredited general hospitals
- Biologically Hazardous Agents at Work and Efforts to Protect Workers' Health: A Review of Recent Reports