Ann Lab Med.
2012 May;32(3):216-219. 10.3343/alm.2012.32.3.216.
Analysis of Factors Influencing the Generation of Unqualified Clinical Samples and Measures to Prevent this Generation
- Affiliations
-
- 1Department of Laboratory Medicine, the First Affiliated Hospital of Chongqing Medical University, Chongqing, P.R. China. lxf224234@hotmail.com
- KMID: 1381688
- DOI: http://doi.org/10.3343/alm.2012.32.3.216
Abstract
- BACKGROUND
We investigated the influence of pre-analytical factors on the results of clinical tests and thereby analyzed approaches to improve quality management in clinical laboratories.
METHODS
Unqualified clinical samples were selected from all the samples received at our clinical laboratory. The data were collected for 2009 and 2010, i.e., the years before and after the establishment of the laboratory quality management system. The rate and causes of generation of unqualified samples were analyzed, and measures to improve the laboratory practices were studied and implemented.
RESULTS
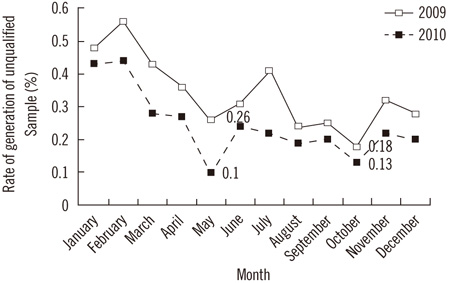
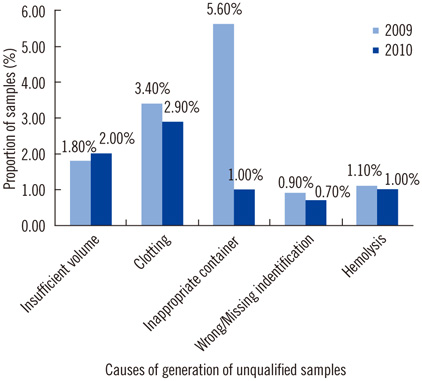
A total of 1,051 unqualified samples were identified from among the 553,158 samples (the overall incidence rate of unqualified samples was 0.19%). The number of unqualified samples substantially varied according to the nature of the sample, and clinical samples collected for routine blood tests or coagulation tests were the predominant unqualified samples. The main causes of generation of unqualified samples were insufficient sample volumes and improper methods of mixing the samples. The rate of generation of unqualified samples decreased significantly after the implementation of improvement measures (0.26% in 2009 vs. 0.13% in 2010, P<0.001).
CONCLUSIONS
The number of unqualified samples decreased significantly after the establishment of the laboratory quality management system, which promoted active communication among and training of the clinical staff to reduce the occurrence of pre-analytical errors. Comprehensive control of pre-analytical factors is an important approach in improving the clinical laboratory practices.
Keyword
MeSH Terms
Figure
Reference
-
1. Stankovic AK. The laboratory is a key partner in assuring patient safety. Clin Lab Med. 2004. 24:1023–1035.
Article2. Rivers PA, Dobalian A, Germinario FA. A review and analysis of the clinical laboratory improvement amendment of 1988: compliance plans and enforcement policy. Health Care Manage Rev. 2005. 30:93–102.3. Johnson PR. The contribution of proficiency testing to improving laboratory performance and ensuring quality patient care. Clin Leadersh Manag Rev. 2004. 18:335–341.4. Howanitz PJ. Errors in laboratory medicine: practical lessons to improve patient safety. Arch Pathol Lab Med. 2005. 129:1252–1261.
Article5. Bonini P, Plebani M, Ceriotti F, Ceriotti F. Errors in laboratory medicine. Clin Chem. 2002. 48:691–698.
Article6. Plebani M. Appropriateness in programs for continuous quality improvement in clinical laboratories. Clin Chim Acta. 2003. 333:131–139.
Article7. Plebani M, Carraro P. Mistakes in a stat laboratory: types and frequency. Clin Chem. 1997. 43:1348–1351.
Article8. Lippi G, Bassi A, Brocco G, Montagnana M, Salvagno GL, Guidi GC. Preanalytic error tracking in a laboratory medicine department: results of a 1-year experience. Clin Chem. 2006. 52:1442–1443.
Article9. Howanitz PJ. Errors in laboratory medicine: practical lessons to improve patient safety. Arch Pathol Lab Med. 2005. 129:1252–1261.
Article10. Saxena S, Kempf R, Wilcox S, Shulman IA, Wong L, Cunningham G, et al. Critical laboratory value notification: a failure mode effects and criticality analysis. Jt Comm J Qual Patient Saf. 2005. 31:495–506.
Article11. Ricós C, Garcia-Victoria M, de la Fuente B. Quality indicators and specifications for the extra-analytical phases in clinical laboratory management. Clin Chem Lab Med. 2004. 42:578–582.
Article12. Barenfanger J, Sautter RL, Lang DL, Collins SM, Hacek DM, Peterson LR, Improving patient. read-back) telephone reports of critical information. Am J Clin Pathol. 2004. 121:801–803.13. Berte LM. Patient safety: getting there from here-quality management is the best patient safety program. Clin Leadersh Manag Rev. 2004. 18:311–315.14. Bachner P. Quality assurance of the analytic process: pre- and postanalytic variation. Clin Lab Med. 1986. 6:613–623.
Article15. Lundberg GD. How clinicians should use the diagnostic laboratory in a changing medical world. Clin Chim Acta. 1999. 280:3–11.
Article16. Coiera E. Communication systems in healthcare. Clin Biochem Rev. 2006. 27:89–98.17. Rai AJ, Vitzthum F. Effects of preanalytical variables on peptide and protein measurement in human serum and plasma; implication of clinical proteomics. Expert Rev Proteomics. 2006. 3:409–426.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Comparison of Suicide Attempt by Generation During the COVID-19 Pandemic: Focusing on the Younger Generation
- Factors Influencing Anxiety in MZ Generation Workers: Occupational Stress, Social Problem Solving, Work as Meaning, and Work-Life Balance
- Factors Influencing Subjective Quality of Life in Male Baby Boom Generation Men
- A structural equation model of organizational commitment by hospital nurses: The moderating effect of each generation through multi-group analysis
- The Mediating Effect of Work Friendship on the Relationship between Grit and Work Engagement among Millennial and Generation Z Nurses in Korea