Cutaneous Melanoma in Asians
- Affiliations
-
- 1Department of Dermatology, Chonnam National University Medical School, Gwangju, Korea. sjyun@chonnam.ac.kr
- KMID: 2432268
- DOI: http://doi.org/10.4068/cmj.2016.52.3.185
Abstract
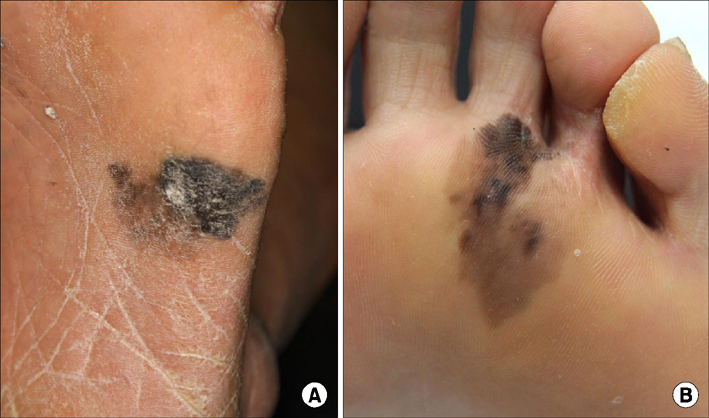
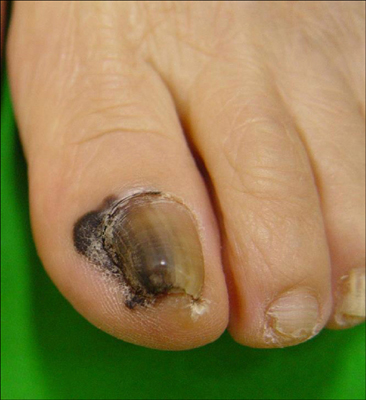
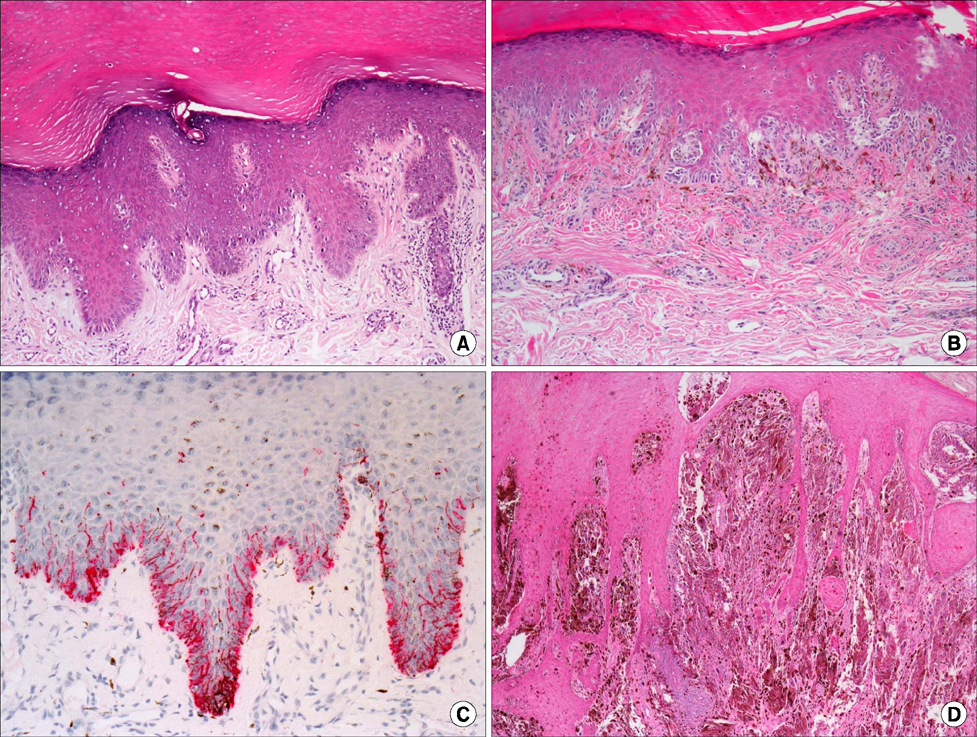
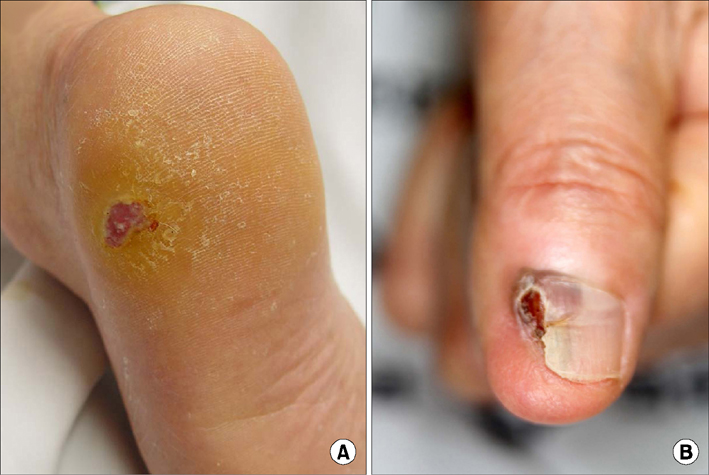
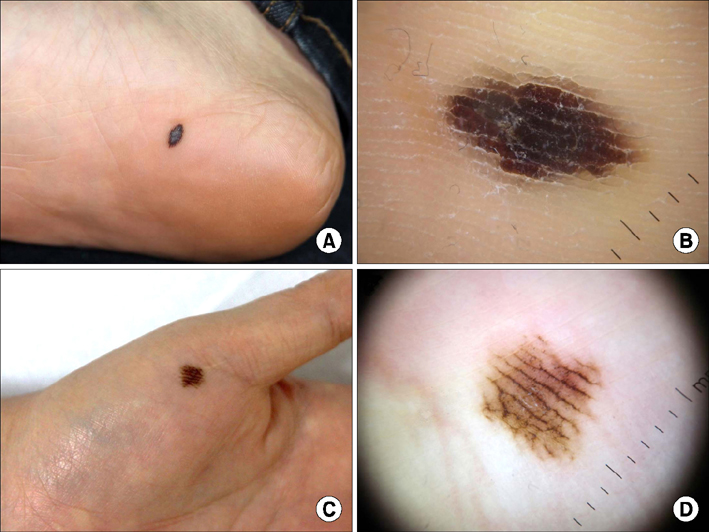
- Malignant melanoma is a rare disease in Asians but potentially the most aggressive form of skin cancer worldwide. It can occur in any melanocyte-containing anatomic site. Four main cutaneous melanoma subtypes are recognized: lentigo maligna melanoma, superficial spreading melanoma, acral lentiginous melanoma (ALM), and nodular melanoma. Generally, excessive exposure to ultraviolet (UV) radiation increases the risk of melanoma. The exception is ALM, which is the most common melanoma subtype in Asians and is not associated with UV radiation. ALM presents as dark brownish to black, irregular maculopatches, nodules, or ulcers on the palms, soles, and nails. The lesions may be misdiagnosed as more benign lesions, such as warts, ulcers, hematomas, foreign bodies, or fungal infections, especially in amelanotic acral melanomas where black pigments are absent. The aim of this brief review is to improve understanding and the rate of early detection thereby reducing mortality, especially regarding cutaneous melanoma in Asians.
MeSH Terms
Figure
Cited by 4 articles
-
Clinicopathologic Features and Prognostic Factors of Primary Cutaneous Melanoma: a Multicenter Study in Korea
Jung Eun Kim, Bo Young Chung, Chang Yoon Sim, A Young Park, Jong Suk Lee, Kyu Uang Whang, Young Lip Park, Hye One Kim, Chun Wook Park, Sung Yul Lee
J Korean Med Sci. 2019;34(16):. doi: 10.3346/jkms.2019.34.e126.Clinical Application of the Unifying Concept of Cutaneous Melanoma
Luca Roncati, Francesco Piscioli, Teresa Pusiol
Chonnam Med J. 2017;53(1):78-80. doi: 10.4068/cmj.2017.53.1.78.Pathogenesis and prevention of skin cancer
Byung-Ho Oh
J Korean Med Assoc. 2018;61(11):644-648. doi: 10.5124/jkma.2018.61.11.644.Sensitivity and Usefulness of VE1 Immunohistochemical Staining in Acral Melanomas with
BRAF Mutation
Min Song Suh, Yoo Duk Choi, Jee-Bum Lee, Seung-Chul Lee, Young Ho Won, Sook Jung Yun
Ann Dermatol. 2018;30(5):556-561. doi: 10.5021/ad.2018.30.5.556.
Reference
-
1. Curado MP, Edwards B, Shin HR, Storm H, Ferlay J, Heanue M, et al. Cancer incidence in five continents. Vol. IX. Lyon: IARC Scientific Pubulications;2007.2. Sneyd M, Cox B. The control of melanoma in New Zealand. N Z Med J. 2006; 119:U2169.3. Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014; 64:9–29.
Article4. Shoo BA, Kashani-Sabet M. Melanoma arising in African-, Asian-, Latino- and Native-American populations. Semin Cutan Med Surg. 2009; 28:96–102.
Article5. Bellew S, Del Rosso JQ, Kim GK. Skin cancer in asians: part 2: melanoma. J Clin Aesthet Dermatol. 2009; 2:34–36.6. Elder DE, Elenitasas R, Murphy GF, Xu X. Benign pigmented lesions and malignant melanoma. In : Elder DE, Elenitasas R, Rosenbach M, Murphy GF, Rubin AI, Xu X, editors. Lever's Histo-Pathology of the Skin. 11th ed. Philadelphia: Wolters Kluwer;2014. p. 853–968.7. Reed RJ. New concepts in surgical pathology of the skin. New York: Wiley;1976. p. 89–90.8. Lee MW, Koh JK, Kwon KS, Kim NI, Kim SW, Kim SN, et al. Clinical and histopathological study of cutaneous melanoma in Korea. Korean J Dermatol. 2003; 41:43–47.9. Jung HJ, Kweon SS, Lee JB, Lee SC, Yun SJ. A clinicopathologic analysis of 177 acral melanomas in Koreans: relevance of spreading pattern and physical stress. JAMA Dermatol. 2013; 149:1281–1288.
Article10. Yun SJ, Kim SJ. Images in clinical medicine. Hutchinson's nail sign. N Engl J Med. 2011; 364:e38.11. Soon SL, Solomon AR Jr, Papadopoulos D, Murray DR, McAlpine B, Washington CV. Acral lentiginous melanoma mimicking benign disease: the Emory experience. J Am Acad Dermatol. 2003; 48:183–188.
Article12. Choi YD, Chun SM, Jin SA, Lee JB, Yun SJ. Amelanotic acral melanomas: clinicopathological, BRAF mutation, and KIT aberration analyses. J Am Acad Dermatol. 2013; 69:700–707.
Article13. Park SL, Le Marchand L, Wilkens LR, Kolonel LN, Henderson BE, Zhang ZF, et al. Risk factors for malignant melanoma in white and non-white/non-African American populations: the multiethnic cohort. Cancer Prev Res (Phila). 2012; 5:423–434.
Article14. Yun SJ, Kwon OS, Han JH, Kweon SS, Lee MW, Lee DY, et al. Clinical characteristics and risk of melanoma development from giant congenital melanocytic naevi in Korea: a nationwide retrospective study. Br J Dermatol. 2012; 166:115–123.
Article15. Elwood JM, Jopson J. Melanoma and sun exposure: an overview of published studies. Int J Cancer. 1997; 73:198–203.
Article16. Tadokoro T, Kobayashi N, Zmudzka BZ, Ito S, Wakamatsu K, Yamaguchi Y, et al. UV-induced DNA damage and melanin content in human skin differing in racial/ethnic origin. FASEB J. 2003; 17:1177–1179.
Article17. Sheehan JM, Cragg N, Chadwick CA, Potten CS, Young AR. Repeated ultraviolet exposure affords the same protection against DNA photodamage and erythema in human skin types II and IV but is associated with faster DNA repair in skin type IV. J Invest Dermatol. 2002; 118:825–829.
Article18. Eide MJ, Weinstock MA. Association of UV index, latitude, and melanoma incidence in nonwhite populations--US Surveillance, Epidemiology, and End Results (SEER) Program, 1992 to 2001. Arch Dermatol. 2005; 141:477–481.
Article19. Veierød MB, Weiderpass E, Thörn M, Hansson J, Lund E, Armstrong B, et al. A prospective study of pigmentation, sun exposure, and risk of cutaneous malignant melanoma in women. J Natl Cancer Inst. 2003; 95:1530–1538.
Article20. Holly EA, Aston DA, Cress RD, Ahn DK, Kristiansen JJ. Cutaneous melanoma in women. II. Phenotypic characteristics and other host-related factors. Am J Epidemiol. 1995; 141:934–942.
Article21. Garbe C, Büttner P, Weiss J, Soyer HP, Stocker U, Krüger S, et al. Associated factors in the prevalence of more than 50 common melanocytic nevi, atypical melanocytic nevi, and actinic lentigines: multicenter case-control study of the Central Malignant Melanoma Registry of the German Dermatological Society. J Invest Dermatol. 1994; 102:700–705.
Article22. Tucker MA, Halpern A, Holly EA, Hartge P, Elder DE, Sagebiel RW, et al. Clinically recognized dysplastic nevi. A central risk factor for cutaneous melanoma. JAMA. 1997; 277:1439–1444.
Article23. Curtin JA, Fridlyand J, Kageshita T, Patel HN, Busam KJ, Kutzner H, et al. Distinct sets of genetic alterations in melanoma. N Engl J Med. 2005; 353:2135–2147.
Article24. Curtin JA, Busam K, Pinkel D, Bastian BC. Somatic activation of KIT in distinct subtypes of melanoma. J Clin Oncol. 2006; 24:4340–4346.
Article25. Kong Y, Si L, Zhu Y, Xu X, Corless CL, Flaherty KT, et al. Large-scale analysis of KIT aberrations in Chinese patients with melanoma. Clin Cancer Res. 2011; 17:1684–1691.
Article26. Ashida A, Takata M, Murata H, Kido K, Saida T. Pathological activation of KIT in metastatic tumors of acral and mucosal melanomas. Int J Cancer. 2009; 124:862–868.
Article27. Jin SA, Chun SM, Choi YD, Kweon SS, Jung ST, Shim HJ, et al. BRAF mutations and KIT aberrations and their clinicopathological correlation in 202 Korean melanomas. J Invest Dermatol. 2013; 133:579–582.
Article28. Nagore E, Oliver V, Moreno-Picot S, Fortea JM. Primary cutaneous melanoma in hidden sites is associated with thicker tumours - a study of 829 patients. Eur J Cancer. 2001; 37:79–82.
Article29. Weinstock MA. Cutaneous melanoma: public health approach to early detection. Dermatol Ther. 2006; 19:26–31.
Article30. Saida T, Koga H, Uhara H. Key points in dermoscopic differentiation between early acral melanoma and acral nevus. J Dermatol. 2011; 38:25–34.
Article31. Kim YC, Lee MG, Choe SW, Lee MC, Chung HG, Cho SH. Acral lentiginous melanoma: an immunohistochemical study of 20 cases. Int J Dermatol. 2003; 42:123–129.
Article32. Balch CM, Gershenwald JE, Soong SJ, Thompson JF, Atkins MB, Byrd DR, et al. Final version of 2009 AJCC melanoma staging and classification. J Clin Oncol. 2009; 27:6199–6206.
Article33. Tsao H, Atkins MB, Sober AJ. Management of cutaneous melanoma. N Engl J Med. 2004; 351:998–1012.
Article34. Khayat D, Rixe O, Martin G, Soubrane C, Banzet M, Bazex JA, et al. Surgical margins in cutaneous melanoma (2 cm versus 5 cm for lesions measuring less than 2.1-mm thick). Cancer. 2003; 97:1941–1946.
Article35. Krown SE, Chapman PB. Defining adequate surgery for primary melanoma. N Engl J Med. 2004; 350:823–825.
Article36. Veronesi U, Cascinelli N. Narrow excision (1-cm margin). A safe procedure for thin cutaneous melanoma. Arch Surg. 1991; 126:438–441.37. Morton DL, Cochran AJ, Thompson JF, Elashoff R, Essner R, Glass EC, et al. Sentinel node biopsy for early-stage melanoma: accuracy and morbidity in MSLT-I, an international multicenter trial. Ann Surg. 2005; 242:302–311. discussion 311-3.38. Morton DL, Thompson JF, Cochran AJ, Mozzillo N, Elashoff R, Essner R, et al. Sentinel-node biopsy or nodal observation in melanoma. N Engl J Med. 2006; 355:1307–1317.
Article39. Sabel MS, Griffith K, Sondak VK, Lowe L, Schwartz JL, Cimmino VM, et al. Predictors of nonsentinel lymph node positivity in patients with a positive sentinel node for melanoma. J Am Coll Surg. 2005; 201:37–47.
Article40. Chang AE, Karnell LH, Menck HR. The National Cancer Data Base report on cutaneous and noncutaneous melanoma: a summary of 84,836 cases from the past decade. The American College of Surgeons Commission on Cancer and the American Cancer Society. Cancer. 1998; 83:1664–1678.
Article41. Chang JW, Yeh KY, Wang CH, Yang TS, Chiang HF, Wei FC, et al. Malignant melanoma in Taiwan: a prognostic study of 181 cases. Melanoma Res. 2004; 14:537–541.
Article42. Kirkwood JM, Ibrahim JG, Sondak VK, Richards J, Flaherty LE, Ernstoff MS, et al. High- and low-dose interferon alfa-2b in high-risk melanoma: first analysis of intergroup trial E1690/S9111/C9190. J Clin Oncol. 2000; 18:2444–2458.
Article43. Acquavella N, Kluger H, Rhee J, Farber L, Tara H, Ariyan S, et al. Toxicity and activity of a twice daily high-dose bolus interleukin 2 regimen in patients with metastatic melanoma and metastatic renal cell cancer. J Immunother. 2008; 31:569–576.
Article44. Kilbridge KL, Weeks JC, Sober AJ, Haluska FG, Slingluff CL, Atkins MB, et al. Patient preferences for adjuvant interferon alfa-2b treatment. J Clin Oncol. 2001; 19:812–823.
Article45. Treisman J, Garlie N. Systemic therapy for cutaneous melanoma. Clin Plast Surg. 2010; 37:127–146.46. Del Prete SA, Maurer LH, O'Donnell J, Forcier RJ, LeMarbre P. Combination chemotherapy with cisplatin, carmustine, dacarbazine, and tamoxifen in metastatic melanoma. Cancer Treat Rep. 1984; 68:1403–1405.47. Legha SS, Ring S, Papadopoulos N, Plager C, Chawla S, Benjamin R. A prospective evaluation of a triple-drug regimen containing cisplatin, vinblastine, and dacarbazine (CVD) for metastatic melanoma. Cancer. 1989; 64:2024–2029.
Article48. Hofmann M, Kiecker F, Wurm R, Schlenger L, Budach V, Sterry W, et al. Temozolomide with or without radiotherapy in melanoma with unresectable brain metastases. J Neurooncol. 2006; 76:59–64.
Article49. Carvajal RD, Antonescu CR, Wolchok JD, Chapman PB, Roman RA, Teitcher J, et al. KIT as a therapeutic target in metastatic melanoma. JAMA. 2011; 305:2327–2334.
Article50. Niezgoda A, Niezgoda P, Czajkowski R. Novel approaches to treatment of advanced melanoma: a review on targeted therapy and immunotherapy. Biomed Res Int. 2015; 2015:851387.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Cutaneous Malignant Melanoma Associated with Papillary Thyroid Cancer
- A Case of Cutaneous Metastatic Melanoma Treated with Topical Diphencyprone (DPCP)
- Cutaneous Ulceration after Injection of Interferon Alpha in a Melanoma Patient
- Three Cases of Malignant Melanoma Possibly Arising in a Long Standing Melanocytic Nevus
- Primary cutaneous malignant melanoma of the breast