Pediatr Gastroenterol Hepatol Nutr.
2016 Dec;19(4):251-258. 10.5223/pghn.2016.19.4.251.
Acanthosis Nigricans as a Clinical Predictor of Insulin Resistance in Obese Children
- Affiliations
-
- 1Department of Pediatrics, Chosun University School of Medicine, Gwangju, Korea. krmoon@chosun.ac.kr
- KMID: 2364766
- DOI: http://doi.org/10.5223/pghn.2016.19.4.251
Abstract
- PURPOSE
This study aimed to evaluate the utility of acanthosis nigricans (AN) severity as an index for predicting insulin resistance in obese children.
METHODS
The subjects comprised 74 obese pediatric patients who attended the Department of Pediatrics at Chosun University Hospital between January 2013 and March 2016. Waist circumference; body mass index; blood pressure; fasting glucose and fasting insulin levels; lipid profile; aspartate transaminase, alanine transaminase, glycated hemoglobin, C-peptide, and uric acid levels; and homeostatic model assessment insulin resistance (HOMA-IR) and quantitative insulin check sensitivity index (QUICKI) scores were compared between subjects with AN and those without AN. Receiver operating characteristic curves were used to investigate the utility of the AN score in predicting insulin resistance. HOMA-IR and QUICKI were compared according to AN severity.
RESULTS
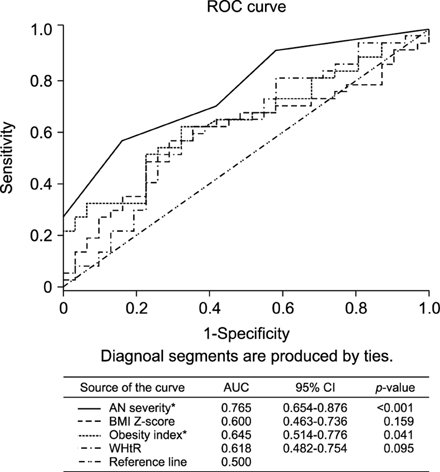
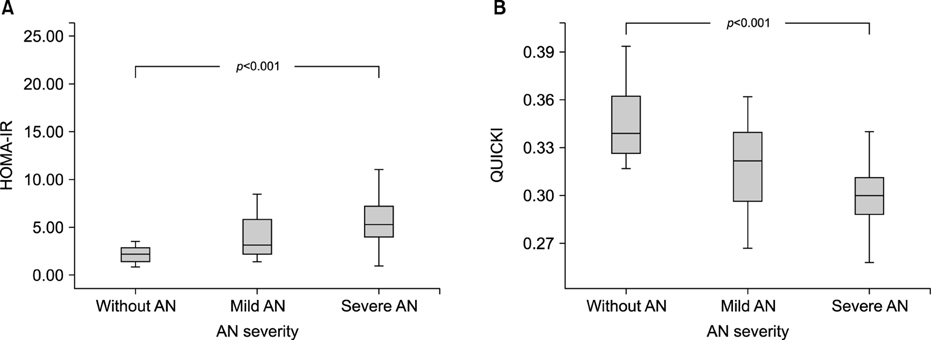
The With AN group had higher fasting insulin levels (24.1±21.0 mU/L vs. 9.8±3.6 mU/L, p<0.001) and HOMA-IR score (5.74±4.71 vs. 2.14±0.86, p<0.001) than the Without AN group. The AN score used to predict insulin resistance was 3 points or more (sensitivity 56.8%, specificity 83.9%). HOMA-IR scores increased with AN severity, from the Without AN group (mean, 2.15; 95% confidence interval [CI], 1.72-2.57) to the Mild AN (mean, 4.15; 95% CI, 3.04-5.25) and Severe AN groups (mean, 7.22; 95% CI, 5.08-9.35; p<0.001).
CONCLUSION
Insulin resistance worsens with increasing AN severity, and patients with Severe AN (AN score ≥3) are at increased risk of insulin resistance.
MeSH Terms
-
Acanthosis Nigricans*
Alanine Transaminase
Aspartate Aminotransferases
Blood Pressure
Body Mass Index
C-Peptide
Child*
Fasting
Glucose
Hemoglobin A, Glycosylated
Humans
Hyperinsulinism
Insulin Resistance*
Insulin*
Pediatric Obesity
Pediatrics
ROC Curve
Sensitivity and Specificity
Uric Acid
Waist Circumference
Alanine Transaminase
Aspartate Aminotransferases
C-Peptide
Glucose
Insulin
Uric Acid
Figure
Reference
-
1. Seo JW. Obesity in children and adolescents. Korean J Pediatr. 2009; 52:1311–1320.
Article2. Weiss R, Kaufman FR. Metabolic complications of childhood obesity: identifying and mitigating the risk. Diabetes Care. 2008; 31:Suppl 2. S310–S316.3. Hong YM. Metabolic syndrome in children and adolescents. Korean J Pediatr. 2009; 52:737–744.
Article4. Chueh HW, Cho GR, Yoo JH. Clinical significance of acanthosis nigricans in children and adolescents with obesity induced metabolic complications. Korean J Pediatr. 2007; 50:987–994.
Article5. Kahn CR, Flier JS, Bar RS, Archer JA, Gorden P, Martin MM, et al. The syndromes of insulin resistance and acanthosis nigricans. Insulin-receptor disorders in man. N Engl J Med. 1976; 294:739–745.
Article6. Dietz WH. Overweight in childhood and adolescence. N Engl J Med. 2004; 350:855–857.
Article7. Slyper AH, Kashmer L, Huang WM, Re'em Y. Acanthosis nigricans, vitamin D, and insulin resistance in obese children and adolescents. J Pediatr Endocrinol Metab. 2014; 27:1107–1111.
Article8. Patidar PP, Ramachandra P, Philip R, Saran S, Agarwal P, Gutch M, et al. Correlation of acanthosis nigricans with insulin resistance, anthropometric, and other metabolic parameters in diabetic Indians. Indian J Endocrinol Metab. 2012; 16:S436–S437.
Article9. Centers for Disease Control and Prevention. 2007 Korean National Growth Charts [Internet]. Osong: 2008. [cited 2008 Aug 21]. Available from: http://cdc.go.kr/CDC/notice/CdcKrInfo0201.jsp?menuIds=HOME001-MNU1154-MNU0005-MNU1889&cid=1235.10. Keskin M, Kurtoglu S, Kendirci M, Atabek ME, Yazici C. Homeostasis model assessment is more reliable than the fasting glucose/insulin ratio and quantitative insulin sensitivity check index for assessing insulin resistance among obese children and adolescents. Pediatrics. 2005; 115:e500–e503.
Article11. Burke JP, Hale DE, Hazuda HP, Stern MP. A quantitative scale of acanthosis nigricans. Diabetes Care. 1999; 22:1655–1659.
Article12. American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2014; 37:Suppl 1. S81–S90.13. National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III) final report. Circulation. 2002; 106:3143–3421.14. Yosipovitch G, DeVore A, Dawn A. Obesity and the skin: skin physiology and skin manifestations of obesity. J Am Acad Dermatol. 2007; 56:901–916.
Article15. Hermanns-Lê T, Scheen A, Piérard GE. Acanthosis nigricans associated with insulin resistance: pathophysiology and management. Am J Clin Dermatol. 2004; 5:199–203.16. Dunaif A, Green G, Phelps RG, Lebwohl M, Futterweit W, Lewy L. Acanthosis nigricans, insulin action, and hyperandrogenism: clinical, histological, and biochemical findings. J Clin Endocrinol Metab. 1991; 73:590–595.
Article17. Schwartz RA. Acanthosis nigricans. J Am Acad Dermatol. 1994; 31:1–19.
Article18. Reaven GM. Role of insulin resistance in human disease (syndrome X): an expanded definition. Annu Rev Med. 1993; 44:121–131.
Article19. Caprio S, Plewe G, Diamond MP, Simonson DC, Boulware SD, Sherwin RS, et al. Increased insulin secretion in puberty: a compensatory response to reductions in insulin sensitivity. J Pediatr. 1989; 114:963–967.
Article20. Stuart CA, Smith MM, Gilkison CR, Shaheb S, Stahn RM. Acanthosis nigricans among native Americans: an indicator of high diabetes risk. Am J Public Health. 1994; 84:1839–1842.
Article21. Stuart CA, Peters EJ, Prince MJ, Richards G, Cavallo A, Meyer WJ 3rd. Insulin resistance with acanthosis nigricans: the roles of obesity and androgen excess. Metabolism. 1986; 35:197–205.
Article22. Venkatswami S, Anandam S. Acanthosis nigricans: a flag for insulin resistance. J Endocrinol Metab Diabetes South Afr. 2014; 19:68–74.
Article23. Noviarti D, Rini EA, Oenzil F. Association of resistin level with acanthosis nigricans in obese adolescents. Paediatr Indones. 2016; 56:32–36.
Article24. Kong AS, Williams RL, Rhyne R, Urias-Sandoval V, Cardinali G, Weller NF, et al. Acanthosis nigricans: high prevalence and association with diabetes in a practice-based research network consortium--a PRImary care Multi-Ethnic network (PRIME Net) study. J Am Board Fam Med. 2010; 23:476–485.
Article25. Yamazaki H, Ito S, Yoshida H. Acanthosis nigricans is a reliable cutaneous marker of insulin resistance in obese Japanese children. Pediatr Int. 2003; 45:701–705.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Acanthosis Nigricans Detected in Obese Infertile Woman
- Coexistence of Scleredema and Acanthosis Nigricans in a Patient with Diabetes Mellitus
- A case of generalized lipodystrophy
- Syndromal acanthosis nigricans with insulin resistance
- Three Cases of Concomitant Acanthosis Nigricans with Confluent and Reticulated Papillomatosis in Obese Patients