Intest Res.
2018 Apr;16(2):255-266. 10.5217/ir.2018.16.2.255.
Comparison of efficacy of once daily multimatrix mesalazine 2.4 g/day and 4.8 g/day with other 5-aminosalicylic acid preparation in active ulcerative colitis: a randomized, double-blind study
- Affiliations
-
- 1Center for Diagnostic and Therapeutic Endoscopy, Keio University Hospital, Tokyo, Japan. hogata@z8.keio.jp
- 2Yokoyama Hospital for Gastroenterological Diseases, Nagoya, Japan.
- 3Clinical Development Department, Mochida Pharmaceutical Co., Ltd., Tokyo, Japan.
- 4Center for Advanced IBD Research and Treatment, Kitasato Institute Hospital, Kitasato University, Tokyo, Japan.
- KMID: 2417677
- DOI: http://doi.org/10.5217/ir.2018.16.2.255
Abstract
- BACKGROUND/AIMS
This study compared the efficacy of multimatrix mesalazine 2.4 g/day and 4.8 g/day with controlled-release mesalazine 2.25 g/day.
METHODS
In this multicenter, randomized, double-blind study, 251 patients with mildly to moderately active ulcerative colitis received multimatrix mesalazine 2.4 g/day once daily (Multimatrix-2.4), 4.8 g/day once daily (Multimatrix-4.8), or controlled-release (time-dependent) mesalazine 2.25 g/day 3 times daily (Time-2.25) for 8 weeks. The primary efficacy endpoint was the change in the ulcerative colitis-disease activity index (UC-DAI) score.
RESULTS
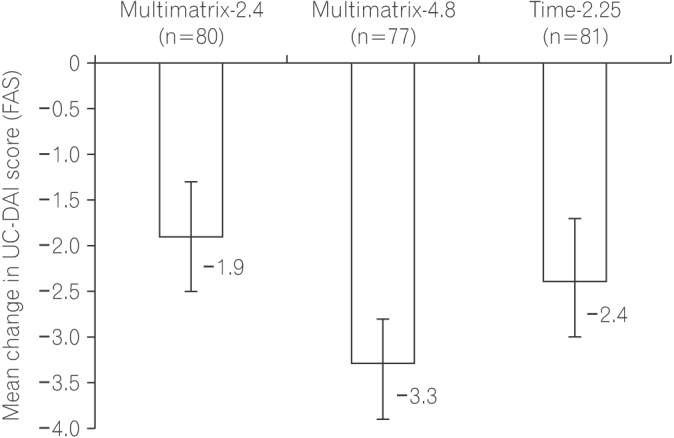
The mean change in the UC-DAI score and standard deviation in the per protocol set was −1.9±2.5 for Multimatrix-2.4 and −2.4±2.8 for Time-2.25. The difference between Multimatrix-2.4 and Time-2.25 was 0.3 (two-sided 95% confidence interval [CI], −0.5 to 1.1), thus non-inferiority was not demonstrated based on the pre-defined non-inferiority margin (1.0). In the full analysis set, the difference between Multimatrix-4.8 and Time-2.25 was −1.2 (two-sided 95% CI, −2.0 to −0.5), and the mean change in UC-DAI score in the FAS was −3.3 (two-sided 95% CI, −3.9 to −2.8) for Multimatrix-4.8 and −1.9 (two-sided 95% CI, −2.5 to −1.3) for Multimatrix-2.4, indicating that Multimatrix-4.8 was more effective than Time-2.25 and Multimatrix-2.4. There was no difference among the treatment groups in terms of safety.
CONCLUSIONS
This study showed that the efficacy of multimatrix mesalazine 2.4 g/day was comparable to controlled release mesalazine 2.25 g/day, although non-inferiority was not demonstrated. Importantly, this was the first study to indicate that multimatrix mesalazine 4.8 g/day was more effective than 2.4g/day with no associated safety concerns.
Keyword
Figure
Cited by 1 articles
-
Is once daily multimatrix mesalazine therapy effective regardless of the dose in patients with mild to moderate ulcerative colitis?
Seong Ran Jeon
Intest Res. 2018;16(2):163-165. doi: 10.5217/ir.2018.16.2.163.
Reference
-
1. Lichtenstein GR, Kamm MA, Boddu P, et al. Effect of once- or twice-daily MMX mesalamine (SPD476) for the induction of remission of mild to moderately active ulcerative colitis. Clin Gastroenterol Hepatol. 2007; 5:95–102. PMID: 17234558.
Article2. Kamm MA, Sandborn WJ, Gassull M, et al. Once-daily, high-concentration MMX mesalamine in active ulcerative colitis. Gastroenterology. 2007; 132:66–75. PMID: 17241860.
Article3. Frieri G, Giacomelli R, Pimpo M, et al. Mucosal 5-aminosalicylic acid concentration inversely correlates with severity of colonic inflammation in patients with ulcerative colitis. Gut. 2000; 47:410–414. PMID: 10940280.
Article4. Sutherland LR, Martin F, Greer S, et al. 5-Aminosalicylic acid enema in the treatment of distal ulcerative colitis, proctosigmoiditis, and proctitis. Gastroenterology. 1987; 92:1894–1898. PMID: 3569765.
Article5. Hibi T. Ministry of Health, Labour and Welfare. Atlas of endoscopy in inflammatory bowel disease. Research Group for Intractable Inflammatory Bowel Disease (Watanabe Group). Health and labour science research grants from the Japanese Ministry of Health, Labour and Welfare and research on measures for intractable disease: 2007 research report. Tokyo: Ministry of Health, Labour and Welfare;2008. p. 423–457.6. Ito H, Iida M, Matsumoto T, et al. Direct comparison of two different mesalamine formulations for the induction of remission in patients with ulcerative colitis: a double-blind, randomized study. Inflamm Bowel Dis. 2010; 16:1567–1574. PMID: 20049950.
Article7. Miner P, Hanauer S, Robinson M, Schwartz J, Arora S. Pentasa UC Maintenance Study Group. Safety and efficacy of controlled-release mesalamine for maintenance of remission in ulcerative colitis. Dig Dis Sci. 1995; 40:296–304. PMID: 7851193.
Article8. Lichtenstein GR, Kamm MA, Sandborn WJ, Lyne A, Joseph RE. MMX mesalazine for the induction of remission of mild-to-moderately active ulcerative colitis: efficacy and tolerability in specific patient subpopulations. Aliment Pharmacol Ther. 2008; 27:1094–1102. PMID: 18363894.
Article9. Hanauer SB, Sandborn WJ, Dallaire C, et al. Delayed-release oral mesalamine 4.8 g/day (800 mg tablets) compared to 2.4 g/day (400 mg tablets) for the treatment of mildly to moderately active ulcerative colitis: the ASCEND I trial. Can J Gastroenterol. 2007; 21:827–834. PMID: 18080055.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Comparison of efficacy of multimatrix mesalazine 4.8 g/day once-daily with other high-dose mesalazine in active ulcerative colitis: a randomized, double-blind study
- Comparison of efficacies of once-daily dose multimatrix mesalazine and multiple-dose mesalazine for the maintenance of remission in ulcerative colitis: a randomized, double-blind study
- Is once daily multimatrix mesalazine therapy effective regardless of the dose in patients with mild to moderate ulcerative colitis?
- Acute Pancreatitis Induced by 5-Aminosalicylic Acid in a Patient with Ulcerative Colitis
- A Case of Acute Pancreatitis Caused by 5-aminosalicylic Acid Suppositories in a Patient with Ulcerative Colitis