Cancer Res Treat.
2005 Apr;37(2):103-108.
Mast Cell and Macrophage Counts and Microvessel Density in Invasive Breast Carcinoma-Comparison Analysis with Clinicopathological Parameters
- Affiliations
-
- 1Department of Pathology, Chung-Ang University College of Medicine, Seoul, Korea. esp@cau.ac.kr
Abstract
- PURPOSE
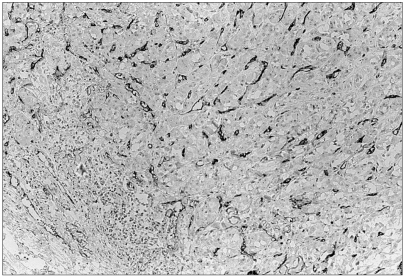
The purpose of this study was to evaluate the clinicopathological significance of the microvessel density and macrophage and mast cell counts in invasive breast carcinomas. MATERIALS AND METHODS: 45 invasive breast carcinomas were immunohistochemically stained with the endothelial antigen, CD34, and macrophage marker, CD68. 0.1% toluidine blue was used to highlight mast cells. The microvessel and mast cell counts were performed at x200 magnification and the macrophages at x400 magnification. RESULTS: With the 45 invasive breast carcinomas, there were no statistically significant associations between the mast cell, macrophage and microvessel counts and the tumor size and lymph node status. ER and PR negative mast cells infiltrated more than in cases of positive stati, with statistical significance (p-value=0.010 and 0.005, respectively). The macrophage counts were negatively correlated with the PR status (p-value=0.030). With respect to the c-erbB-2 status, there was no significance correlation with the mast cell, macrophage and microvessel counts. The mast cell counts showed significantly positive correlation with the microvessel counts in the invasive breast carcinomas (p-value=0.015). In a comparison of the macrophage counts with the microvessel counts, a positive tendency for both parameters, but without statistical significance (p-value=0.310). CONCLUSION: Increasing numbers of mast cells and macrophages were recruited in invasive breast carcinomas, which contribute to angiogenesis. The microvessel density in invasive breast carcinomas had no statistically significant association with the tumor size, lymph node status, and histological grade, presence of DCIS component, estrogen/progesterone receptor status and cerbB-2 status. The evaluation of angiogenesis using these methods is not thought to provide an independent clinicopathological factor in invasive breast carcinomas.
MeSH Terms
Figure
Reference
-
1. Polverini PJ. How the extracellular matrix and macrophages contribute to angiogenesis-dependent diseases. Eur J Cancer. 1996; 32A:2430–2437. PMID: 9059331.
Article2. Kessler DA, Langer RS, Pless NA, Folkman J. Mast cells ant tumor angiogenesis. Int J Cancer. 1976; 18:703–709. PMID: 62725.3. Starkey JR, Crowle PK, Taubenberger S. Mast cell-deficient W/Wv mice exhibit a decreased rate of tumor angiogenesis. Int J Cancer. 1988; 42:48–52. PMID: 2455691.4. Morra B, Ferrero V, Bussi M, Pacchioni D, Cerrato M, Bussolati G. Peri and intratumoral macrophage infiltration in laryngeal carcinoma. An immunohistochemical study. Acta Otolaryngol. 1991; 111:444–448. PMID: 2068934.5. Bo L, Mork SJ, Nyland H. An immunohistochemical study of mononuclear cells in meningiomas. Neuropathol Appl Neurobiol. 1992; 18:548–558. PMID: 1488087.
Article6. Folkman J. Clinical applications of research on angiogenesis. N Engl J Med. 1995; 333:1757–1763. PMID: 7491141.
Article7. Mignatti P, Rifkin DB. Biology and biochemistry of proteinases in tumor invasion. Physiol Rev. 1993; 73:161–195. PMID: 8419965.
Article8. Hildenbrand R, Dilger I, Horlin A, Stutte HJ. Urokinase and macrophages in tumour angiogenesis. Br J Cancer. 1995; 72:818–823. PMID: 7547226.
Article9. Leek RD, Lewis CE, Whitehouse R, Greenall M, Clarke J, Harris AL. Association of macrophage infiltration with angiogenesis and prognosis in invasive breast carcinoma. Cancer Res. 1996; 56:4625–4629. PMID: 8840975.10. Bigler SA, Deering RE, Brawer MK. Comparison of microscopic vascularity in benign and malignant prostate tissue. Hum Pathol. 1993; 24:220–226. PMID: 8432518.
Article11. Weidner N, Semple JP, Welch WR, Folkman J. Tumor angiogenesis and metastasis-correlation in invasive breast carcinoma. N Engl J Med. 1991; 324:1–8. PMID: 1701519.12. Liotta LA, Kleinerman J, Saidel GM. Quantitative relationships of intravascular tumor cells, tumor vessels, and pulmonary metastases following tumor implantation. Cancer Res. 1974; 34:997–1004. PMID: 4841969.13. Uzzan B, Nicolas P, Cucherat M, Perret GY. Microvessel density as a prognostic factor in women with breast cancer: a systematic review of the literature and meta-analysis. Cancer Res. 2004; 64:2941–2955. PMID: 15126324.14. Cozzolino F, Torcia M, Aldinucci D, Rubartelli A, Miliani A, Shaw AR, et al. Production of interleukin-1 by bone marrow myeloma cells. Blood. 1989; 74:380–387. PMID: 2665838.
Article15. Kang HJ, Gong G, Jang SJ, Jung PJ, Park CK. Expression of cyclooxygenase-2 in human breast carcinoma: Relevance to tumor angiogenesis and expression of estrogen receptor. Cancer Res Treat. 2001; 33:286–295.
Article16. Meininger CJ, Zetter BR. Mast cells and angiogenesis. Semin Cancer Biol. 1992; 3:73–79. PMID: 1378312.17. Qu Z, Liebler JM, Powers MR, Galey T, Ahmadi P, Huang XN, et al. Mast cells are a major source of basic fibroblast growth factor in chronic inflammation and cutaneous hemangioma. Am J Pathol. 1995; 147:564–573. PMID: 7545872.18. Motro B, Itin A, Sachs L, Keshet E. Pattern of interleukin 6 gene expression in vivo suggests a role for this cytokine in angiogenesis. Proc Natl Acad Sci USA. 1990; 87:3092–3096. PMID: 1691500.
Article19. Bussolino F, Ziche M, Wang JM, Alessi D, Morbidelli L, Cremona O, et al. In vitro and in vivo activation of endothelial cells by colony-stimulating factors. J Clin Invest. 1991; 87:986–995. PMID: 1705569.
Article20. Roberts AB, Sporn MB, Assoian RK, Smith JM, Roche NS, Wakefield LM, et al. Transforming growth factor type beta: rapid induction of fibrosis and angiogenesis in vivo and stimulation of collagen formation in vitro. Proc Natl Acad Sci USA. 1986; 83:4167–4171. PMID: 2424019.
Article21. Raju KS, Alessandri G, Gullino PM. Characterization of a chemoattractant for endothelium induced by angiogenesis effectors. Cancer Res. 1984; 44:1579–1584. PMID: 6200213.22. Norrby K. Heparin and angiogenesis: a low-molecular-weight fraction inhibits and a high-molecular-weight fraction stimulates angiogenesis systemically. Haemostasis. 1993; 23(Suppl. 1):141–149. PMID: 7684350.
Article23. Wilks JW, Scott PS, Vrba LK, Cocuzza JM. Inhibition of angiogenesis with combination treatments of angiostatic steroids and suramin. Int J Radiat Biol. 1991; 60:73–77. PMID: 1713944.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Angiogenesis in Cervical Intraepithelial Neoplasia and Invasive Cervical Cancer: an Immunohistochemical Study
- Image Analysis Quantifying Microvessel Density in Renal Cell Carcinoma
- VEGF Expression and Microvessel Density in Oral Squamous Cell Carcinomas
- Analysis of Tumor Angiogenesis in Invasive Cervical Cancer
- Correlation between Tumor Angiogenesis (Microvessel Density), Metastasis and Tumor Cell Proliferation in Colorectal Carcinomas