Dement Neurocogn Disord.
2016 Dec;15(4):129-134. 10.12779/dnd.2016.15.4.129.
Analysis of the Expectation of Stem Cell Therapy in Patients with Alzheimer’s Disease
- Affiliations
-
- 1Department of Neurology, Hanyang University College of Medicine, Seoul, Korea. ksh213@hanyang.ac.kr
- KMID: 2442838
- DOI: http://doi.org/10.12779/dnd.2016.15.4.129
Abstract
- BACKGROUND AND PURPOSE
Alzheimer's disease (AD) is the most common form of dementia which typically manifests as loss of memory and cognitive functions. Currently, available treatments for AD provide only symptomatic improvement and the benefit is minimal. Stem cell therapy (SCT) has been considered a promising treatment option for AD. We investigated the caregiver's perception about implementation of SCT in their AD patients, and determined the factors related to SCT.
METHODS
A total of 100 caregivers, who cared for their AD patients, were interviewed at two hospitals. Structured open and closed questions about SCT for AD were asked by trained interviewers using the conventional in-person method. In addition, 60 dementia-related physicians were randomly interviewed via an e-mail questionnaire.
RESULTS
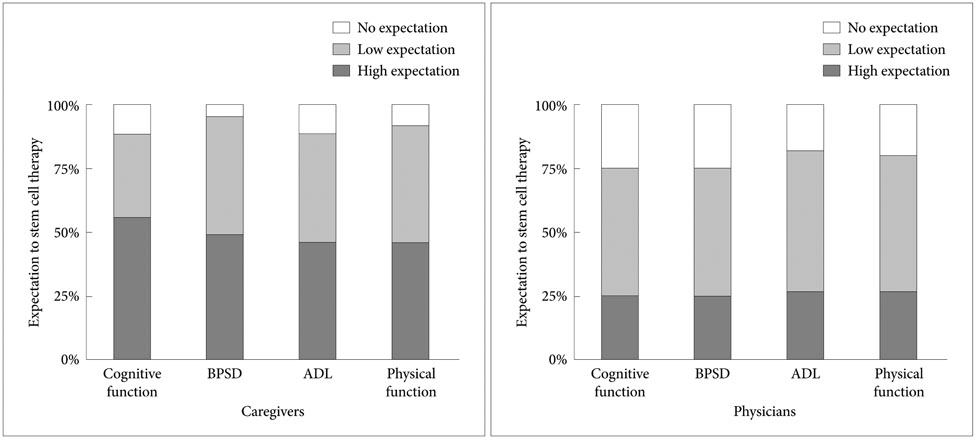
Of the 100 subjects, 61 caregivers replied that they wanted their AD patients to receive SCT. Approximately 50% of the caregivers expected high improvement in cognitive function, behavioral and psychological symptoms, and activities of daily living, and physical improvements among their AD patients. However, physicians had much lower expectations of improvements in the above parameters. Multi-variate analysis revealed that female gender [odds ratio (OR): 3.747, 95% confidence interval (CI): 1.425-9.851] and familiarity with stem cells (OR: 3.873, 95% CI: 1.290-11.622) were independently associated with caregivers' desire that their AD patients should undergo SCT. The major source of information on SCT was television (76.7%), and the most reliable source of information on SCT was physicians (83.6%).
CONCLUSIONS
In this study, many caregivers of AD patients fantasized and overestimated the need for SCT in comparison with physicians' expectation. Therefore, it is necessary for physicians to develop strategies for educating caregivers about the appropriate risks and benefits of SCT.
Keyword
MeSH Terms
Figure
Reference
-
1. Wimo A, Winblad B, Jönsson L. The worldwide societal costs of dementia: estimates for 2009. Alzheimers Dement. 2010; 6:98–103.
Article2. Mangialasche F, Solomon A, Winblad B, Mecocci P, Kivipelto M. Alzheimer’s disease: clinical trials and drug development. Lancet Neurol. 2010; 9:702–716.
Article3. Hansen RA, Gartlehner G, Webb AP, Morgan LC, Moore CG, Jonas DE. Efficacy and safety of donepezil, galantamine, and rivastigmine for the treatment of Alzheimer’s disease: a systematic review and meta-analysis. Clin Interv Aging. 2008; 3:211–225.4. Roberson ED, Mucke L. 100 years and counting: prospects for defeating Alzheimer’s disease. Science. 2006; 314:781–784.
Article5. Maggini M, Vanacore N, Raschetti R. Cholinesterase inhibitors: drugs looking for a disease? PLoS Med. 2006; 3:e140.
Article6. Lindvall O, Hyun I. Medical innovation versus stem cell tourism. Science. 2009; 324:1664–1665.
Article7. Amariglio N, Hirshberg A, Scheithauer BW, Cohen Y, Loewenthal R, Trakhtenbrot L, et al. Donor-derived brain tumor following neural stem cell transplantation in an ataxia telangiectasia patient. PLoS Med. 2009; 6:e1000029.
Article8. Pontes-Neto OM, Silva GS, Feitosa MR, de Figueiredo NL, Fiorot JA Jr, Rocha TN, et al. Stroke awareness in Brazil: alarming results in a community-based study. Stroke. 2008; 39:292–296.9. Enserink M. Biomedicine. Selling the stem cell dream. Science. 2006; 313:160–163.10. Gaskell G, Einsiedel E, Hallman W, Priest SH, Jackson J, Olsthoorn J. Communication. Social values and the governance of science. Science. 2005; 310:1908–1909.11. Ryan KA, Sanders AN, Wang DD, Levine AD. Tracking the rise of stem cell tourism. Regen Med. 2010; 5:27–33.
Article12. Shineha R, Kawakami M, Kawakami K, Nagata M, Tada T, Kato K. Familiarity and prudence of the Japanese public with research into induced pluripotent stem cells, and their desire for its proper regulation. Stem Cell Rev. 2010; 6:1–7.
Article13. Einsiedel E, Premji S, Geransar R, Orton NC, Thavaratnam T, Bennett LK. Diversity in public views toward stem cell sources and policies. Stem Cell Rev. 2009; 5:102–107.
Article14. Kim YS, Chung DI, Choi H, Baek W, Kim HY, Heo SH, et al. Fantasies about stem cell therapy in chronic ischemic stroke patients. Stem Cells Dev. 2013; 22:31–36.
Article15. Kang Y, Na DL, Hahn S. A validity study on the Korean Mini-Mental State Examination (K-MMSE) in dementia patients. J Korean Neurol Assoc. 1997; 15:300–308.16. Morris JC. The Clinical Dementia Rating (CDR): current version and scoring rules. Neurology. 1993; 43:2412–2414.17. Chen WW, Blurton-Jones M. Concise review: can stem cells be used to treat or model Alzheimer’s disease? Stem Cells. 2012; 30:2612–2618.
Article18. Dantuma E, Merchant S, Sugaya K. Stem cells for the treatment of neurodegenerative diseases. Stem Cell Res Ther. 2010; 1:37.
Article19. Lee JS, Hong JM, Moon GJ, Lee PH, Ahn YH, Bang OY. A long-term follow-up study of intravenous autologous mesenchymal stem cell transplantation in patients with ischemic stroke. Stem Cells. 2010; 28:1099–1106.
Article20. Honmou O, Houkin K, Matsunaga T, Niitsu Y, Ishiai S, Onodera R, et al. Intravenous administration of auto serum-expanded autologous mesenchymal stem cells in stroke. Brain. 2011; 134(Pt 6):1790–1807.
Article21. Zarzeczny A, Caulfield T. Stem cell tourism and doctors’ duties to minors--a view from Canada. Am J Bioeth. 2010; 10:3–15.
Article22. Regenberg AC, Hutchinson LA, Schanker B, Mathews DJ. Medicine on the fringe: stem cell-based interventions in advance of evidence. Stem Cells. 2009; 27:2312–2319.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Developmental Strategy for Stem Cell Therapy Products in Regulatory Considerations
- Cell Biological Characteristics of Adult Stem Cells
- Clinical application of stem cell in cardiovascular diseases
- Current Trends and Prospect of Cell Therapy using Hematopoietic Stem Cells
- Stem Cell Properties of Therapeutic Potential