Ann Surg Treat Res.
2024 Mar;106(3):155-168. 10.4174/astr.2024.106.3.155.
Pioneering PGC-1αα–boosted secretome: a novel approach to combating liver fibrosis
- Affiliations
-
- 1Department of Surgery, Bucheon St. Mary’s Hospital, College of Medicine, the Catholic University of Korea, Seoul, Korea
- 2Department of Surgery, Seoul St. Mary’s Hospital, College of Medicine, the Catholic University of Korea, Seoul, Korea
- 3Catholic Central Laboratory of Surgery, College of Medicine, the Catholic University of Korea, Seoul, Korea
- 4Department of Surgery, Eunpyeong St. Mary’s Hospital, College of Medicine, the Catholic University of Korea, Seoul, Korea
- 5Translational Research Team, Surginex Co., Ltd., Seoul, Korea
- 6Department of Surgery, Daejeon St. Mary’s Hospital, College of Medicine, the Catholic University of Korea, Seoul, Korea
- 7Department of Surgery, Uijeongbu St. Mary’s Hospital, College of Medicine, the Catholic University of Korea, Seoul, Korea
- KMID: 2553385
- DOI: http://doi.org/10.4174/astr.2024.106.3.155
Abstract
- Purpose
Liver fibrosis is a critical health issue with limited treatment options. This study investigates the potential of PGC-Sec, a secretome derived from peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α)-overexpressing adipose-derived stem cells (ASCs), as a novel therapeutic strategy for liver fibrosis.
Methods
Upon achieving a cellular confluence of 70%–80%, ASCs were transfected with pcDNA-PGC-1α. PGC-Sec, obtained through concentration of conditioned media using ultrafiltration units with a 3-kDa cutoff, was assessed through in vitro assays and in vitro mouse models.
Results
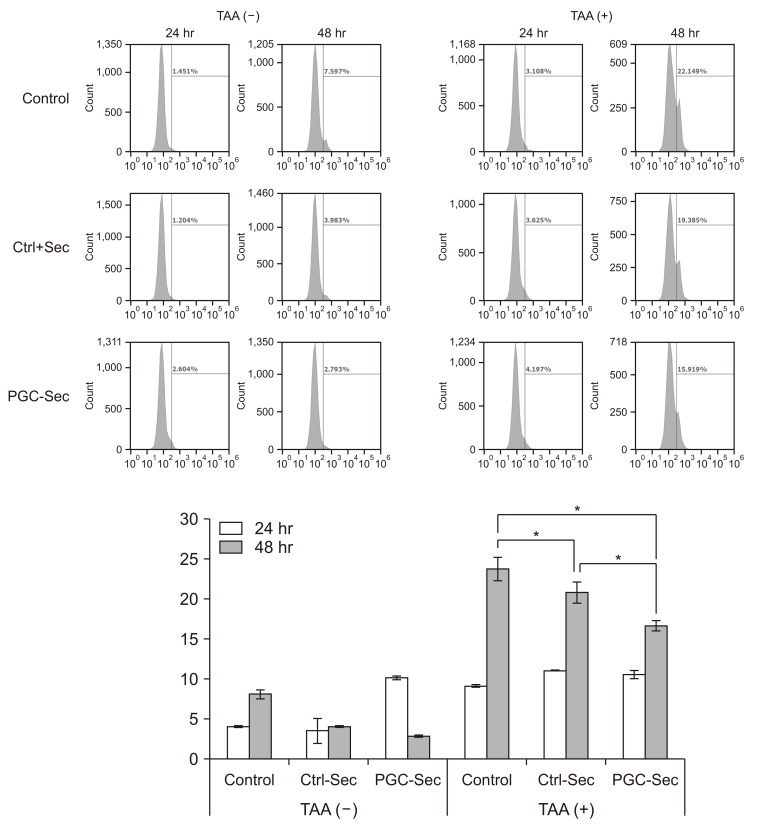
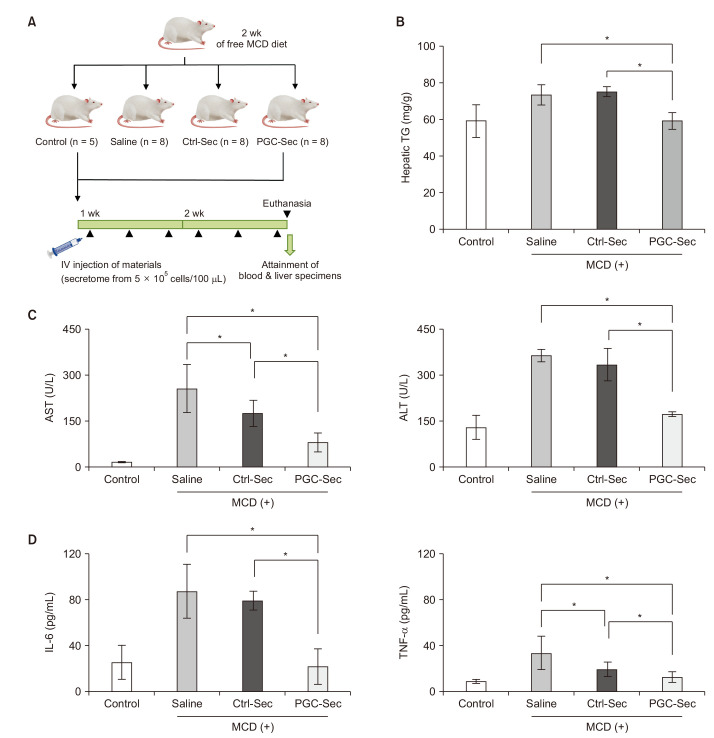
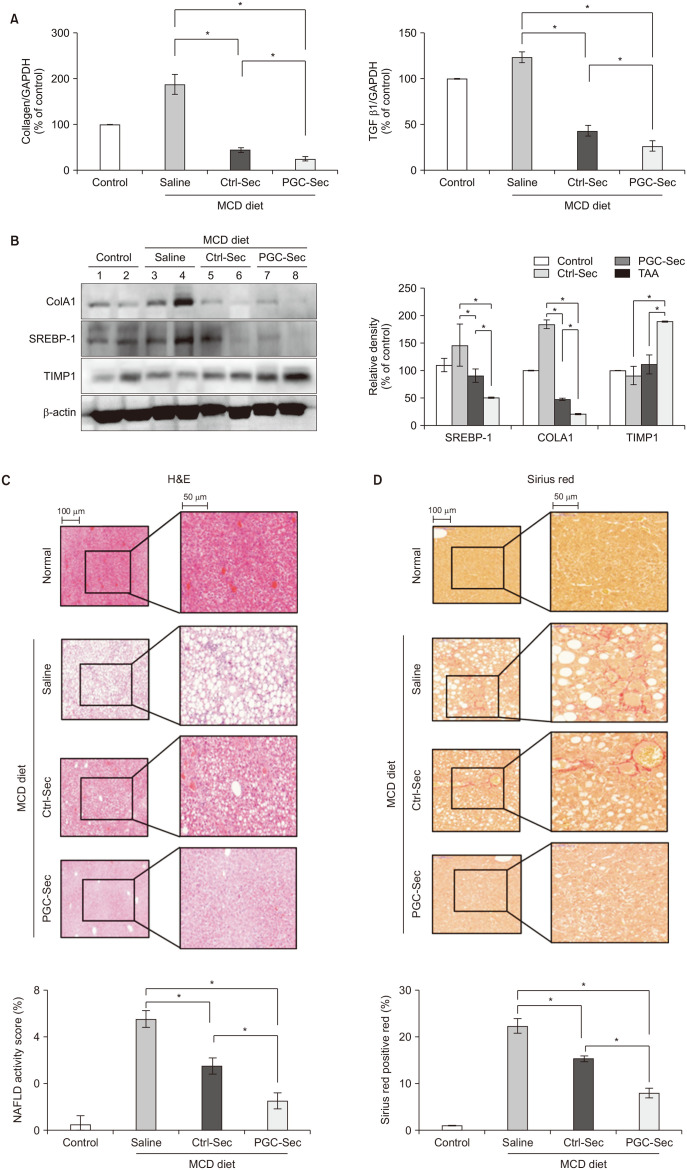
In vitro, PGC-Sec significantly reduced LX2 human hepatic stellate cell proliferation and mitigated mitochondrial oxidative stress compared to the control-secretome. In an in vivo mouse model, PGC-Sec treatment led to notable reductions in hepatic enzyme activity, serum proinflammatory cytokine concentrations, and fibrosis-related marker expression. Histological analysis demonstrated improved liver histology and reduced fibrosis severity in PGC-Sec–treated mice. Immunohistochemical staining confirmed enhanced expression of PGC-1α, optic atrophy 1 (a mitochondrial function marker), and peroxisome proliferator-activated receptor alpha (an antifibrogenic marker) in the PGC-Sec–treated group, along with reduced collagen type 1A expression (a profibrogenic marker).
Conclusion
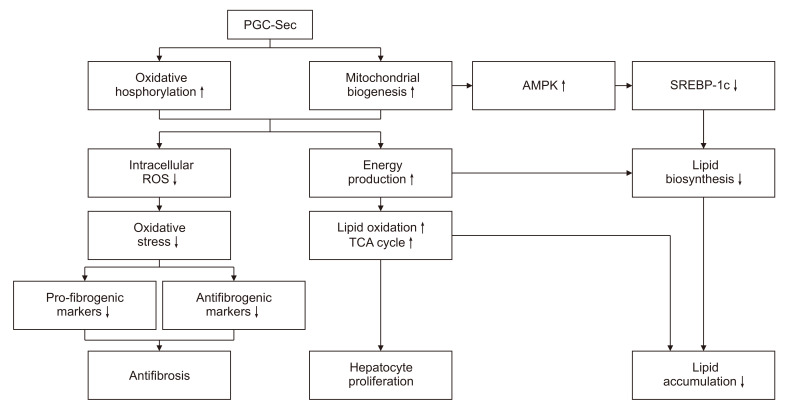
These findings highlight the therapeutic potential of PGC-Sec in combating liver fibrosis by enhancing mitochondrial biogenesis and function, and promoting antifibrotic processes. PGC-Sec holds promise as a novel treatment strategy for liver fibrosis.
Keyword
Figure
Reference
-
1. Pellicoro A, Ramachandran P, Iredale JP, Fallowfield JA. Liver fibrosis and repair: immune regulation of wound healing in a solid organ. Nat Rev Immunol. 2014; 14:181–194. PMID: 24566915.2. Geissler S, Textor M, Kühnisch J, Könnig D, Klein O, Ode A, et al. Functional comparison of chronological and in vitro aging: differential role of the cytoskeleton and mitochondria in mesenchymal stromal cells. PLoS One. 2012; 7:e52700. PMID: 23285157.3. Phinney DG, Pittenger MF. Concise review: MSC-derived exosomes for cell-free therapy. Stem Cells. 2017; 35:851–858. PMID: 28294454.4. Schieber M, Chandel NS. ROS function in redox signaling and oxidative stress. Curr Biol. 2014; 24:R453–R462. PMID: 24845678.5. Sies H, Jones DP. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat Rev Mol Cell Biol. 2020; 21:363–383. PMID: 32231263.6. Austin S, St-Pierre J. PGC1α and mitochondrial metabolism--emerging concepts and relevance in ageing and neurodegenerative disorders. J Cell Sci. 2012; 125:4963–4971. PMID: 23277535.7. LeBleu VS, O'Connell JT, Gonzalez Herrera KN, Wikman H, Pantel K, Haigis MC, et al. PGC-1α mediates mitochondrial biogenesis and oxidative phosphorylation in cancer cells to promote metastasis. Nat Cell Biol. 2014; 16:992–1003. 1–15. PMID: 25241037.8. Lee SC, Jeong HJ, Lee SK, Kim SJ. Hypoxic conditioned medium from human adipose-derived stem cells promotes mouse liver regeneration through JAK/STAT3 signaling. Stem Cells Transl Med. 2016; 5:816–825. PMID: 27102647.9. Kim OH, Hong HE, Seo H, Kwak BJ, Choi HJ, Kim KH, et al. Generation of induced secretome from adipose-derived stem cells specialized for disease-specific treatment: an experimental mouse model. World J Stem Cells. 2020; 12:70–86. PMID: 32110276.10. Paik KY, Kim KH, Park JH, Lee JI, Kim OH, Hong HE, et al. A novel antifibrotic strategy utilizing conditioned media obtained from miR-150-transfected adipose-derived stem cells: validation of an animal model of liver fibrosis. Exp Mol Med. 2020; 52:438–449. PMID: 32152450.11. Kim JG, Lee SC, Kim OH, Kim KH, Song KY, Lee SK, et al. HSP90 inhibitor 17-DMAG exerts anticancer effects against gastric cancer cells principally by altering oxidant-antioxidant balance. Oncotarget. 2017; 8:56473–56489. PMID: 28915605.12. Scarpulla RC. Metabolic control of mitochondrial biogenesis through the PGC-1 family regulatory network. Biochim Biophys Acta. 2011; 1813:1269–1278. PMID: 20933024.13. Summermatter S, Santos G, Pérez-Schindler J, Handschin C. Skeletal muscle PGC-1α controls whole-body lactate homeostasis through estrogen-related receptor α-dependent activation of LDH B and repression of LDH A. Proc Natl Acad Sci U S A. 2013; 110:8738–8743. PMID: 23650363.14. Lai L, Leone TC, Zechner C, Schaeffer PJ, Kelly SM, Flanagan DP, et al. Transcriptional coactivators PGC-1alpha and PGC-lbeta control overlapping programs required for perinatal maturation of the heart. Genes Dev. 2008; 22:1948–1961. PMID: 18628400.15. Zhang Q, Chen W, Xie C, Dai X, Ma J, Lu J. The role of PGC-1α in digestive system malignant tumours. Anticancer Agents Med Chem. 2020; 20:276–285. PMID: 31702508.16. Zhang Y, Shen L, Zhu H, Dreissigacker K, Distler D, Zhou X, et al. PGC-1α regulates autophagy to promote fibroblast activation and tissue fibrosis. Ann Rheum Dis. 2020; 79:1227–1233. PMID: 32482644.17. Wu L, Mo W, Feng J, Li J, Yu Q, Li S, et al. Astaxanthin attenuates hepatic damage and mitochondrial dysfunction in non-alcoholic fatty liver disease by up-regulating the FGF21/PGC-1α pathway. Br J Pharmacol. 2020; 177:3760–3777. PMID: 32446270.18. Lee G, Uddin MJ, Kim Y, Ko M, Yu I, Ha H. PGC-1α, a potential therapeutic target against kidney aging. Aging Cell. 2019; 18:e12994. PMID: 31313501.19. Han Y, Xu X, Tang C, Gao P, Chen X, Xiong X, et al. Reactive oxygen species promote tubular injury in diabetic nephropathy: the role of the mitochondrial ros-txnip-nlrp3 biological axis. Redox Biol. 2018; 16:32–46. PMID: 29475133.20. Cheng CF, Ku HC, Lin H. PGC-1α as a pivotal factor in lipid and metabolic regulation. Int J Mol Sci. 2018; 19:3447. PMID: 30400212.21. Baglio SR, Rooijers K, Koppers-Lalic D, Verweij FJ, Pérez Lanzón M, Zini N, et al. Human bone marrow- and adipose-mesenchymal stem cells secrete exosomes enriched in distinctive miRNA and tRNA species. Stem Cell Res Ther. 2015; 6:127. PMID: 26129847.22. Rani S, Ryan AE, Griffin MD, Ritter T. Mesenchymal stem cell-derived extracellular vesicles: toward cell-free therapeutic applications. Mol Ther. 2015; 23:812–823. PMID: 25868399.23. Jäger S, Handschin C, St-Pierre J, Spiegelman BM. AMP-activated protein kinase (AMPK) action in skeletal muscle via direct phosphorylation of PGC-1alpha. Proc Natl Acad Sci U S A. 2007; 104:12017–12022. PMID: 17609368.24. Suwa M, Egashira T, Nakano H, Sasaki H, Kumagai S. Metformin increases the PGC-1alpha protein and oxidative enzyme activities possibly via AMPK phosphorylation in skeletal muscle in vivo. J Appl Physiol (1985). 2006; 101:1685–1692. PMID: 16902066.25. Yap F, Craddock L, Yang J. Mechanism of AMPK suppression of LXR-dependent Srebp-1c transcription. Int J Biol Sci. 2011; 7:645–650. PMID: 21647332.26. Li W, Li Y, Wang Q, Yang Y. Crude extracts from Lycium barbarum suppress SREBP-1c expression and prevent diet-induced fatty liver through AMPK activation. Biomed Res Int. 2014; 2014:196198. PMID: 25013763.27. Han Y, Hu Z, Cui A, Liu Z, Ma F, Xue Y, et al. Post-translational regulation of lipogenesis via AMPK-dependent phosphorylation of insulin-induced gene. Nat Commun. 2019; 10:623. PMID: 30733434.28. Abu Shelbayeh O, Arroum T, Morris S, Busch KB. PGC-1α is a master regulator of mitochondrial lifecycle and ROS stress response. Antioxidants (Basel). 2023; 12:1075. PMID: 37237941.29. Halling JF, Pilegaard H. PGC-1α-mediated regulation of mitochondrial function and physiological implications. Appl Physiol Nutr Metab. 2020; 45:927–936. PMID: 32516539.30. Estornut C, Milara J, Bayarri MA, Belhadj N, Cortijo J. Targeting oxidative stress as a therapeutic approach for idiopathic pulmonary fibrosis. Front Pharmacol. 2021; 12:794997. PMID: 35126133.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Isolation of Secretome with Enhanced Antifibrotic Properties from miR-214-Transfected Adipose-Derived Stem Cells
- PGC1-alpha plays a role in hypothermic renal protection of renal fibrosis after acute kidney injury
- Hypothermia protects against renal fibrosis after ischemia reperfusion injury
- A novel strategy to promote liver regeneration: utilization of secretome obtained from survivin-overexpressing adipose-derived stem cells
- Effect of Ritonavir-boosting on Atazanavir Discontinuation due to Jaundice in HIV-infected Koreans