Clin Orthop Surg.
2009 Jun;1(2):74-80. 10.4055/cios.2009.1.2.74.
Surgical Results of Intradural Extramedullary Tumors
- Affiliations
-
- 1Department of Orthopaedic Surgery, Kangdong Sacred Heart Hospital, Hallym University School of Medicine, Seoul, Korea. skw@hallym.or.kr
- KMID: 1122636
- DOI: http://doi.org/10.4055/cios.2009.1.2.74
Abstract
-
BACKGROUND: To report the treatment results of 12 patients who underwent a total excision of intradural extramedullary tumors.
METHODS
Twelve cases of histopathologically confirmed intradural extramedullary tumors were treated surgically between February 2002 and March 2005. There were 8 males and 4 females with an average age of 42.6 years. The mean postoperative follow-up period was 24.2 months. The histopathological findings, locations of the tumors, and clinical results were analyzed. The neurological findings obtained during the preoperative stage and the postoperative follow-up were evaluated according to the Frankel classification.
RESULTS
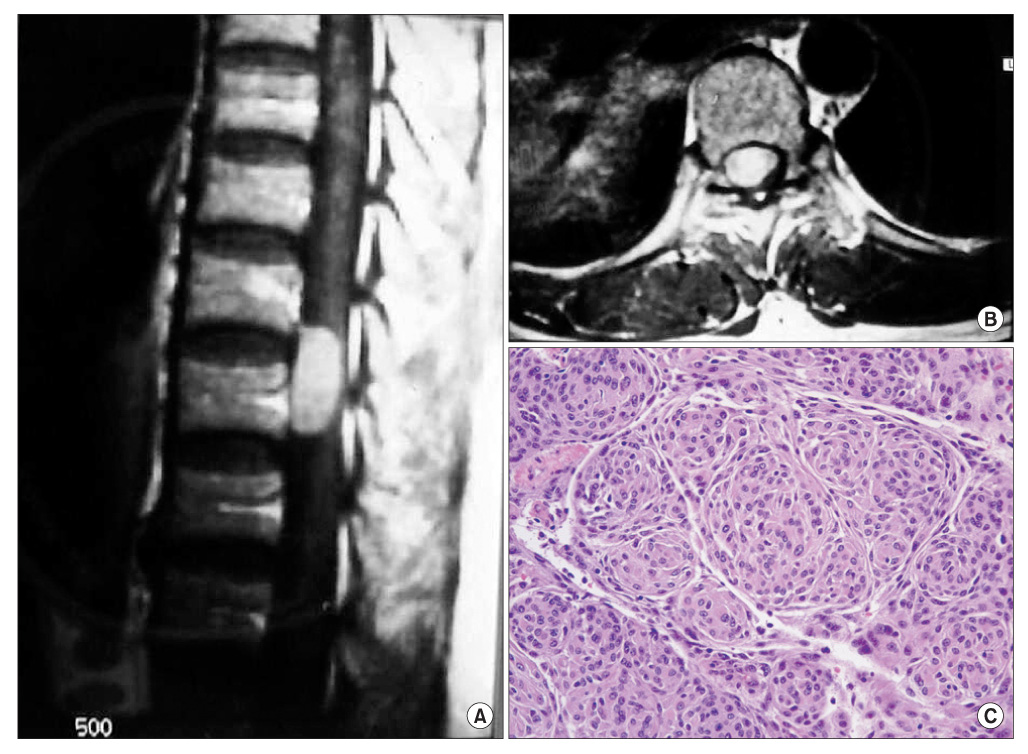
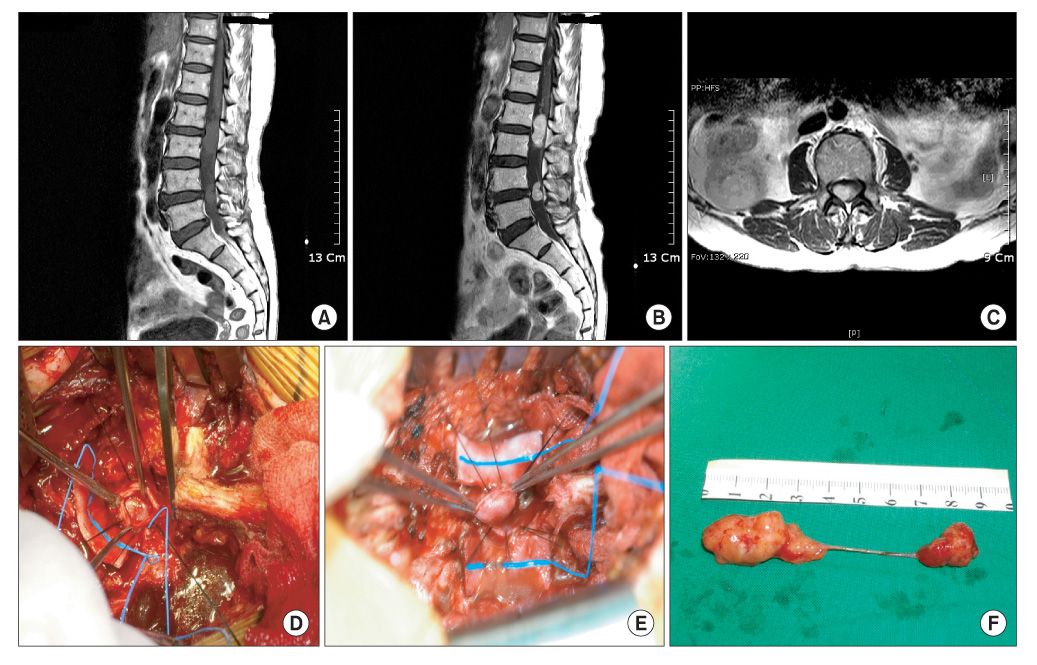
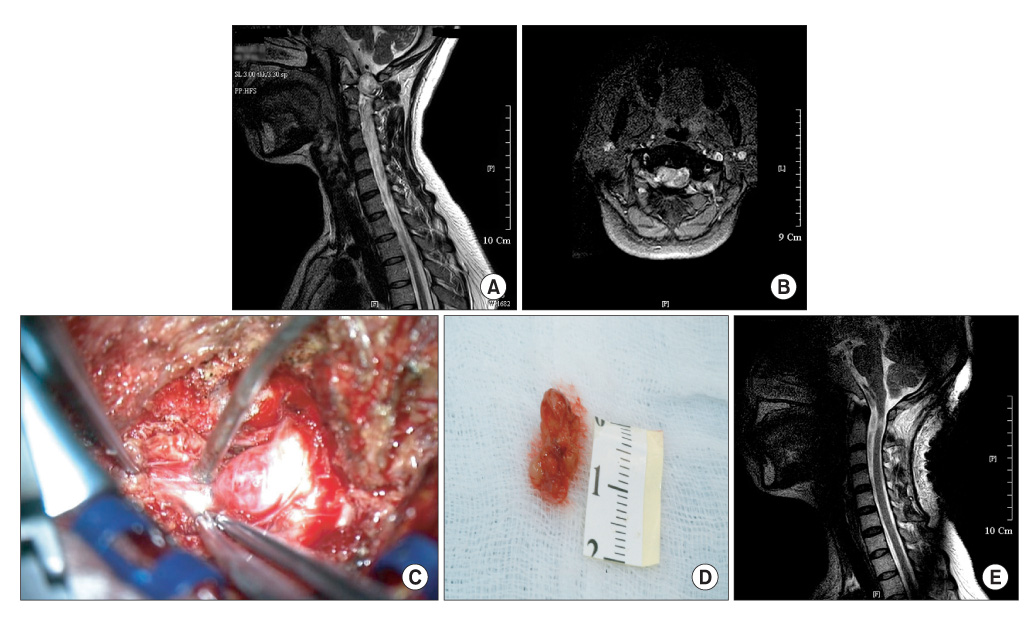
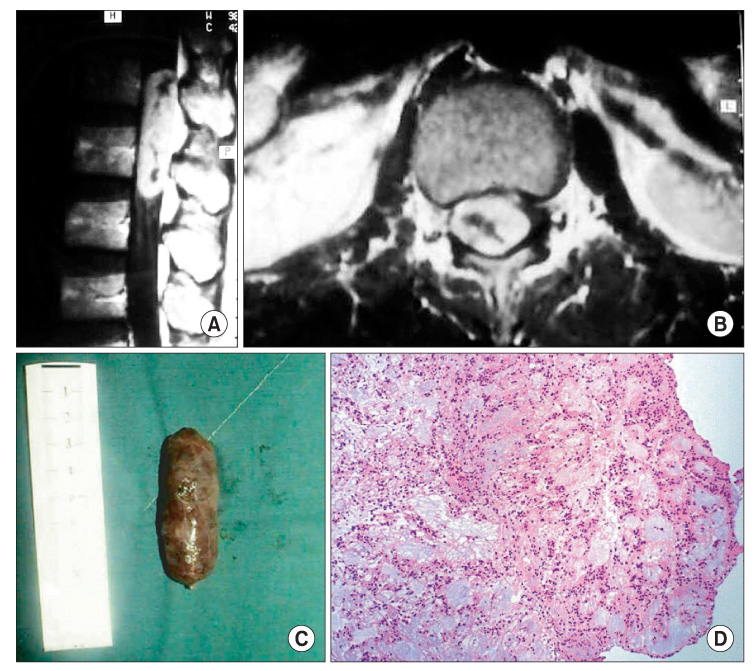
The histopathological results are as follows: 4 cases of a meningioma, 4 cases of a schwannoma, 2 cases of an epidermoid cyst, 1 case of an arachnoid cyst, and 1 case of an ependymoma. The locations of the tumors were as follows: 7 cases in the thoracic region, 4 cases in the lumbar region, and 1 case in the cervical region. At the final follow-up, a 2-grade and 1-grade improvement was observed in 1 and 7 cases, respectively. There were no changes in the Frankel grade in 4 cases. The preoperative neurological deficit improved within 8 postoperative weeks in most cases and within 1 postoperative year in all cases. Postoperatively, there were 2 cases of cerebrospinal fluid leakage and 2 cases of paresthesia.
CONCLUSIONS
Intradural extramedullary tumors detected by MRI are mostly benign and good clinical results can be obtained when treated surgically. Therefore, more active surgical approaches by orthopedic surgeons are recommended to decrease morbidity.
Keyword
MeSH Terms
Figure
Reference
-
1. Nittner K. Vinken PJ, Bruyn GW, editors. Spinal meningiomas, neurinomas, and neurofibroma-hourglass tumors. Handbook of clinical neurology. 1976. Vol. 20. Amsterdam: North-Holland Publishing Co;177–322.2. Cheng MK. Spinal cord tumors in the People's Republic of China: a statistical review. Neurosurgery. 1982. 10(1):22–24.3. Simeone FA. Yomans JR, editor. Spinal cord tumors in adult. Neurological surgery. 1990. 3rd ed. Philadelphia: WB Saunders Co;3531–3547.4. Shin BJ, Lee JC, Yoon TK, et al. Surgical treatments of intradural extramedullary tumor. J Korean Soc Spine Surg. 2002. 9(3):230–237.
Article5. Nittner K. Vinken PJ, Bruyn GW, editors. Tumors of the spine and spinal cord. Handbook of clinical neurology. 1976. Vol. 20. Amsterdam: North-Holland Publishing Co;238–289.6. Harry N. The spine. 1998. 4th ed. Philadelphia: WB Saunders Co;1366.7. Kono K, Inoue Y, Nakamura H, Shakudo M, Nakayama K. MR imaging of a case of a dumbbell-shaped spinal schwannoma with intramedullary and intradural-extramedullary components. Neuroradiology. 2001. 43(10):864–867.
Article8. Gelabert-Gonzalez M, Garcia-Allut A, Martinez-Rumbo R. Spinal meningiomas. Neurocirugia (Astur). 2006. 17(2):125–131.9. Cohen AR, Wisoff JH, Allen JC, Epstein F. Malignant astrocytomas of the spinal cord. J Neurosurg. 1989. 70(1):50–54.
Article10. Cooper PR, Epstein F. Radical resection of intramedullary spinal cord tumors in adults: recent experience in 29 patients. J Neurosurg. 1985. 63(4):492–499.11. Cristante L, Herrmann HD. Surgical management of intramedullary spinal cord tumors: functional outcome and sources of morbidity. Neurosurgery. 1994. 35(1):69–74.12. Samii M, Klekamp J. Surgical results of 100 intramedullary tumors in relation to accompanying syringomyelia. Neurosurgery. 1994. 35(5):865–873.
Article13. Klekamp J, Samii M. Surgical results for spinal meningiomas. Surg Neurol. 1999. 52(6):552–562.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Intradural Extramedullary Non-infiltrated Solitary Metastatic Tumor
- Removal of Intradural-Extramedullary Spinal Cord Tumors with Unilateral Limited Laminectomy
- Non-Enhancing Intradural Extramedullary Ependymoma: A Case Report
- Intraspinal Myxoid Chondrosarcoma: Case Report
- Exploring Unilateral Biportal Endoscopy for Lumbar Intradural Lesions: A Technical and Video Report on Benefits and Key Considerations