J Vet Sci.
2010 Mar;11(1):27-34. 10.4142/jvs.2010.11.1.27.
Fatal cases of Theileria annulata infection in calves in Portugal associated with neoplastic-like lymphoid cell proliferation
- Affiliations
-
- 1Instituto de Ciencias Agrarias e Ambientais Mediterranicas, Departamento de Medicina Veterinaria, Universidade de Evora, Polo da Mitra, Ap.94, 7002-554 Evora, Portugal. smbb@uevora.pt
- 2University of California, Veterinary Medical Center, 10435 Sorrento Valley Road, Suite 101 San Diego, USA.
- 3Instituto de Investigacao Cientifica Tropical, CVZ, Faculdade de Medicina Veterinaria, Avenida da Universidade Tecnica, 1300-477 Lisboa, Portugal.
- 4Centro de Investigacao Interdisciplinar em Sanidade Animal (CIISA), Faculdade de Medicina Veterinaria, Avenida da Universidade Tecnica, 1300-477 Lisboa, Portugal.
- 5Vet+ Servicos Veterinarios Lda., Largo Machado Santos Lote 16, 7050-125 Montemor-o-Novo, Portugal.
- 6Cooperativa Agricola de Compra e Venda de Montemor-o-Novo, CRL (COPRAPEC), Rua 5 Outubro 76-1 degrees, 7050-355 Montemor-o-Novo, Portugal.
- 7Centro de Investigacao em Patobiologia Molecular, Instituto Portugues de Oncologia, Rua Prof. Lima Basto, 1099-023 Lisboa, Portugal.
- 8Laboratorio Nacional de Investigacao Veterinaria, Estrada de Benfica 701, 1549-011 Lisboa, Portugal.
- 9Division of Infection and Immunity, Institute of Comparative Medicine, Faculty of Veterinary Medicine, University of Glasgow, Bearsden Road, Glasgow G61 1QH, Scotland.
- KMID: 1106154
- DOI: http://doi.org/10.4142/jvs.2010.11.1.27
Abstract
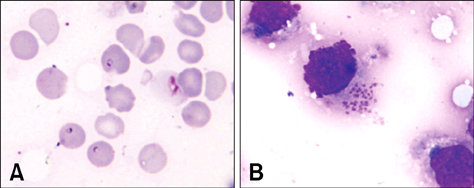
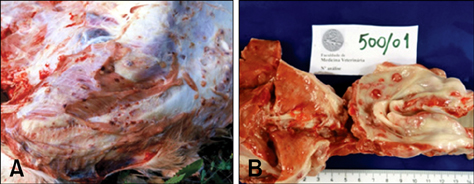
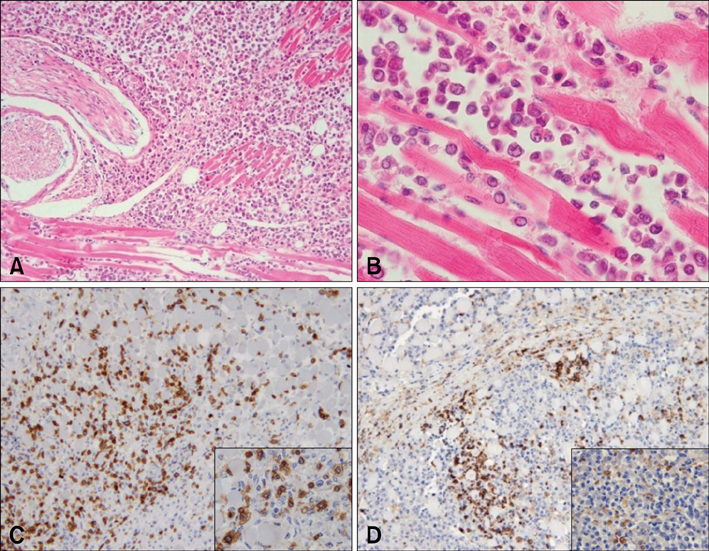
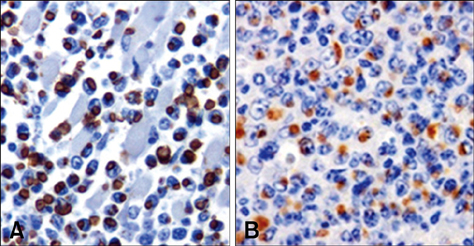
- This study was carried out to investigate fifteen cases of acute lethal infection of calves (< or = 4 months of age) by the protozoan parasite Theileria (T.) annulata in the south of Portugal. Calves developed multifocal to coalescent nodular skin lesions, similar to multicentric malignant lymphoma. Infestation with ticks (genus Hyalomma) was intense. Theileria was seen in blood and lymph node smears, and T. annulata infection was confirmed by isolation of schizont-transformed cells and sequencing of hypervariable region 4 of the 18S rRNA gene. At necropsy, hemorrhagic nodules or nodules with a hemorrhagic halo were seen, particularly in the skin, subcutaneous tissue, skeletal and cardiac muscles, pharynx, trachea and intestinal serosa. Histologically, nodules were formed by large, round, lymphoblastoid neoplastic-like cells. Immunohistochemistry (IHC) identified these cells as mostly CD3 positive T lymphocytes and MAC387 positive macrophages. A marker for B lymphocytes (CD79alphacy) labeled very few cells. T. annulata infected cells in these nodules were also identified by IHC through the use of two monoclonal antibodies (1C7 and 1C12) which are diagnostic for the parasite. It was concluded that the pathological changes observed in the different organs and tissues were caused by proliferation of schizont-infected macrophages, which subsequently stimulate a severe uncontrolled proliferation of uninfected T lymphocytes.
MeSH Terms
-
Animals
Base Sequence
Cattle
Cattle Diseases/epidemiology/*parasitology/pathology
Cell Growth Processes/physiology
DNA, Protozoan/chemistry/genetics
Disease Outbreaks/*veterinary
Female
Immunohistochemistry/veterinary
Lymphocytes/parasitology/*pathology
Male
Molecular Sequence Data
Polymerase Chain Reaction/veterinary
Portugal/epidemiology
RNA, Ribosomal, 18S/chemistry/genetics
Sequence Analysis, DNA
Skin Diseases/epidemiology/parasitology/pathology/*veterinary
Theileria annulata/*isolation & purification
Theileriasis/epidemiology/parasitology/*pathology
Figure
Reference
-
1. Adamson R, Hall R. A role for matrix metalloproteinases in the pathology and attenuation of Theileria annulata infections. Parasitol Today. 1997. 13:390–393.
Article2. Adamson R, Logan M, Kinnaird J, Langsley G, Hall R. Loss of matrix metalloproteinase 9 activity in Theileria annulata-attenuated cells is at the transcriptional level and is associated with differentially expressed AP-1 species. Mol Biochem Parasitol. 2000. 106:51–61.
Article3. Ahmed JS, Schnittger L, Mehlhorn H. Review: Theileria schizonts induce fundamental alterations in their host cells. Parasitol Res. 1999. 85:527–538.
Article4. Biermann R, Schnittger L, Beyer D, Ahmed JS. Initiation of translation and cellular localization of Theileria annulata casein kinase IIα: implication for its role in host cell transformation. J Cell Physiol. 2003. 196:444–453.
Article5. Brown CGD. Control of Tropical Theileriosis (Theileria annulata infection) of cattle. Parassitologia. 1990. 32:23–31.6. Brown CGD. Dynamics and impact of tick-borne diseases of cattle. Trop Anim Health Prod. 1997. 29:1S–3S.
Article7. Brown DJ, Campbell JD, Russell GC, Hopkins J, Glass EJ. T cell activation by Theileria annulata-infected macrophages correlates with cytokine production. Clin Exp Immunol. 1995. 102:507–514.
Article8. Campbell JD, Brown DJ, Glass EJ, Hall FR, Spooner RL. Theileria annulata sporozoite targets. Parasite Immunol. 1994. 16:501–505.9. Campbell JD, Howie SE, Odling KA, Glass EJ. Theileria annulata induces abberrant T cell activation in vitro and in vivo. Clin Exp Immunol. 1995. 99:203–210.
Article10. Campbell JDM, Spooner RL. Macrophages behaving badly: infected cells and subversion of immune responses to Theileria annulata. Parasitol Today. 1999. 15:10–16.11. Collins RA, Sopp P, Gelder KI, Morrison WI, Howard CJ. Bovine gamma/delta TcR+ T lymphocytes are stimulated to proliferate by autologous Theileria annulata-infected cells in the presence of interleukin-2. Scand J Immunol. 1996. 44:444–452.
Article12. DeMartini JC, Moulton JE. Responses of the bovine lymphatic system to infection by Theileria parva. I. Histology and ultrastructure of lymph nodes in experimentally-infected calves. J Comp Pathol. 1973. 83:281–298.
Article13. Dobbelaere D, Heussler V. Transformation of leukocytes by Theileria parva and T. annulata. Annu Rev Microbiol. 1999. 53:1–42.14. D'Oliveira C, van der Weide M, Habela MA, Jacquiet P, Jongejan F. Detection of Theileria annulata in blood samples of carrier cattle by PCR. J Clin Microbiol. 1995. 33:2665–2669.15. Forsyth LMG, Minns FC, Kirvar E, Adamson RE, Hall FR, McOrist S, Brown CGD, Preston PM. Tissue damage in cattle infected with Theileria annulata accompanied by metastasis of cytokine-producing, schizont-infected mononuclear phagocytes. J Comp Pathol. 1999. 120:39–57.
Article16. Gill BS, Bhattacharyulu Y, Kaur D. Symptoms and Pathology of experimental bovine tropical theileriosis (Theileria annulata infection). Ann Parasitol Hum Comp. 1977. 52:597–608.
Article17. Glascodine J, Tetley L, Tait A, Brown D, Shiels B. Developmental expression of a Theileria annulata merozoite surface antigen. Mol Biochem Parasitol. 1990. 40:105–112.
Article18. Glass EJ. The balance between protective immunity and pathogenesis in tropical theileriosis: what we need to know to design effective vaccines for the future. Res Vet Sci. 2001. 70:71–75.
Article19. Graham SP, Brown DJ, Vatansever Z, Waddington D, Taylor LH, Nichani AK, Campbell JDM, Adamson RE, Glass EJ, Spooner RL. Proinflammatory cytokine expression by Theileria annulata infected cell lines correlates with the pathology they cause in vivo. Vaccine. 2001. 19:2932–2944.
Article20. Leemans I, Brown D, Hooshmand-Rad P, Kirvar E, Uggla A. Infectivity and cross-immunity studies of Theileria lestoquardi and Theileria annulata in sheep and cattle: I. In vivo responses. Vet Parasitol. 1999. 82:179–192.
Article21. Leitão JLS. Teileriose bovina em Portugal. Repositório de Trabalhos do Laboratório Central de Patologia Veterinária. 1945. II:1–8.22. Manuja A, Malhotra DV, Sikka VK, Sangwan AK, Sharma R, Kumar B, Mehta BD, Gulati BR, Nichani AK. Isolates of Theileria annulata collected from different parts of India show phenotypic and genetic diversity. Vet Parasitol. 2006. 137:242–252.
Article23. Mehlhorn H, Schein E. The piroplasms: life cycle and sexual stages. Adv Parasitol. 1984. 23:37–103.
Article24. Moreau M, Thibaud J, Miled LB, Chaussepied M, Baumgartner M, Davis WC, Minoprio P, Langsley G. Theileria annulata in CD5+ macrophages and B1 B cells. Infect Immun. 1999. 67:6678–6682.
Article25. Nichani AK, Thorp BH, Brown CGD, Campbell JDM, Brown DJ, Ritchie M, Spooner RL. In vivo development of Theileria annulata: major changes in efferent lymph following infection with sporozoites or allogeneic schizont-infected mononuclear cells. Parasitology. 1999. 118:327–333.
Article26. Preston PM, Brown CGD, Entrican G, Richardson W, Boid R. Synthesis of tumour necrosis factor-alpha and interferons by mononuclear cells from Theileria annulata-infected cattle. Parasite Immunol. 1993. 15:525–534.
Article27. Preston PM, Brown CG, Bell-Sakyi L, Richardson W, Sanderson A. Tropical theileriosis in Bos taurus and Bos taurus cross Bos indicus calves: response to infection with graded doses of sporozoites of Theileria annulata. Res Vet Sci. 1992. 53:230–243.
Article28. Preston PM, Hall FR, Glass EJ, Campbell JDM, Darghouth MA, Ahmed JS, Shiels BR, Spooner RL, Jongejan F, Brown CGD. Innate and adaptive immune responses co-operate to protect cattle against Theileria annulata. Parasitol Today. 1999. 15:268–274.
Article29. Preston PM, Jongejan F. Protective immune mechanisms to ticks and tick-borne diseases of ruminants. Parasitol Today. 1999. 15:255–258.
Article30. Renneker S, Kullmann B, Gerber S, Dobschanski J, Bakheit MA, Geysen D, Shiels B, Tait A, Ahmed JS, Seitzer U. Development of a competitive ELISA for detection of Theileria annulata infection. Transbound Emerg Dis. 2008. 55:249–256.
Article31. Rozen S, Skaletsky H. Misener S, Krawetz SA, editors. Primer3 on the WWW for general users and for biologist programmers. Bioinformatics Methods and Protocols. 2000. Totowa: Humana Press;365–386.
Article32. Sager H, Bertoni G, Jungi TW. Differences between B cell and macrophage transformation by the bovine parasite, Theileria annulata: a clonal approach. J Immunol. 1998. 161:335–341.33. Sager H, Davis WC, Dobbelaere DA, Jungi TW. Macrophage-parasite relationship in theileriosis. Reversible phenotypic and functional dedifferentiation of macrophages infected with Theileria annulata. J Leukoc Biol. 1997. 61:459–468.
Article34. Sharma G, Sharma RD, Nichani AK. Successful long-term in vitro cultivation of Theileria annulata schizonts in media supplemented with homologous and heterologous sera. Vet Parasitol. 1998. 79:135–141.
Article35. Shiels B, Langsley G, Weir W, Pain A, McKellar S, Dobbelaere D. Alteration of host cell phenotype by Theileria annulata and Theileria parva: mining for manipulators in the parasite genomes. Int J Parasitol. 2006. 36:9–21.
Article36. Shiels B, McDougall C, Tait A, Brown CGD. Antigenic diversity of Theileria annulata macroschizonts. Vet Parasitol. 1986. 21:1–10.37. Shiels BR, McDougall C, Tait A, Brown CGD. Identification of Infection-associated antigens in Theileria annulata transformed cells. Parasite Immunol. 1986. 8:69–77.
Article38. Spitalska E, Torina A, Cannella V, Caracappa S, Sparagano OA. Discrimination between Theileria lestoquardi and Theileria annulata in their vectors and hosts by RFLP based on the 18S rRNA gene. Parasitol Res. 2004. 94:318–320.
Article39. Spooner RL, Innes EA, Glass EJ, Brown CGD. Theileria annulata and T. parva infect and transform different bovine mononuclear cells. Immunology. 1989. 66:284–288.40. Valli VEO. Maxie MG, editor. The hematopoietic system. Jubb, Kennedy & Palmer's Pathology of Domestic Animals. 2007. Vol. 2:5th ed. Philadelphia: Saunders;199–201.41. Zhang Z, Schwartz S, Wagner L, Miller W. A greedy algorithm for aligning DNA sequences. J Comput Biol. 2000. 7:203–214.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Treatment of natural tropical theileriosis with the extract of the plant Peganum harmala
- Biochemical, Hematological, and Electrocardiographic Changes in Buffaloes Naturally Infected with Theileria annulata
- Validation of Reference Genes for Quantitative Real-Time PCR in Bovine PBMCs Transformed and Non-transformed by Theileria annulata
- The genus Hypoxylon, Wood Decay Fungi - II. Teleomorph of Annulata Section
- Intravascular Large B-cell Lymphoma with Bone Marrow Involvement: A Case Successfully Treated with a Rituximab-CHOP Combination Regimen