Nat Prod Sci.
2017 Jun;23(2):92-96. 10.20307/nps.2017.23.2.92.
Inhibitory Effect of a Sesquiterpene from Artemisia iwayomogi on Expression of Inducible Nitric Oxide Synthase by Suppression of I-κBα Degradation in LPS-stimulated RAW 264.7 Cells
- Affiliations
-
- 1College of Pharmacy, Sookmyung Women's University, Seoul 04310, Republic of Korea. ryuha@sookmyung.ac.kr
- 2Department of Natural Medicine Resources, Semyung University, Jecheon, Chungbuk 27136, Republic of Korea. hwalee@semyung.ac.kr
- KMID: 2387057
- DOI: http://doi.org/10.20307/nps.2017.23.2.92
Abstract
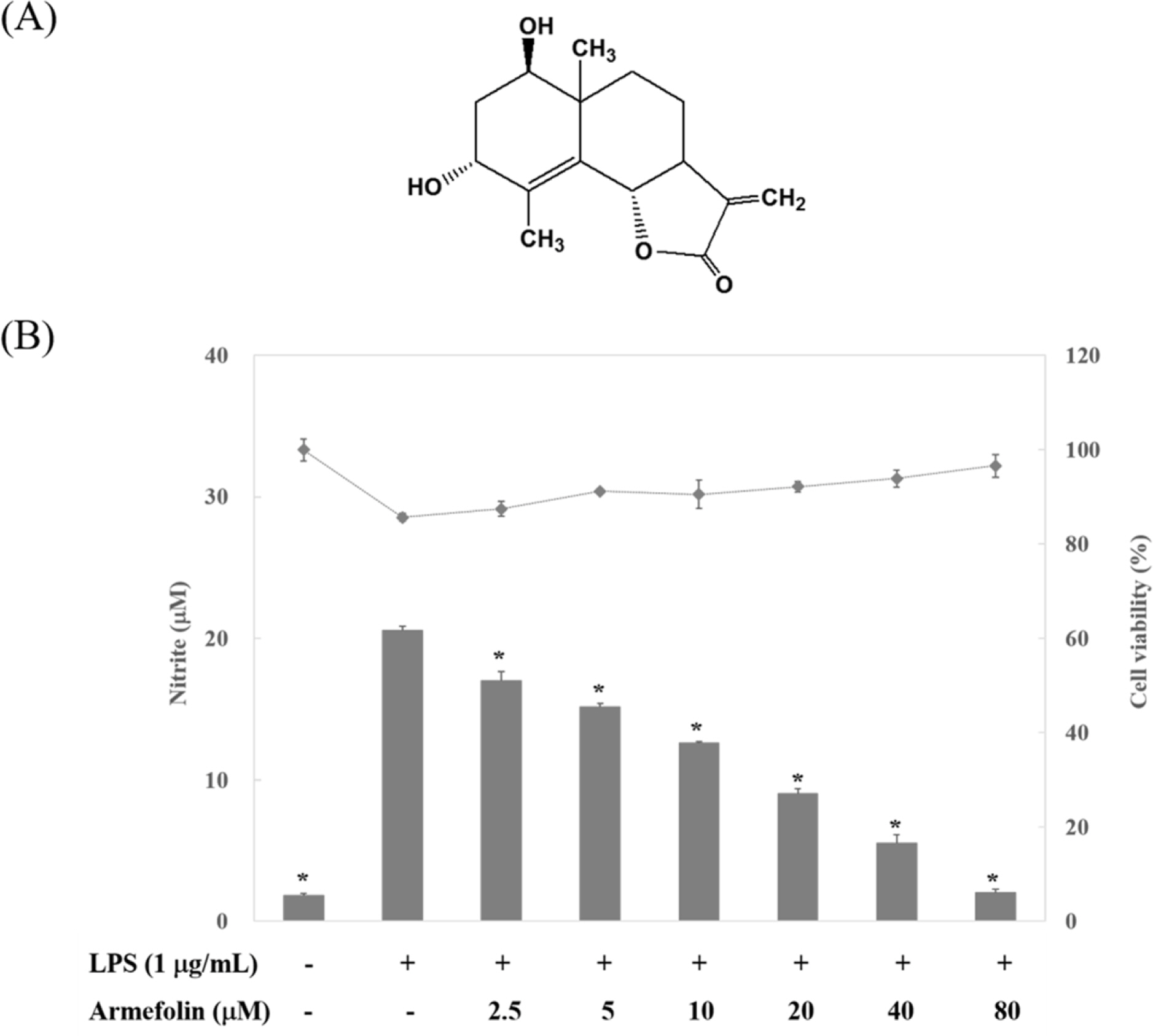
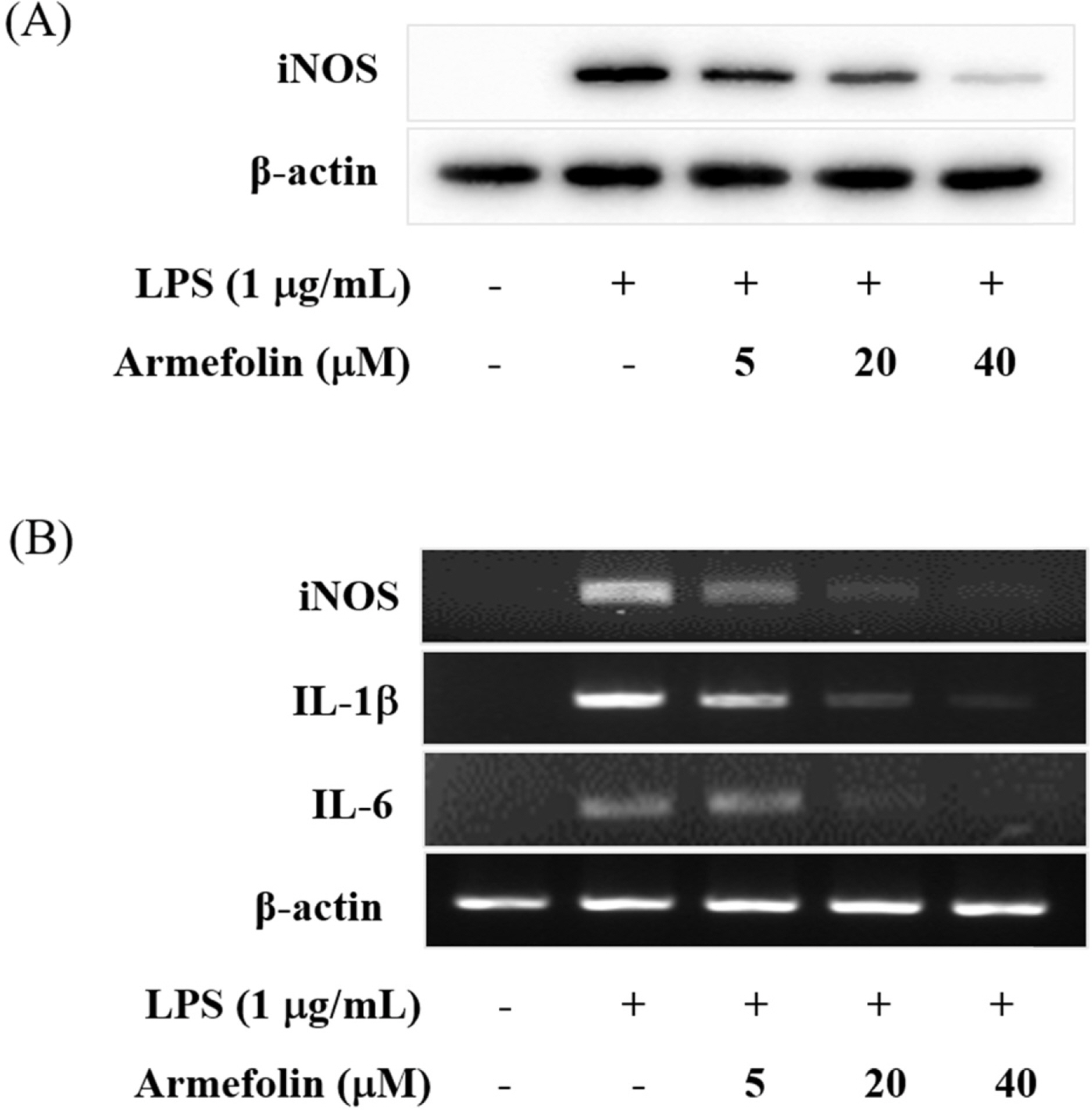
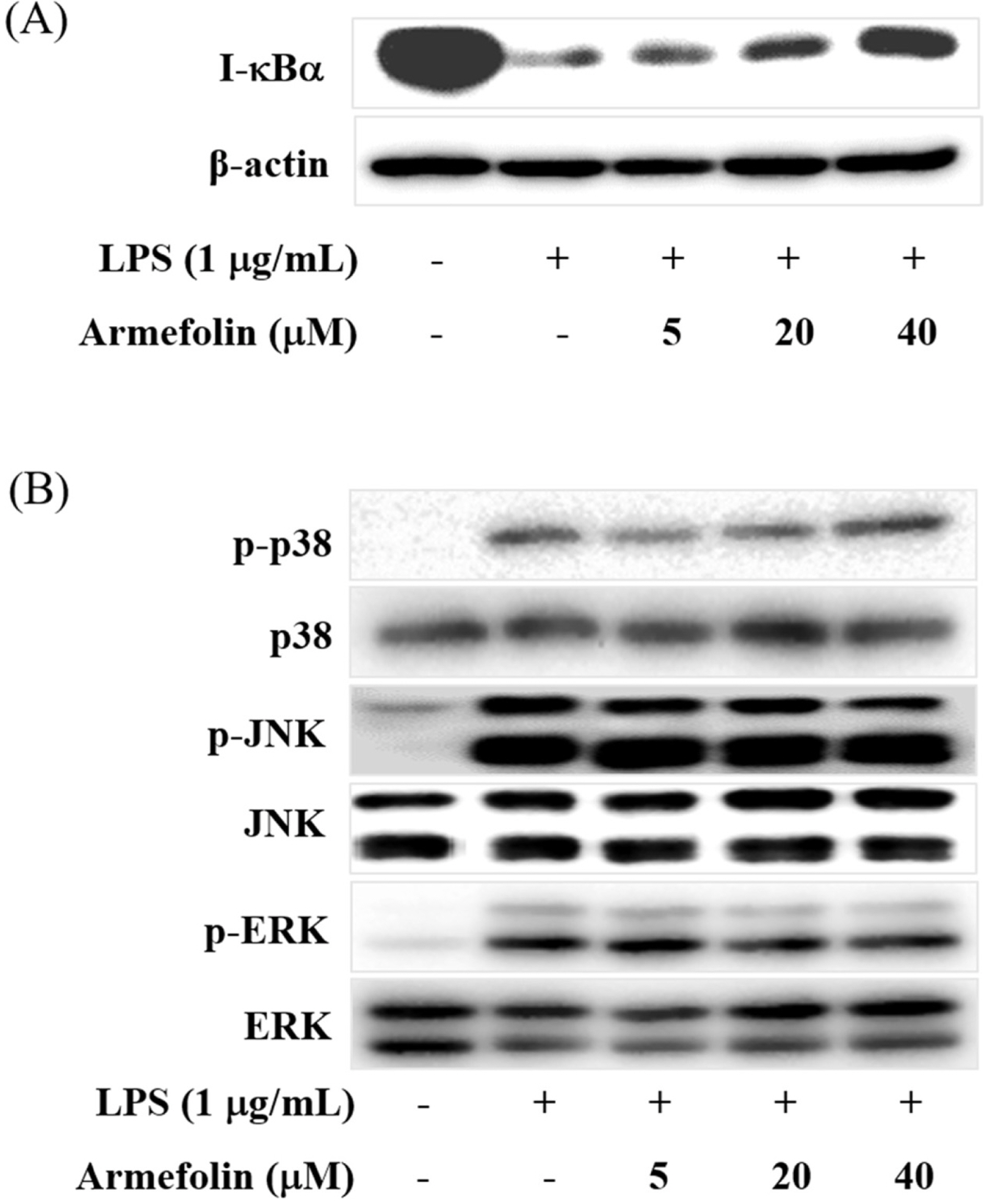
- A sesquiterpene was purified from Artemisia iwayomogi methanolic extract during the course of searching anti-inflammatory principle from medicinal plants. A sesquiterpene identified as armefolin inhibited the production of nitric oxide (NO) and attenuated inducible nitric oxide synthase (iNOS) protein level in lipopolysaccharide (LPS)-activated RAW 264.7 cells. Armefolin also down-regulated mRNA expressions of iNOS and pro-inflammatory cytokines, interleukin-1β and interleukin-6 in LPS-activated macrophages. Moreover, armefolin suppressed the degradation of inhibitory-κBα (I-κBα) in LPS-activated macrophages. These data suggest that armefolin from A. iwayomogi can suppress the LPS-induced production of NO and the expression of iNOS gene through inhibiting the degradation of I-κBα. Taken together, armefolin from A. iwayomogi might be a candidate as promising anti-inflammatory agent.
Keyword
MeSH Terms
Figure
Reference
-
References
(1). Kim J. K.Illustrated Natural Drugs Encyclopedia; Namsandang publisher: Seoul,. 1989. , p. 79.(2). Yan X. T.., Ding Y.., Lee S. H.., Li W.., Sun Y. N.., Yang S. Y.., Jang H. D.., Kim Y. H.Nat. Prod. Sci. 2014. 20:176–181.(3). Ha H.., Lee H.., Seo C. S.., Lim H. S.., Lee M. Y.., Lee J. K.., Shin H.Evid. Based Complement. Alternat. Med. 2014. 2014:673286.(4). Wang J. H.., Choi M. K.., Shin J. W.., Hwang S. Y.., Son C. G. J.Ethnopharmacol. 2012. 140:179–185.(5). Yu H. H.., Kim Y. H.., Kil B. S.., Kim K. J.., Jeong S. I.., You Y. O.Planta Med. 2003. 69:1159–1162.(6). Kim A. R.., Zou Y. N.., Park T. H.., Shim K. H.., Kim M. S.., Kim N. D.., Kim J. D.., Bae S. J.., Choi J. S.., Chung H. Y.Phytother. Res. 2004. 18:1–7.(7). Ahn H.., Kim J. Y.., Lee H. J.., Kim Y. K.., Ryu J. H.Arch. Pharm. Res. 2003. 26:301–305.(8). Nathan C.., Xie Q. W. J.Biol. Chem. 1994. 269:13725–13728.(9). Thomsen L. L.., Miles D. W.Cancer Metastasis Rev. 1998. 17:107–118.
Article(10). Weinberg J. B.., Fermor B.., Guilak F.Subcell. Biochem. 2007. 42:31–62.(11). Mittal R.., Gonzalez-Gomez I.., Goth K. A.., Prasadarao N. V.Am. J. Pathol. 2010. 176:1292–1305.(12). Kapur S.., Picard F.., Perreault M.., Deshaies Y.., Marette A.Int. J. Obes. Relat. Metab. Disord. 2000. 24:S36–S40.(13). Förstermann U.., Schmidt H. H.., Pollock J. S.., Sheng H.., Mitchell J. A.., Warner T. D.., Nakane M.., Murad F.Biochem. Pharmacol. 1991. 42:1849–1857.(14). Förstermann U.., Pollock J. S.., Tracey W. R.., Nakane M.Methods Enzymol. 1994. 233:258–264.(15). Hiraku Y.., Kawanishi S.., Ichinose T.., Murata M. Ann. N. Y.Acad. Sci. 2010. 1203:15–22.(16). Hiraku Y.., Kawanishi S.Methods Mol. Biol. 2009. 512:3–13.(17). Mata R.., Delgado G.., de Vivar A. R.Phytochemistry. 1984. 23:1665–1668.(18). Gasparini C.., Feldmann M.Curr. Pharm. Des. 2012. 18:5735–5745.(19). Kanarek N.., Ben-Neriah Y.Immunol. Rev. 2012. 246:77–94.(20). Valera F. C.., Umezawa K.., Brassesco M. S.., Castro-Gamero A. M.., Queiroz R. G. D. P.., Scrideli C. A.., Tone L. G.., Anselmo-Lima W. T.Cell. Physiol. Biochem. 2012. 30:13–22.(21). Ganai A. A.., Khan A. A.., Malik Z. A.., Farooqi H.Toxicol. Appl. Pharmacol. 2015. 283:139–146.(22). Kaminska B.Biochim. Biophys. Acta. 2005. 1754:253–262.(23). Kim E. K.., Choi E. J.Arch. Toxicol. 2015. 89:867–882.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Anti-inflammatory activities of Scolopendra subspinipes mutilans in RAW 264.7 cells
- Anti-inflammatory Activity of Standardized Fraction from Inula helenium L. via Suppression of NF-κB Pathway in RAW 264.7 Cells
- Differential regulation of inducible nitric oxide synthase and cyclooxygenase-2 expression by superoxide dismutase in lipopolysaccharide stimulated RAW 264.7 cells
- Anti-inflammatory effect of the water fraction from hawthorn fruit on LPS-stimulated RAW 264.7 cells
- Inhibitory Effects of Paeonia Suffruticosa on the Expression of Inducible Nitric Oxide Synthase Gene in RAW264.7 Macrophages