Protective Effects of Oleic Acid Against Palmitic Acid-Induced Apoptosis in Pancreatic AR42J Cells and Its Mechanisms
- Affiliations
-
- 1Department of Physiology, College of Medicine, College of Medicine, Hallym University, Chuncheon 200-702, Korea. hykwon@hallym.ac.kr
- 2Department of Biomedical Sciences, College of Natural Sciences, Hallym University, Chuncheon 200-702, Korea.
- 3Department of Microbiology, College of Medicine, Hallym University, Chuncheon 200-702, Korea.
- KMID: 1432762
- DOI: http://doi.org/10.4196/kjpp.2013.17.1.43
Abstract
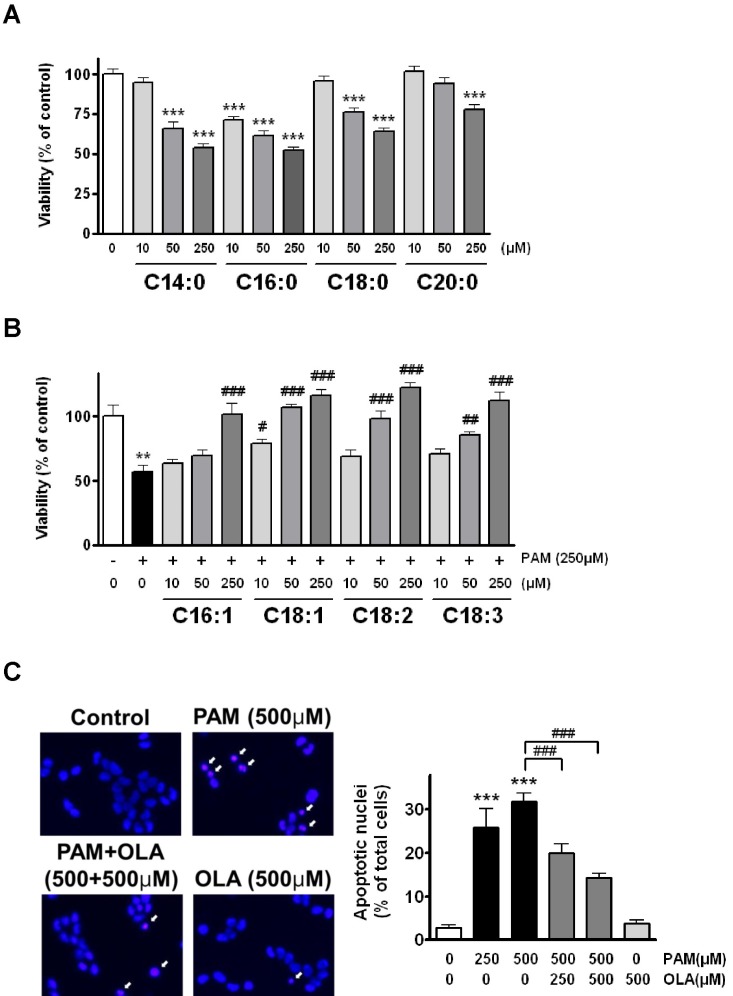
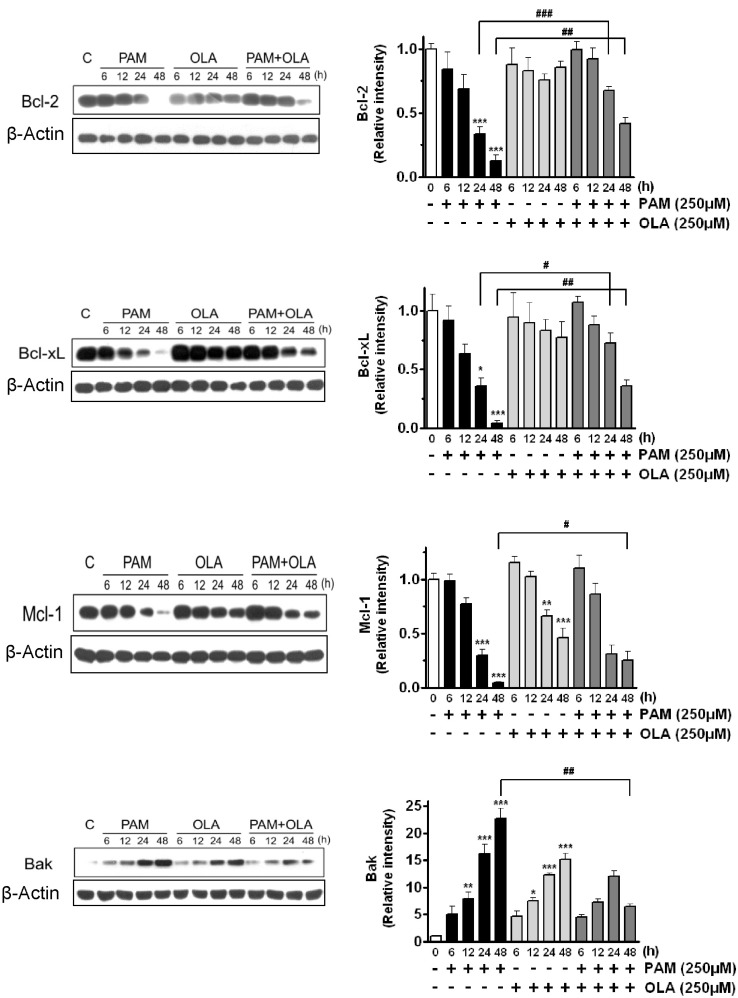
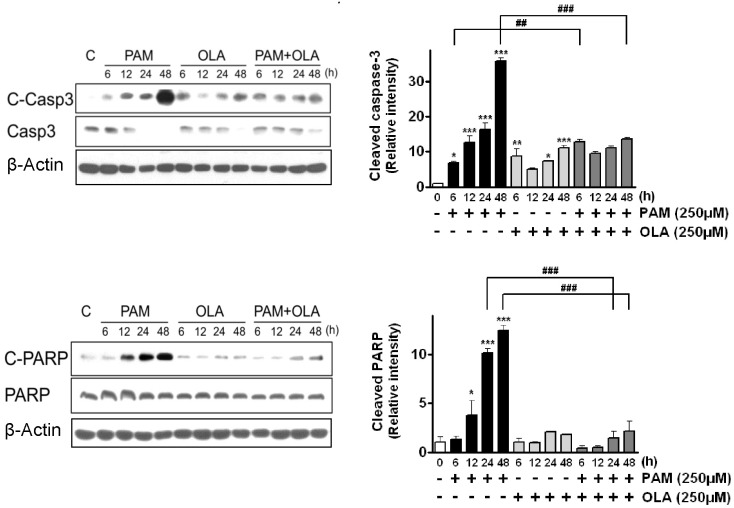
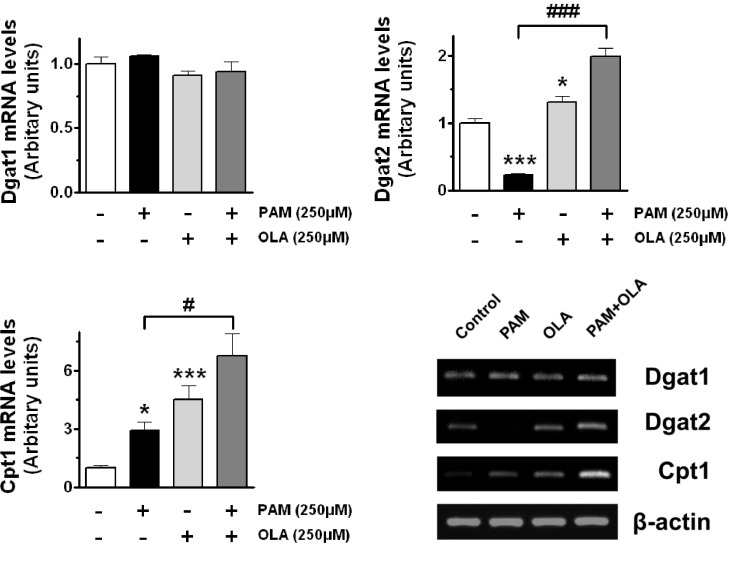
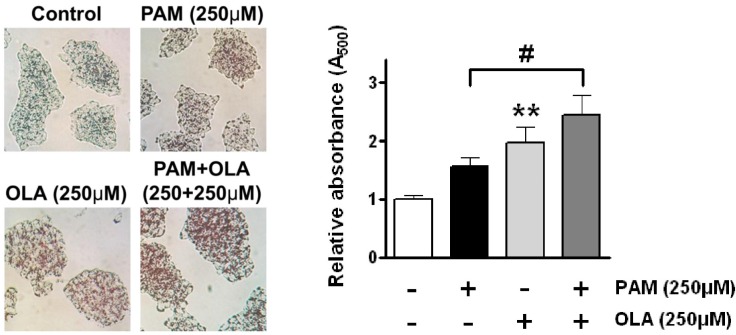
- Palmitic acid (PAM), one of the most common saturated fatty acid (SFA) in animals and plants, has been shown to induce apoptosis in exocrine pancreatic AR42J cells. In this study, we investigated cellular mechanisms underlying protective effects of oleic acid (OLA) against the lipotoxic actions of PAM in AR42J cells. Exposure of cells to long-chain SFA induced apoptotic cell death determined by MTT cell viability assay and Hoechst staining. Co-treatment of OLA with PAM markedly protected cells against PAM-induced apoptosis. OLA significantly attenuated the PAM-induced increase in the levels of pro-apoptotic Bak protein, cleaved forms of apoptotic proteins (caspase-3, PARP). On the contrary, OLA restored the decreased levels of anti-apoptotic Bcl-2 family proteins (Bcl-2, Bcl-xL, and Mcl-1) in PAM-treated cells. OLA also induced up-regulation of the mRNA expression of Dgat2 and Cpt1 genes which are involved in triacylglycerol (TAG) synthesis and mitochondrial beta-oxidation, respectively. Intracellular TAG accumulation was increased by OLA supplementation in accordance with enhanced expression of Dgat2 gene. These results indicate that restoration of anti-apoptotic/pro-apoptotic protein balance from apoptosis toward cell survival is involved in the cytoprotective effects of OLA against PAM-induced apoptosis in pancreatic AR42J cells. In addition, OLA-induced increase in TAG accumulation and up-regulation of Dgat2 and Cpt1 gene expressions may be possibly associated in part with the ability of OLA to protect cells from deleterious actions of PAM.
Keyword
MeSH Terms
Figure
Cited by 3 articles
-
Multiple Effects of a Novel Epothilone Analog on Cellular Processes and Signaling Pathways Regulated by Rac1 GTPase in the Human Breast Cancer Cells
Hong Zhang, Fan An, Li Tang, Rongguo Qiu
Korean J Physiol Pharmacol. 2014;18(2):109-120. doi: 10.4196/kjpp.2014.18.2.109.The Stimulatory Effect of Essential Fatty Acids on Glucose Uptake Involves Both Akt and AMPK Activation in C2C12 Skeletal Muscle Cells
So Yeon Park, Min Hye Kim, Joung Hoon Ahn, Su Jin Lee, Jong Ho Lee, Won Sik Eum, Soo Young Choi, Hyeok Yil Kwon
Korean J Physiol Pharmacol. 2014;18(3):255-261. doi: 10.4196/kjpp.2014.18.3.255.Novel functional roles of caspase-related genes in the regulation of apoptosis and autophagy
Ju-Hyun Shin, Sang-Hyun Min
Korean J Physiol Pharmacol. 2016;20(6):573-580. doi: 10.4196/kjpp.2016.20.6.573.
Reference
-
1. Rasouli N, Molavi B, Elbein SC, Kern PA. Ectopic fat accumulation and metabolic syndrome. Diabetes Obes Metab. 2007; 9:1–10. PMID: 17199713.
Article2. Unger RH. Lipotoxicity in the pathogenesis of obesity-dependent NIDDM. Genetic and clinical implications. Diabetes. 1995; 44:863–870. PMID: 7621989.
Article3. Shimabukuro M, Wang MY, Zhou YT, Newgard CB, Unger RH. Protection against lipoapoptosis of beta cells through leptin-dependent maintenance of Bcl-2 expression. Proc Natl Acad Sci USA. 1998; 95:9558–9561. PMID: 9689119.4. Unger RH, Orci L. Lipoapoptosis: its mechanism and its diseases. Biochim Biophys Acta. 2002; 1585:202–212. PMID: 12531555.
Article5. Kusminski CM, Shetty S, Orci L, Unger RH, Scherer PE. Diabetes and apoptosis: lipotoxicity. Apoptosis. 2009; 14:1484–1495. PMID: 19421860.
Article6. Mu YM, Yanase T, Nishi Y, Tanaka A, Saito M, Jin CH, Mukasa C, Okabe T, Nomura M, Goto K, Nawata H. Saturated FFAs, palmitic acid and stearic acid, induce apoptosis in human granulosa cells. Endocrinology. 2001; 142:3590–3597. PMID: 11459807.
Article7. Eitel K, Staiger H, Brendel MD, Brandhorst D, Bretzel RG, Haring HU, Kellerer M. Different role of saturated and unsaturated fatty acids in beta-cell apoptosis. Biochem Biophys Res Commun. 2002; 299:853–856. PMID: 12470657.8. Akazawa Y, Cazanave S, Mott JL, Elmi N, Bronk SF, Kohno S, Charlton MR, Gores GJ. Palmitoleate attenuates palmitate-induced Bim and PUMA up-regulation and hepatocyte lipoapoptosis. J Hepatol. 2010; 52:586–593. PMID: 20206402.
Article9. Suzuki J, Akahane K, Nakamura J, Naruse K, Kamiya H, Himeno T, Nakamura N, Shibata T, Kondo M, Nagasaki H, Fujiya A, Oiso Y, Hamada Y. Palmitate induces apoptosis in Schwann cells via both ceramide-dependent and independent pathways. Neuroscience. 2011; 176:188–198. PMID: 21145948.
Article10. Turpin SM, Lancaster GI, Darby I, Febbraio MA, Watt MJ. Apoptosis in skeletal muscle myotubes is induced by ceramides and is positively related to insulin resistance. Am J Physiol Endocrinol Metab. 2006; 291:E1341–E1350. PMID: 16849630.
Article11. Cazanave SC, Elmi NA, Akazawa Y, Bronk SF, Mott JL, Gores GJ. CHOP and AP-1 cooperatively mediate PUMA expression during lipoapoptosis. Am J Physiol Gastrointest Liver Physiol. 2010; 299:G236–G243. PMID: 20430872.
Article12. Soumura M, Kume S, Isshiki K, Takeda N, Araki S, Tanaka Y, Sugimoto T, Chin-Kanasaki M, Nishio Y, Haneda M, Koya D, Kashiwagi A, Maegawa H, Uzu T. Oleate and eicosapentaenoic acid attenuate palmitate-induced inflammation and apoptosis in renal proximal tubular cell. Biochem Biophys Res Commun. 2010; 402:265–271. PMID: 20937260.
Article13. Yuzefovych L, Wilson G, Rachek L. Different effects of oleate vs. palmitate on mitochondrial function, apoptosis, and insulin signaling in L6 skeletal muscle cells: role of oxidative stress. Am J Physiol Endocrinol Metab. 2010; 299:E1096–E1105. PMID: 20876761.
Article14. Kovanlikaya A, Mittelman SD, Ward A, Geffner ME, Dorey F, Gilsanz V. Obesity and fat quantification in lean tissues using three-point Dixon MR imaging. Pediatr Radiol. 2005; 35:601–607. PMID: 15785930.
Article15. Yan MX, Li YQ, Meng M, Ren HB, Kou Y. Long-term high-fat diet induces pancreatic injuries via pancreatic microcirculatory disturbances and oxidative stress in rats with hyperlipidemia. Biochem Biophys Res Commun. 2006; 347:192–199. PMID: 16814251.
Article16. Cnop M, Hannaert JC, Hoorens A, Eizirik DL, Pipeleers DG. Inverse relationship between cytotoxicity of free fatty acids in pancreatic islet cells and cellular triglyceride accumulation. Diabetes. 2001; 50:1771–1777. PMID: 11473037.
Article17. Gumbs AA. Obesity, pancreatitis, and pancreatic cancer. Obes Surg. 2008; 18:1183–1187. PMID: 18563497.
Article18. Habara Y, Uehara A, Takasugi Y, Namiki M, Kanno T. Characterization of secretory responses in exocrine pancreas of genetically obese Zucker rats. Int J Pancreatol. 1991; 10:237–245. PMID: 1724009.
Article19. Hardt PD, Krauss A, Bretz L, Porsch-Ozcürümez M, Schnell-Kretschmer H, Mser E, Bretzel RG, Zekhorn T, Klör HU. Pancreatic exocrine function in patients with type 1 and type 2 diabetes mellitus. Acta Diabetol. 2000; 37:105–110. PMID: 11277309.
Article20. Ammann RW, Raimondi S, Maisonneuve P, Mullhaupt B. Zurich Pancreatitis Study Group. Is obesity an additional risk factor for alcoholic chronic pancreatitis? Pancreatology. 2010; 10:47–53. PMID: 20332661.21. Landau Z, Forti E, Alcaly M, Birk RZ. Palmitate induced lipoapoptosis of exocrine pancreas AR42J cells. Apoptosis. 2006; 11:717–724. PMID: 16532273.
Article22. Spector AA. Fatty acid binding to plasma albumin. J Lipid Res. 1975; 16:165–179. PMID: 236351.
Article23. Park YH, Kim YJ, Son IH, Yang HD. Inhibition of beta-amyloid(1-40) Peptide Aggregation and Neurotoxicity by Citrate. Korean J Physiol Pharmacol. 2009; 13:273–279. PMID: 19885010.24. Hirasawa A, Hara T, Katsuma S, Adachi T, Tsujimoto G. Free fatty acid receptors and drug discovery. Biol Pharm Bull. 2008; 31:1847–1851. PMID: 18827341.
Article25. Chavez JA, Summers SA. Characterizing the effects of saturated fatty acids on insulin signaling and ceramide and diacylglycerol accumulation in 3T3-L1 adipocytes and C2C12 myotubes. Arch Biochem Biophys. 2003; 419:101–109. PMID: 14592453.
Article26. Azevedo-Martins AK, Monteiro AP, Lima CL, Lenzen S, Curi R. Fatty acid-induced toxicity and neutral lipid accumulation in insulin-producing RINm5F cells. Toxicol In Vitro. 2006; 20:1106–1113. PMID: 16644178.
Article27. Otsuki M, Yamamoto M, Yamaguchi T. Animal models of chronic pancreatitis. Gastroenterol Res Pract. 2010; 2010:403295. PMID: 21197438.
Article28. Han MS, Park SY, Shinzawa K, Kim S, Chung KW, Lee JH, Kwon CH, Lee KW, Lee JH, Park CK, Chung WJ, Hwang JS, Yan JJ, Song DK, Tsujimoto Y, Lee MS. Lysophosphatidylcholine as a death effector in the lipoapoptosis of hepatocytes. J Lipid Res. 2008; 49:84–97. PMID: 17951222.
Article29. Maedler K, Oberholzer J, Bucher P, Spinas GA, Donath MY. Monounsaturated fatty acids prevent the deleterious effects of palmitate and high glucose on human pancreatic beta-cell turnover and function. Diabetes. 2003; 52:726–733. PMID: 12606514.30. Masuoka HC, Mott J, Bronk SF, Werneburg NW, Akazawa Y, Kaufmann SH, Gores GJ. Mcl-1 degradation during hepatocyte lipoapoptosis. J Biol Chem. 2009; 284:30039–30048. PMID: 19734538.
Article31. Allagnat F, Cunha D, Moore F, Vanderwinden JM, Eizirik DL, Cardozo AK. Mcl-1 downregulation by pro-inflammatory cytokines and palmitate is an early event contributing to β-cell apoptosis. Cell Death Differ. 2011; 18:328–337. PMID: 20798690.
Article32. Coll T, Eyre E, Rodriguez-Calvo R, Palomer X, Sánchez RM, Merlos M, Laguna JC, Vazquez-Carrera M. Oleate reverses palmitate-induced insulin resistance and inflammation in skeletal muscle cells. J Biol Chem. 2008; 283:11107–11116. PMID: 18281277.
Article33. Nemcova-Furstova V, James RF, Kovar J. Inhibitory effect of unsaturated fatty acids on saturated fatty acid-induced apoptosis in human pancreatic β-cells: activation of caspases and ER stress induction. Cell Physiol Biochem. 2011; 27:525–538. PMID: 21691070.
Article34. Nolan CJ, Larter CZ. Lipotoxicity: why do saturated fatty acids cause and monounsaturates protect against it? J Gastroenterol Hepatol. 2009; 24:703–706. PMID: 19646010.
Article35. Cases S, Smith SJ, Zheng YW, Myers HM, Lear SR, Sande E, Novak S, Collins C, Welch CB, Lusis AJ, Erickson SK, Farese RV Jr. Identification of a gene encoding an acyl CoA:diacylglycerol acyltransferase, a key enzyme in triacylglycerol synthesis. Proc Natl Acad Sci USA. 1998; 95:13018–13023. PMID: 9789033.
Article36. Oelkers P, Behari A, Cromley D, Billheimer JT, Sturley SL. Characterization of two human genes encoding acyl coenzyme A:cholesterol acyltransferase-related enzymes. J Biol Chem. 1998; 273:26765–26771. PMID: 9756920.
Article37. Jogl G, Tong L. Crystal structure of carnitine acetyltransferase and implications for the catalytic mechanism and fatty acid transport. Cell. 2003; 112:113–122. PMID: 12526798.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Differential effects of saturated and unsaturated fatty acids on vascular reactivity in isolated mesenteric and femoral arteries of rats
- Serum Phospholipid Fatty Acids in Benign Breast Tumor and Breast Cancer
- Effects of C18 Fatty Acids on Intracellular Ca2+ Mobilization and Histamine Release in RBL-2H3 Cells
- Emodin exerts protective effect against palmitic acid-induced endoplasmic reticulum stress in HepG2 cells
- Serum Fatty Acid and Carnitine Levels in Obese Children with Fatty Livers