Korean J Radiol.
2014 Feb;15(1):20-28. 10.3348/kjr.2014.15.1.20.
Breast Metastases from Extramammary Malignancies: Typical and Atypical Ultrasound Features
- Affiliations
-
- 1Department of Radiology and Center for Imaging Science, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul 135-710, Korea. claudel@skku.edu
- 2Department of Radiology, Catholic University of Daegu College of Medicine, Daegu 712-702, Korea.
- 3Department of Radiology, Inje University College of Medicine, Busan Paik Hospital, Busan 614-735, Korea.
- 4Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul 135-710, Korea.
- KMID: 1711473
- DOI: http://doi.org/10.3348/kjr.2014.15.1.20
Abstract
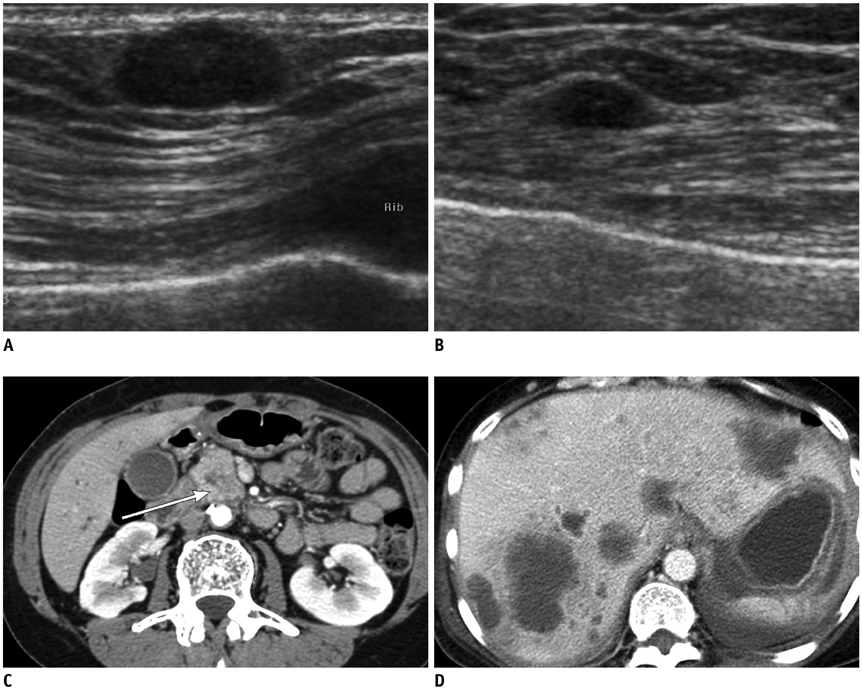
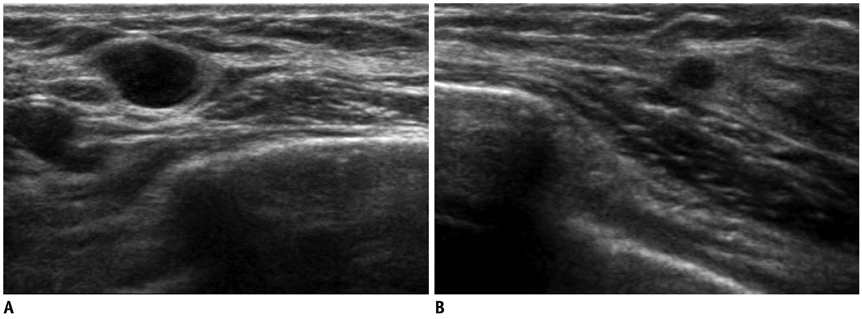
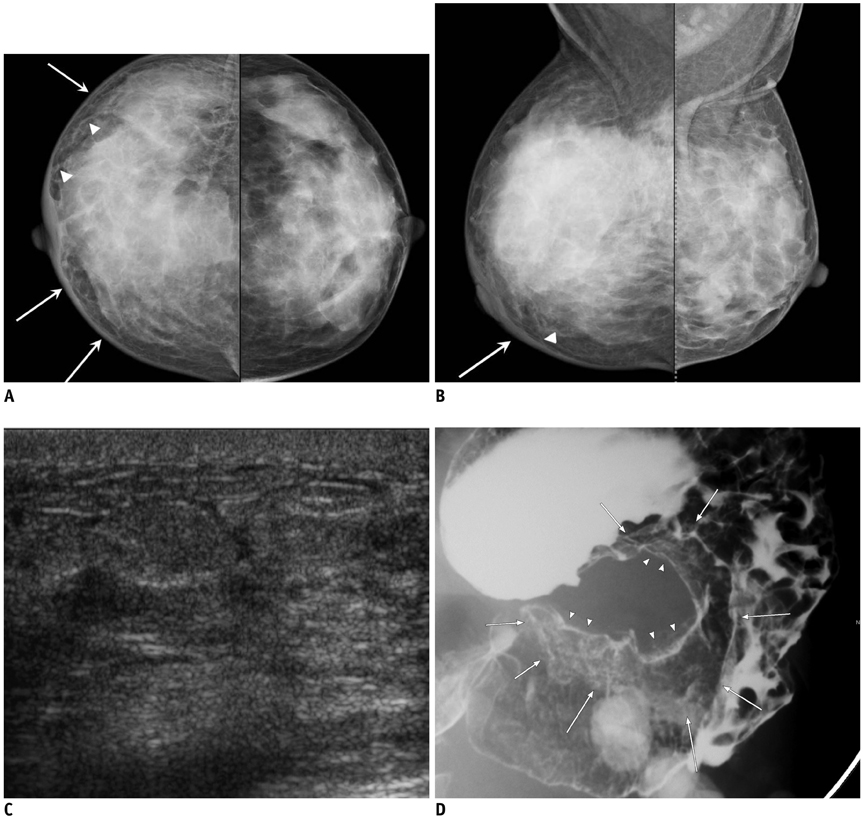
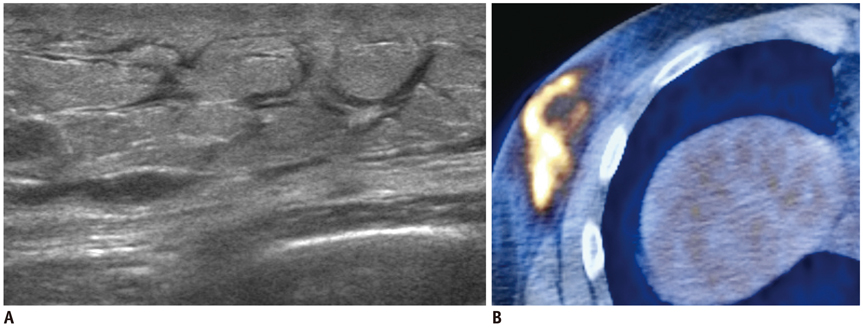
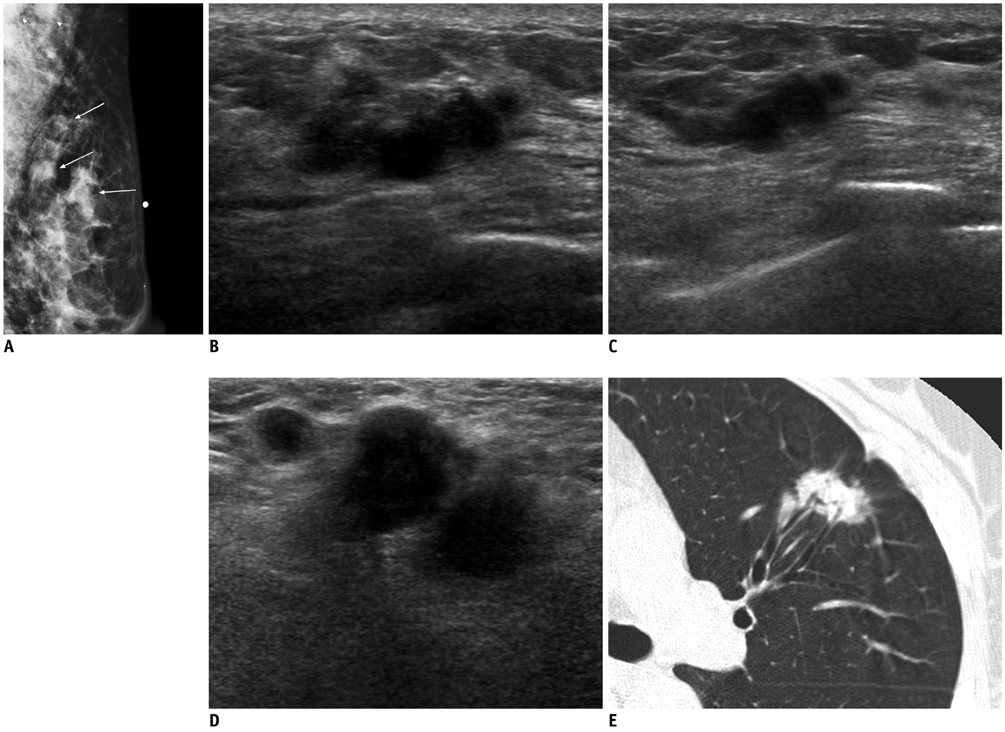
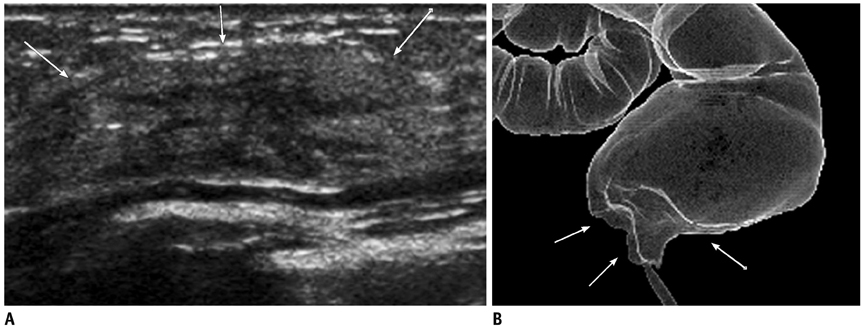
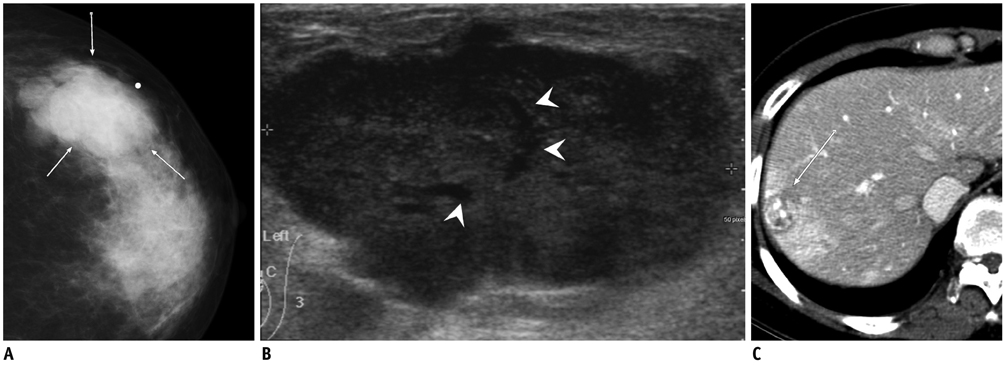
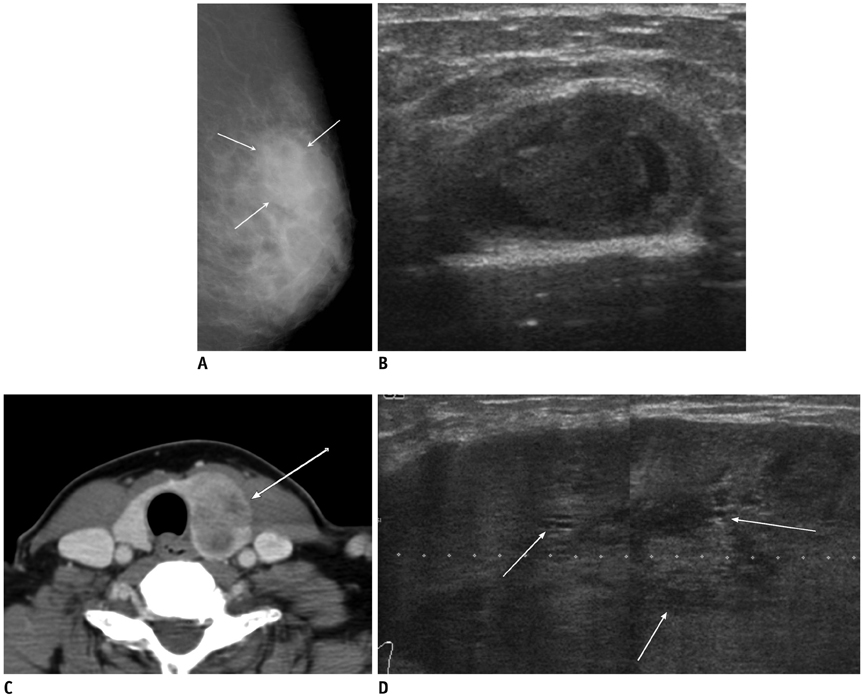
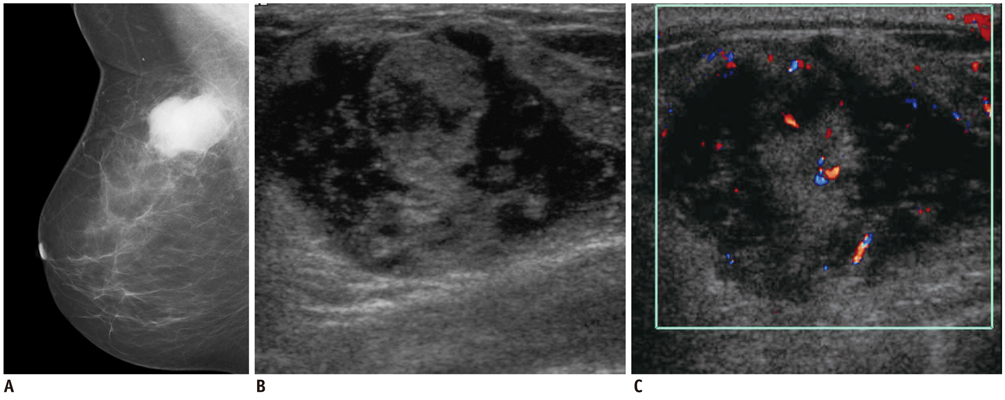
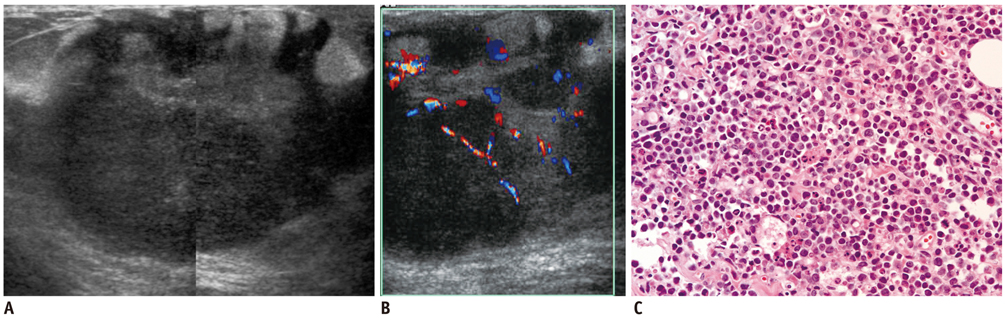
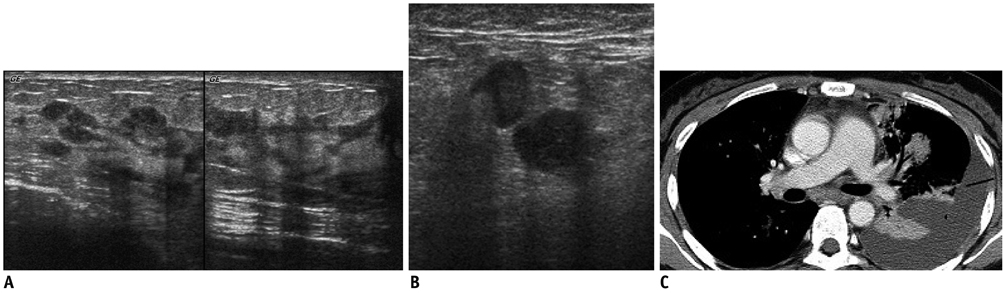
- Breast metastases from extramammary malignancies are uncommon. The most common sources are lymphomas/leukemias and melanomas. Some of the less common sources include carcinomas of the lung, ovary, and stomach, and infrequently, carcinoid tumors, hypernephromas, carcinomas of the liver, tonsil, pleura, pancreas, cervix, perineum, endometrium and bladder. Breast metastases from extramammary malignancies have both hematogenous and lymphatic routes. According to their routes, there are common radiological features of metastatic diseases of the breast, but the features are not specific for metastases. Typical ultrasound (US) features of hematogenous metastases include single or multiple, round to oval shaped, well-circumscribed hypoechoic masses without spiculations, calcifications, or architectural distortion; these masses are commonly located superficially in subcutaneous tissue or immediately adjacent to the breast parenchyma that is relatively rich in blood supply. Typical US features of lymphatic breast metastases include diffusely and heterogeneously increased echogenicities in subcutaneous fat and glandular tissue and a thick trabecular pattern with secondary skin thickening, lymphedema, and lymph node enlargement. However, lesions show variable US features in some cases, and differentiation of these lesions from primary breast cancer or from benign lesions is difficult. In this review, we demonstrate various US appearances of breast metastases from extramammary malignancies as typical and atypical features, based on the results of US and other imaging studies performed at our institution. Awareness of the typical and atypical imaging features of these lesions may be helpful to diagnose metastatic lesions of the breast.
Keyword
MeSH Terms
-
Adenocarcinoma/secondary/ultrasonography
Adolescent
Adult
Breast Neoplasms/*secondary/*ultrasonography
Breast Neoplasms, Male/secondary/ultrasonography
Carcinoma/secondary/ultrasonography
Female
Humans
Lymphatic Metastasis/ultrasonography
Lymphoma, Extranodal NK-T-Cell/pathology/ultrasonography
Lymphoma, Large B-Cell, Diffuse/pathology/ultrasonography
Male
Melanoma/secondary
Middle Aged
Multiple Myeloma/secondary/ultrasonography
Neoplastic Cells, Circulating/pathology
Figure
Cited by 2 articles
-
Metastatic Osteosarcoma to the Breast Presenting as a Densely Calcified Mass on Mammography
Jonghyeon Kim, Ha Young Woo, Eun-Kyung Kim, Min Jung Kim, Hee Jung Moon, Jung Hyun Yoon
J Breast Cancer. 2016;19(1):87-91. doi: 10.4048/jbc.2016.19.1.87.Salivary Duct Cancer Metastasis Mimicking Primary Breast Cancer: A Case Report and Review
Aisha M. Alzuhair, Gyungyub Gong, Hee Jung Shin, Jong Won Lee, Sae Byul Lee, Jisun Kim, Beom Seok Ko, Byung Ho Son, Sei Hyun Ahn, Il Yong Chung
J Breast Cancer. 2019;22(4):653-660. doi: 10.4048/jbc.2019.22.e45.
Reference
-
1. Lee SH, Park JM, Kook SH, Han BK, Moon WK. Metastatic tumors to the breast: mammographic and ultrasonographic findings. J Ultrasound Med. 2000; 19:257–262.2. McCrea ES, Johnston C, Haney PJ. Metastases to the breast. AJR Am J Roentgenol. 1983; 141:685–690.3. Muttarak M, Nimmonrat A, Chaiwun B. Metastatic carcinoma to the male and female breast. Australas Radiol. 1998; 42:16–19.4. Bohman LG, Bassett LW, Gold RH, Voet R. Breast metastases from extramammary malignancies. Radiology. 1982; 144:309–312.5. Silverman JF, Feldman PS, Covell JL, Frable WJ. Fine needle aspiration cytology of neoplasms metastatic to the breast. Acta Cytol. 1987; 31:291–300.6. Yeh CN, Lin CH, Chen MF. Clinical and ultrasonographic characteristics of breast metastases from extramammary malignancies. Am Surg. 2004; 70:287–290.7. Vergier B, Trojani M, de Mascarel I, Coindre JM, Le Treut A. Metastases to the breast: differential diagnosis from primary breast carcinoma. J Surg Oncol. 1991; 48:112–116.8. Amichetti M, Perani B, Boi S. Metastases to the breast from extramammary malignancies. Oncology. 1990; 47:257–260.9. Nielsen M, Andersen JA, Henriksen FW, Kristensen PB, Lorentzen M, Ravn V, et al. Metastases to the breast from extramammary carcinomas. Acta Pathol Microbiol Scand A. 1981; 89:251–256.10. Hajdu SI, Urban JA. Cancers metastatic to the breast. Cancer. 1972; 29:1691–1696.11. Yang WT, Metreweli C. Sonography of nonmammary malignancies of the breast. AJR Am J Roentgenol. 1999; 172:343–348.12. Bartella L, Kaye J, Perry NM, Malhotra A, Evans D, Ryan D, et al. Metastases to the breast revisited: radiological-histopathological correlation. Clin Radiol. 2003; 58:524–531.13. Derchi LE, Rizzatto G, Giuseppetti GM, Dini G, Garaventa A. Metastatic tumors in the breast: sonographic findings. J Ultrasound Med. 1985; 4:69–74.14. Yang WT, Muttarak M, Ho LW. Nonmammary malignancies of the breast: ultrasound, CT, and MRI. Semin Ultrasound CT MR. 2000; 21:375–394.15. Paulus DD, Libshitz HI. Metastasis to the breast. Radiol Clin North Am. 1982; 20:561–568.16. Cardenosa G. Breast Imaging. Philadelphia: Lippincott Williams & Wilkins;2004. p. 270–276.17. Jackson FI, Lalani ZH. Breast lymphoma: radiologic imaging and clinical appearances. Can Assoc Radiol J. 1991; 42:48–54.18. Yang WT, Lane DL, Le-Petross HT, Abruzzo LV, Macapinlac HA. Breast lymphoma: imaging findings of 32 tumors in 27 patients. Radiology. 2007; 245:692–702.19. Sabaté JM, Gómez A, Torrubia S, Camins A, Roson N, De Las Heras P, et al. Lymphoma of the breast: clinical and radiologic features with pathologic correlation in 28 patients. Breast J. 2002; 8:294–304.20. Paulus DD. Lymphoma of the breast. Radiol Clin North Am. 1990; 28:833–840.21. Chung SY, Oh KK. Imaging findings of metastatic disease to the breast. Yonsei Med J. 2001; 42:497–502.22. Nance FC, MacVaugh H 3rd, Fitts WT Jr. Metastatic tumor to the breast simulating bilateral primary inflammatory carcinoma. Am J Surg. 1966; 112:932–935.23. Krishnan EU, Phillips AK, Randell A, Taylor B, Garg SK. Bilateral metastatic inflammatory carcinoma in the breast from primary ovarian cancer. Obstet Gynecol. 1980; 55:3 Suppl. 94S–96S.24. Toombs BD, Kalisher L. Metastatic disease to the breast: clinical, pathologic, and radiographic features. AJR Am J Roentgenol. 1977; 129:673–676.25. Tateishi U, Hasegawa T, Beppu Y, Satake M, Moriyama N. Synovial sarcoma of the soft tissues: prognostic significance of imaging features. J Comput Assist Tomogr. 2004; 28:140–148.26. Layfield LJ, Gopez EV. Insular carcinoma of the thyroid: report of a case with intact insulae and microfollicular structures. Diagn Cytopathol. 2000; 23:409–413.27. Brancatelli G, Federle MP, Grazioli L, Carr BI. Hepatocellular carcinoma in noncirrhotic liver: CT, clinical, and pathologic findings in 39 U.S. residents. Radiology. 2002; 222:89–94.28. Pope TL Jr, Brenbridge AN, Sloop FB Jr, Morris JR 3rd, Carpenter J. Primary histiocytic lymphoma of the breast: mammographic, sonographic, and pathologic correlation. J Clin Ultrasound. 1985; 13:667–670.29. Liberman L, Giess CS, Dershaw DD, Louie DC, Deutch BM. Non-Hodgkin lymphoma of the breast: imaging characteristics and correlation with histopathologic findings. Radiology. 1994; 192:157–160.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Breast Metastatic Squamous Cell Carcinoma from Lung
- Breast Metastasis from Gastric Adenocarcinoma Mimicking Normal Breast Parenchyma on Ultrasound: A Case Report
- Metastatic Tumors to the Breast from Extramammary Malignancies
- A Case of Extramammary Paget's Disease of the Axilla
- Breast Metastasis from Gastric Cancer Mimicking Benign Breast Mass