Korean J Physiol Pharmacol.
2012 Oct;16(5):343-348. 10.4196/kjpp.2012.16.5.343.
Sustained K+ Outward Currents are Sensitive to Intracellular Heteropodatoxin2 in CA1 Neurons of Organotypic Cultured Hippocampi of Rats
- Affiliations
-
- 1Department of Physiology, School of Medicine, Jeju National University, Jeju 690-756, Korea. jungsc@jejunu.ac.kr
- KMID: 1493968
- DOI: http://doi.org/10.4196/kjpp.2012.16.5.343
Abstract
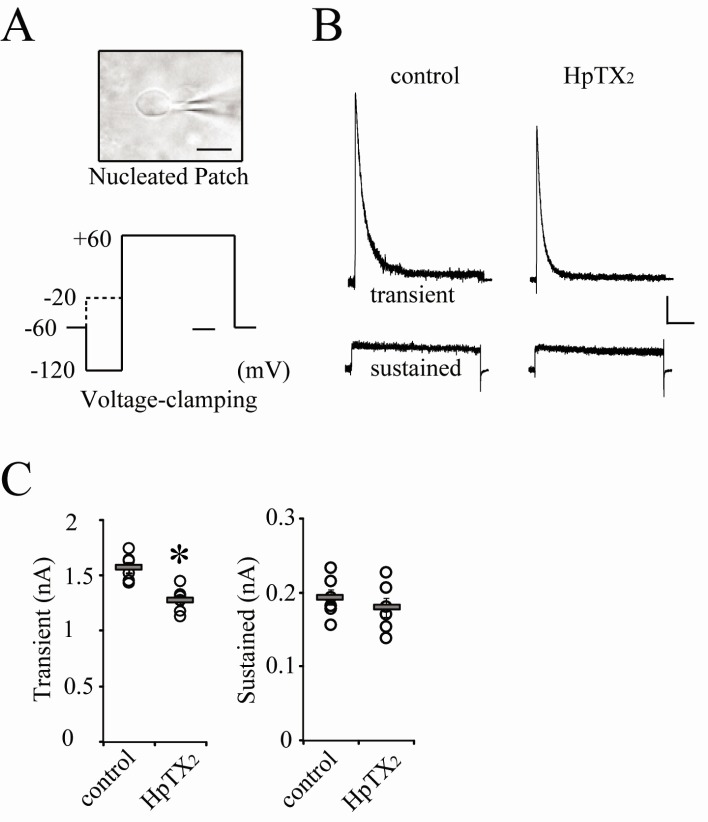
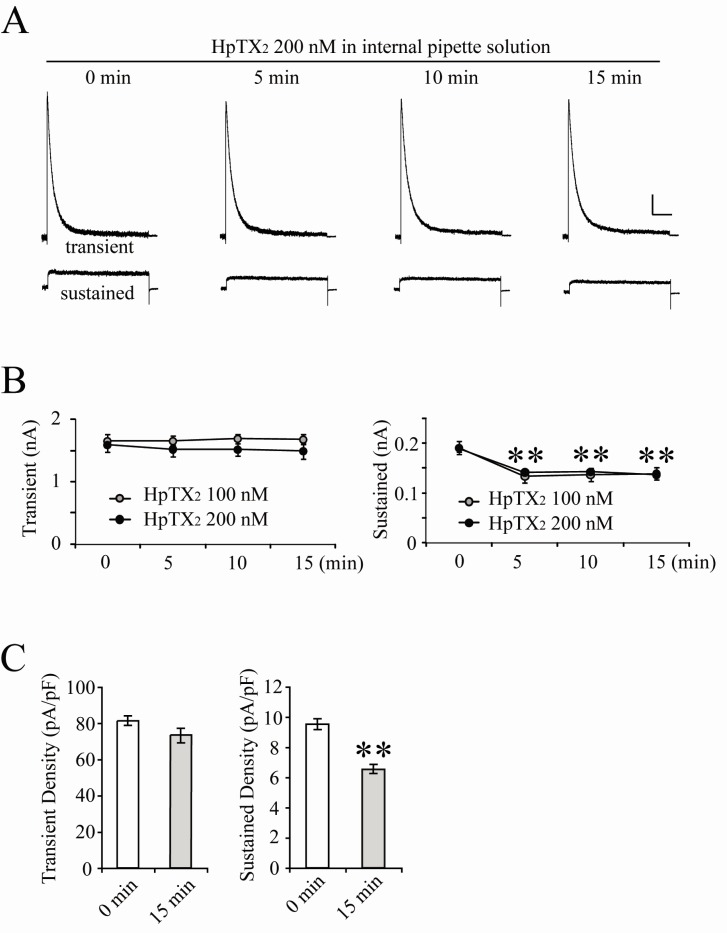
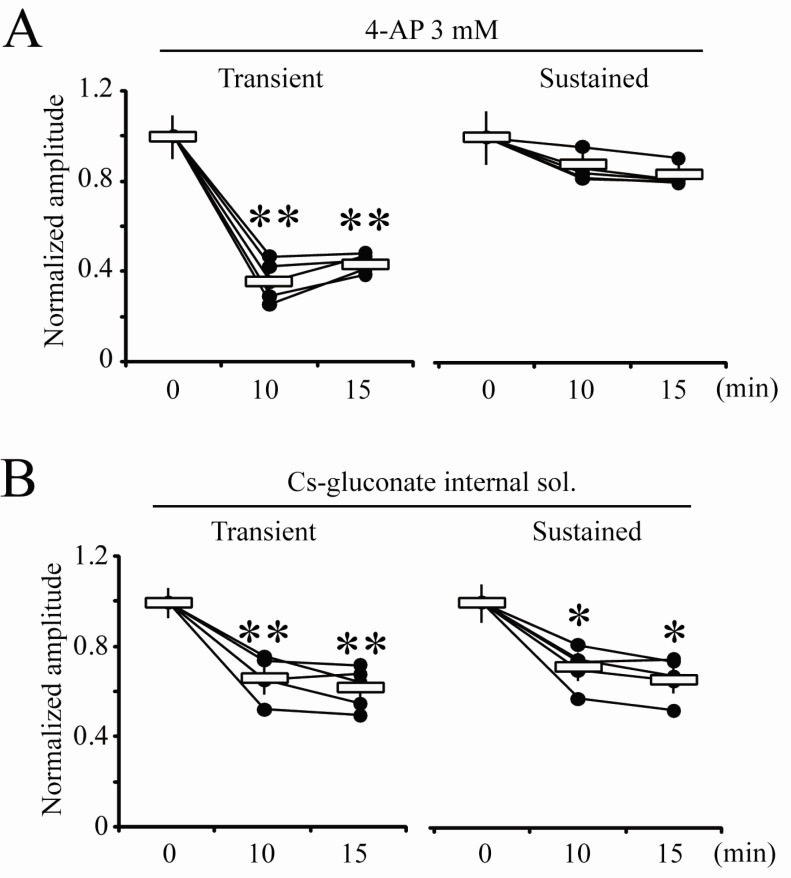
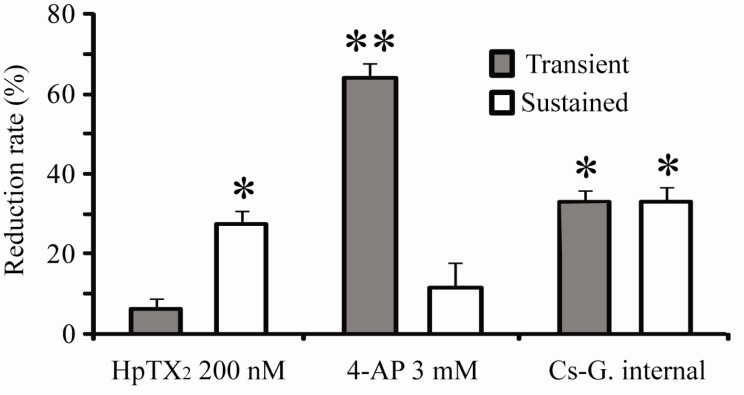
- Blocking or regulating K+ channels is important for investigating neuronal functions in mammalian brains, because voltage-dependent K+ channels (Kv channels) play roles to regulate membrane excitabilities for synaptic and somatic processings in neurons. Although a number of toxins and chemicals are useful to change gating properties of Kv channels, specific effects of each toxin on a particular Kv subunit have not been sufficiently demonstrated in neurons yet. In this study, we tested electrophysiologically if heteropodatoxin2 (HpTX2), known as one of Kv4-specific toxins, might be effective on various K+ outward currents in CA1 neurons of organotypic hippocampal slices of rats. Using a nucleated-patch technique and a pre-pulse protocol in voltage-clamp mode, total K+ outward currents recorded in the soma of CA1 neurons were separated into two components, transient and sustained currents. The extracellular application of HpTX2 weakly but significantly reduced transient currents. However, when HpTX2 was added to internal solution, the significant reduction of amplitudes were observed in sustained currents but not in transient currents. This indicates the non-specificity of HpTX2 effects on Kv4 family. Compared with the effect of cytosolic 4-AP to block transient currents, it is possible that cytosolic HpTX2 is pharmacologically specific to sustained currents in CA1 neurons. These results suggest that distinctive actions of HpTX2 inside and outside of neurons are very efficient to selectively reduce specific K+ outward currents.
Keyword
Figure
Reference
-
1. Bankar GR, Nampurath GK, Nayak PG, Bhattacharya S. A possible correlation between the correction of endothelial dysfunction and normalization of high blood pressure levels by 1,3,4-oxadiazole derivative, an L-type Ca2+ channel blocker in deoxycorticosterone acetate and N(G)-nitro-l-arginine hypertensive rats. Chem Biol Interact. 2010; 183:327–331. PMID: 19912999.2. Cai BX, Li XY, Chen JH, Tang YB, Wang GL, Zhou JG, Qui QY, Guan YY. Ginsenoside-Rd, a new voltage-independent Ca2+ entry blocker, reverses basilar hypertrophic remodeling in stroke-prone renovascular hypertensive rats. Eur J Pharmacol. 2009; 606:142–149. PMID: 19374845.3. Das A, McDowell M, O'Dell CM, Busch ME, Smith JA, Ray SK, Banik NL. Post-treatment with voltage-gated Na(+) channel blocker attenuates kainic acid-induced apoptosis in rat primary hippocampal neurons. Neurochem Res. 2010; 35:2175–2183. PMID: 21127971.
Article4. Dzhala VI, Kuchibhotla KV, Glykys JC, Kahle KT, Swiercz WB, Feng G, Kuner T, Augustine GJ, Bacskai BJ, Staley KJ. Progressive NKCC1-dependent neuronal chloride accumulation during neonatal seizures. J Neurosci. 2010; 30:11745–11761. PMID: 20810895.
Article5. Kobayashi T, Hirai H, Iino M, Fuse I, Mitsumura K, Washiyama K, Kasai S, Ikeda K. Inhibitory effects of the antiepileptic drug ethosuximide on G protein-activated inwardly rectifying K+ channels. Neuropharmacology. 2009; 56:499–506. PMID: 18977371.6. Nakajima S, Iwasaki S, Obata K. Delayed rectification and anomalous rectification in frog's skeletal muscle membrane. J Gen Physiol. 1962; 46:97–115. PMID: 14478119.
Article7. Nicholson GM, Little MJ, Tyler M, Narahashi T. Selective alteration of sodium channel gating by Australian funnel-web spider toxins. Toxicon. 1996; 34:1443–1453. PMID: 9028001.
Article8. Yao S, Zhang MM, Yoshikami D, Azam L, Olivera BM, Bulaj G, Norton RS. Structure, dynamics, and selectivity of the sodium channel blocker mu-conotoxin SIIIA. Biochemistry. 2008; 47:10940–10949. PMID: 18798648.9. Gandolfo G, Gottesmann C, Bidard JN, Lazdunski M. Subtypes of K+ channels differentiated by the effect of K+ channel openers upon K+ channel blocker-induced seizures. Brain Res. 1989; 495:189–192. PMID: 2550110.10. Andersson KE. Pharmacodynamic profiles of different calcium channel blockers. Acta Pharmacol Toxicol (Copenh). 1986; 58(Suppl 2):31–42. PMID: 2424267.
Article11. Cognard C, Lazdunski M, Romey G. Different types of Ca2+ channels in mammalian skeletal muscle cells in culture. Proc Natl Acad Sci USA. 1986; 83:517–521. PMID: 2417248.13. Hoffman DA, Magee JC, Colbert CM, Johnston D. K+ channel regulation of signal propagation in dendrites of hippocampal pyramidal neurons. Nature. 1997; 387:869–875. PMID: 9202119.14. Pan Z, Yamaguchi R, Doi S. Bifurcation analysis and effects of changing ionic conductances on pacemaker rhythm in a sinoatrial node cell model. Biosystems. 2011; 106:9–18. PMID: 21683757.
Article15. Marbán E, Cho HC. Biological pacemakers as a therapy for cardiac arrhythmias. Curr Opin Cardiol. 2008; 23:46–54. PMID: 18281827.
Article16. Jung SC, Hoffman DA. Biphasic somatic A-type K channel downregulation mediates intrinsic plasticity in hippocampal CA1 pyramidal neurons. PLoS One. 2009; 4:e6549. PMID: 19662093.
Article17. Du J, Haak LL, Phillips-Tansey E, Russell JT, McBain CJ. Frequency-dependent regulation of rat hippocampal somatodendritic excitability by the K+ channel subunit Kv2.1. J Physiol. 2000; 522(Pt 1):19–31. PMID: 10618149.18. Sanguinetti MC, Johnson JH, Hammerland LG, Kelbaugh PR, Volkmann RA, Saccomano NA, Mueller AL. Heteropodatoxins: peptides isolated from spider venom that block Kv4.2 potassium channels. Mol Pharmacol. 1997; 51:491–498. PMID: 9058605.19. Zarayskiy VV, Balasubramanian G, Bondarenko VE, Morales MJ. Heteropoda toxin 2 is a gating modifier toxin specific for voltage-gated K+ channels of the Kv4 family. Toxicon. 2005; 45:431–442. PMID: 15733564.20. Swartz KJ. Tarantula toxins interacting with voltage sensors in potassium channels. Toxicon. 2007; 49:213–230. PMID: 17097703.
Article21. Li Y, Hu GY. Huperzine A inhibits the sustained potassium current in rat dissociated hippocampal neurons. Neurosci Lett. 2002; 329:153–156. PMID: 12165400.
Article22. Gu C, Barry J. Function and mechanism of axonal targeting of voltage-sensitive potassium channels. Prog Neurobiol. 2011; 94:115–132. PMID: 21530607.
Article23. Kim J, Jung SC, Clemens AM, Petralia RS, Hoffman DA. Regulation of dendritic excitability by activity-dependent trafficking of the A-type K+ channel subunit Kv4.2 in hippocampal neurons. Neuron. 2007; 54:933–947. PMID: 17582333.24. Jung SC, Kim J, Hoffman DA. Rapid, bidirectional remodeling of synaptic NMDA receptor subunit composition by A-type K+ channel activity in hippocampal CA1 pyramidal neurons. Neuron. 2008; 60:657–671. PMID: 19038222.25. Kim J, Wei DS, Hoffman DA. Kv4 potassium channel subunits control action potential repolarization and frequency-dependent broadening in rat hippocampal CA1 pyramidal neurones. J Physiol. 2005; 569:41–57. PMID: 16141270.
Article26. Willenborg M, Ghaly H, Hatlapatka K, Urban K, Panten U, Rustenbeck I. The signalling role of action potential depolarization in insulin secretion: metabolism-dependent dissociation between action potential increase and secretion increase by TEA. Biochem Pharmacol. 2010; 80:104–112. PMID: 20303336.27. Choi JS, Hahn SJ. Duloxetine blocks cloned Kv4.3 potassium channels. Brain Res. 2012; 1466:15–23. PMID: 22618310.
Article28. Antonucci DE, Lim ST, Vassanelli S, Trimmer JS. Dynamic localization and clustering of dendritic Kv2.1 voltage-dependent potassium channels in developing hippocampal neurons. Neuroscience. 2001; 108:69–81. PMID: 11738132.
Article29. Misonou H, Thompson SM, Cai X. Dynamic regulation of the Kv2.1 voltage-gated potassium channel during brain ischemia through neuroglial interaction. J Neurosci. 2008; 28:8529–8538. PMID: 18716211.
Article30. Mohapatra DP, Misonou H, Pan SJ, Held JE, Surmeier DJ, Trimmer JS. Regulation of intrinsic excitability in hippocampal neurons by activity-dependent modulation of the KV2.1 potassium channel. Channels (Austin). 2009; 3:46–56. PMID: 19276663.
Article31. Nerbonne JM, Gerber BR, Norris A, Burkhalter A. Electrical remodelling maintains firing properties in cortical pyramidal neurons lacking KCND2-encoded A-type K+ currents. J Physiol. 2008; 586:1565–1579. PMID: 18187474.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Ginkgolide B on the Neuronal Excitability of Medial Vestibular Nuclear Neurons in the Rat
- K+ Currents in Medial Vestibular Nuclear Neurons
- Characteristics of Potassium and Calcium Currents of Hepatic Stellate Cells (Ito) in Rat
- Heterogeneous Composition of Voltage-Dependent K+ Currents in Hepatic Stellate Cells
- Four Voltage-Gated Potassium Currents in Trigeminal Root Ganglion Neurons