J Gynecol Oncol.
2013 Apr;24(2):108-113. 10.3802/jgo.2013.24.2.108.
Concurrent chemoradiotherapy with low-dose daily cisplatin for high risk uterine cervical cancer: a long-term follow-up study
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Kurume University School of Medicine, Kurume, Japan. kimi@med.kurume-u.ac.jp
- 2Department of Radiology, Kurume University School of Medicine, Kurume, Japan.
- KMID: 2288540
- DOI: http://doi.org/10.3802/jgo.2013.24.2.108
Abstract
OBJECTIVE
To evaluate the clinical efficacy of concurrent chemoradiotherapy (CCRT) using daily low-dose cisplatin for cervical cancer.
METHODS
Fifty-one patients with locally advanced cervical cancer (FIGO stage IB2, bulky IIA, IIB-IVA) who were treated with CCRT as primary therapy at Kurume University Hospital between 2000 and 2007 were retrospectively reviewed. CCRT consisted of 5 mg/m2/day of cisplatin 5 days per week, and external beam radiotherapy (EBRT) administrated to whole pelvis to 45-50.6 Gy. High-dose-rate intracavitary brachytherapy was delivered in a single dose of 4-5 Gy at point A, once a week after 20-30 Gy of EBRT.
RESULTS
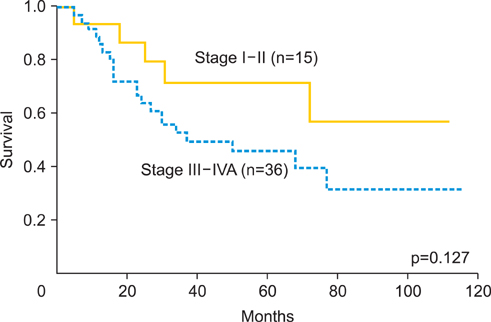
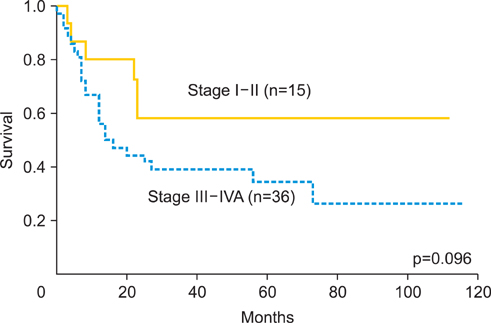
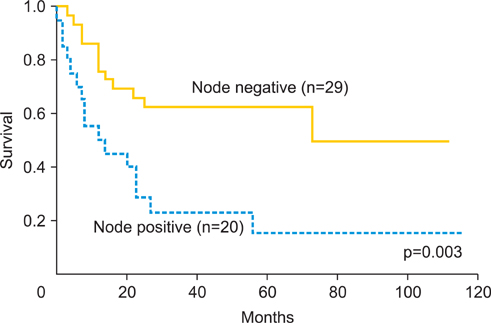
The median follow-up duration was 42 months (range, 5 to 116 months). The overall response rate was 94.1%. Five year overall survival rate was 71.5% and 46.2% in stage I or II, and stage III or IVA, respectively. During follow-up period, 30 recurrences (58.8%) were found, the local failure rate was 39%, and distant failure rate was 35.2%, and both (local and distant) were 15.7%. Hematological toxicities were the most frequent acute toxicities. Grade 3 and 4 neutropenia was observed in 37.3%. Late intestinal toxicities appeared in 7 cases (13.7%), which occurred between 6 and 114 months after treatment. Four cases required bowel surgery.
CONCLUSION
CCRT using daily low-dose cisplatin was tolerable and showed favorable initial response as the primary therapy for locally advanced uterine cervical cancer. But there was no remarkable long-term benefit for patients' survival or local disease control in this study. The incidence of late intestinal toxicity still requires further investigation.
MeSH Terms
Figure
Reference
-
1. Keys HM, Bundy BN, Stehman FB, Muderspach LI, Chafe WE, Suggs CL 3rd, et al. Cisplatin, radiation, and adjuvant hysterectomy compared with radiation and adjuvant hysterectomy for bulky stage IB cervical carcinoma. N Engl J Med. 1999. 340:1154–1161.2. Peters WA 3rd, Liu PY, Barrett RJ 2nd, Stock RJ, Monk BJ, Berek JS, et al. Concurrent chemotherapy and pelvic radiation therapy compared with pelvic radiation therapy alone as adjuvant therapy after radical surgery in high-risk early-stage cancer of the cervix. J Clin Oncol. 2000. 18:1606–1613.3. Morris M, Eifel PJ, Lu J, Grigsby PW, Levenback C, Stevens RE, et al. Pelvic radiation with concurrent chemotherapy compared with pelvic and para-aortic radiation for high-risk cervical cancer. N Engl J Med. 1999. 340:1137–1143.4. Whitney CW, Sause W, Bundy BN, Malfetano JH, Hannigan EV, Fowler WC Jr, et al. Randomized comparison of fluorouracil plus cisplatin versus hydroxyurea as an adjunct to radiation therapy in stage IIB-IVA carcinoma of the cervix with negative para-aortic lymph nodes: a Gynecologic Oncology Group and Southwest Oncology Group study. J Clin Oncol. 1999. 17:1339–1348.5. Rose PG, Bundy BN, Watkins EB, Thigpen JT, Deppe G, Maiman MA, et al. Concurrent cisplatin-based radiotherapy and chemotherapy for locally advanced cervical cancer. N Engl J Med. 1999. 340:1144–1153.6. Cox JD, Stetz J, Pajak TF. Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC). Int J Radiat Oncol Biol Phys. 1995. 31:1341–1346.7. Green JA, Kirwan JM, Tierney JF, Symonds P, Fresco L, Collingwood M, et al. Survival and recurrence after concomitant chemotherapy and radiotherapy for cancer of the uterine cervix: a systematic review and meta-analysis. Lancet. 2001. 358:781–786.8. Chemoradiotherapy for Cervical Cancer Meta-Analysis Collaboration. Reducing uncertainties about the effects of chemoradiotherapy for cervical cancer: a systematic review and meta-analysis of individual patient data from 18 randomized trials. J Clin Oncol. 2008. 26:5802–5812.9. Rose PG. Chemoradiotherapy for cervical cancer. Eur J Cancer. 2002. 38:270–278.10. Rose PG, Ali S, Watkins E, Thigpen JT, Deppe G, Clarke-Pearson DL, et al. Long-term follow-up of a randomized trial comparing concurrent single agent cisplatin, cisplatin-based combination chemotherapy, or hydroxyurea during pelvic irradiation for locally advanced cervical cancer: a Gynecologic Oncology Group Study. J Clin Oncol. 2007. 25:2804–2810.11. Ohno T, Kato S, Tsuji H. Phase I study of weekly cisplatin plus radiotherapy for locally advanced cervical cancer. J Jpn Soc Gynecol Oncol. 2005. 23:564–571.12. Ikushima H, Osaki K, Furutani S, Yamashita K, Kawanaka T, Kishida Y, et al. Chemoradiation therapy for cervical cancer: toxicity of concurrent weekly cisplatin. Radiat Med. 2006. 24:115–121.13. Mitsuhashi A, Uno T, Tanaka N, Suzuka K, Tate S, Yamazawa K, et al. Phase I study of daily cisplatin and concurrent radiotherapy in patients with cervical carcinoma. Gynecol Oncol. 2005. 96:194–197.14. Uno T, Mitsuhashi A, Isobe K, Yamamoto S, Kawakami H, Ueno N, et al. Concurrent daily cisplatin and extended-field radiation therapy for carcinoma of the cervix. Int J Gynecol Cancer. 2008. 18:80–84.15. Kirwan JM, Symonds P, Green JA, Tierney J, Collingwood M, Williams CJ. A systematic review of acute and late toxicity of concomitant chemoradiation for cervical cancer. Radiother Oncol. 2003. 68:217–226.16. Eifel PJ, Winter K, Morris M, Levenback C, Grigsby PW, Cooper J, et al. Pelvic irradiation with concurrent chemotherapy versus pelvic and para-aortic irradiation for high-risk cervical cancer: an update of radiation therapy oncology group trial (RTOG) 90-01. J Clin Oncol. 2004. 22:872–880.17. Choi IJ, Cha MS, Park ES, Han MS, Choi Y, Je GH, et al. The efficacy of concurrent cisplatin and 5-flurouracil chemotherapy and radiation therapy for locally advanced cancer of the uterine cervix. J Gynecol Oncol. 2008. 19:129–134.18. Duenas-Gonzalez A, Zarba JJ, Patel F, Alcedo JC, Beslija S, Casanova L, et al. Phase III, open-label, randomized study comparing concurrent gemcitabine plus cisplatin and radiation followed by adjuvant gemcitabine and cisplatin versus concurrent cisplatin and radiation in patients with stage IIB to IVA carcinoma of the cervix. J Clin Oncol. 2011. 29:1678–1685.19. Wang S, Zhang DS, Pan T, Liu S, Wang MK. Efficacy of concurrent chemoradiotherapy plus adjuvant chemotherapy on advanced cervical cancer. Chin J Cancer. 2010. 29:959–963.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Low Dose Cisplatin as a Radiation Sensitizer in Management of Locally Advanced Squamous Cell Carcinoma of the Uterine Cervix: Evaluation of Acute Toxicity and Early Response
- The Role of Postoperative Adjuvant Chemoradiotherapy for Patients with Invasive Cervical Cancer Discovered after sImple Total Hysterectomy
- Concurrent Cisplatin and 5-Flurouracil Chemotherapy and Radiation Therapy for Locally Advanced Cancer of The Uterine Cervix
- Prognostic significance of neutropenia during adjuvant concurrent chemoradiotherapy in early cervical cancer
- Superior vena cava syndrome secondary to hickman catheter in the advanced cervical cancer patient treated with concurrent chemoradiotherapy: A case report