J Breast Cancer.
2011 Dec;14(4):283-288. 10.4048/jbc.2011.14.4.283.
Chemotherapy Response Assay Test and Prognosis for Breast Cancer Patients Who Have Undergone Anthracycline- and Taxane-Based Chemotherapy
- Affiliations
-
- 1Department of Medicine, Kyung Hee University Graduate School, Seoul, Korea.
- 2Department of Surgery, Ewha Womans University School of Medicine, Seoul, Korea.
- 3Department of Surgery, Kyung Hee University School of Medicine, Seoul, Korea. jeonguni01@hanmail.net
- KMID: 2175721
- DOI: http://doi.org/10.4048/jbc.2011.14.4.283
Abstract
- PURPOSE
A chemotherapy response assay test is performed to evaluate the degree of tumor growth inhibition by a chemotherapeutic agent. Several studies have been done on its usefulness; however, to the best of our knowledge, only a few studies concerning the relationship between chemotherapy response assay test results and breast cancer patients' prognoses have been conducted. Thus, we performed this study to analyze this relationship.
METHODS
Among breast cancer patients who underwent curative surgery and neoadjuvant or adjuvant chemotherapy between August 2004 and December 2009, 102 were enrolled in this study. Chemotherapeutic regimens for patients were doxorubicin plus taxane or doxorubicin plus cyclophosphamide followed by taxane. We divided these patients into two groups (sensitive group [n=19] and resistant group [n=83]) and analyzed the relationship between chemosensitivity results and patient prognosis.
RESULTS
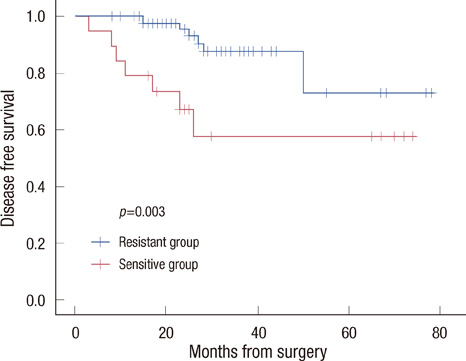
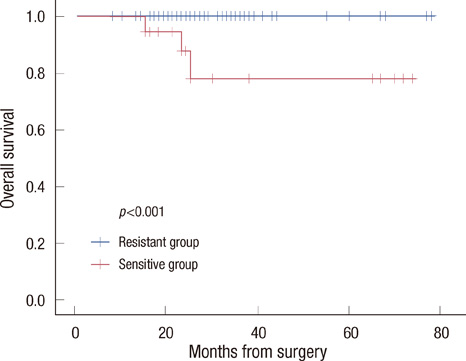
The sensitive group was associated with poor disease-free survival (DFS) (p=0.003) and overall survival (OS) (p<0.001). No significant differences were observed in tumor histology (p=0.548), tumor size (p=0.479), number of metastatic lymph nodes (p=0.326), histologic grade (p=0.077), or nuclear grade (p=0.216) between the two groups. However, in respect to molecular subtype, the HER2-positive type and triple negative breast cancer were more frequently observed in the sensitive group (p=0.001). In a univariate and multivariate analysis for DFS, doxorubicin sensitivity was significantly associated with a poor prognosis (p<0.05).
CONCLUSION
Better chemosensitivity results are associated with a poor prognosis in breast cancer patients who have undergone anthracycline- and taxane-based chemotherapy, however, examination of additional cases and the use of a longer study period are needed.
Keyword
MeSH Terms
Figure
Reference
-
1. Yi SY, Ahn JS, Uhm JE, Lim do H, Ji SH, Jun HJ, et al. Favorable response to doxorubicin combination chemotherapy does not yield good clinical outcome in patients with metastatic breast cancer with triple-negative phenotype. BMC Cancer. 2010. 10:527.
Article2. Keam B, Im SA, Kim HJ, Oh DY, Kim JH, Lee SH, et al. Prognostic impact of clinicopathologic parameters in stage II/III breast cancer treated with neoadjuvant docetaxel and doxorubicin chemotherapy: paradoxical features of the triple negative breast cancer. BMC Cancer. 2007. 7:203.
Article3. Saloustros E, Mavroudis D, Georgoulias V. Paclitaxel and docetaxel in the treatment of breast cancer. Expert Opin Pharmacother. 2008. 9:2603–2616.
Article4. Biesaga B, Niemiec J, Ziobro M, Wysocka J, Kruczak A. Prognostic potential of topoisomerase IIα and HER2 in a retrospective analysis of early advanced breast cancer patients treated with adjuvant anthracycline chemotherapy. Breast. 2011. 20:338–350.
Article5. Moretti E, Oakman C, Di Leo A. Predicting anthracycline benefit: have we made any progress? Curr Opin Oncol. 2009. 21:507–515.
Article6. Cheang MC, Voduc D, Bajdik C, Leung S, McKinney S, Chia SK, et al. Basal-like breast cancer defined by five biomarkers has superior prognostic value than triple-negative phenotype. Clin Cancer Res. 2008. 14:1368–1376.
Article7. Bidard FC, Matthieu MC, Chollet P, Raoefils I, Abrial C, Dômont J, et al. p53 status and efficacy of primary anthracyclines/alkylating agentbased regimen according to breast cancer molecular classes. Ann Oncol. 2008. 19:1261–1265.
Article8. Carey LA, Dees EC, Sawyer L, Gatti L, Moore DT, Collichio F, et al. The triple negative paradox: primary tumor chemosensitivity of breast cancer subtypes. Clin Cancer Res. 2007. 13:2329–2334.
Article9. Liedtke C, Mazouni C, Hess KR, André F, Tordai A, Mejia JA, et al. Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J Clin Oncol. 2008. 26:1275–1281.
Article10. Rouzier R, Perou CM, Symmans WF, Ibrahim N, Cristofanilli M, Anderson K, et al. Breast cancer molecular subtypes respond differently to preoperative chemotherapy. Clin Cancer Res. 2005. 11:5678–5685.
Article11. Koechli OR, Avner BP, Sevin BU, Avner B, Perras JP, Robinson DS, et al. Application of the adenosine triphosphate-cell viability assay in human breast cancer chemosensitivity testing: a report on the first results. J Surg Oncol. 1993. 54:119–125.
Article12. Kim HA, Yom CK, Moon BI, Choe KJ, Sung SH, Han WS, et al. The use of an in vitro adenosine triphosphate-based chemotherapy response assay to predict chemotherapeutic response in breast cancer. Breast. 2008. 17:19–26.
Article13. Choi SK, Jeong J, Lee SA, Hwang SH, Ahn SG, Jung WH, et al. Heterogeneous chemosensitivity of breast cancer determined by adeonsine triphosphate based chemotherapy response assay. J Breast Cancer. 2010. 13:180–186.
Article14. Sevin BU, Peng ZL, Perras JP, Ganjei P, Penalver M, Averette HE. Application of an ATP-bioluminescence assay in human tumor chemosensitivity testing. Gynecol Oncol. 1988. 31:191–204.
Article15. Andreotti PE, Cree IA, Kurbacher CM, Hartmann DM, Linder D, Harel G, et al. Chemosensitivity testing of human tumors using a microplate adenosine triphosphate luminescence assay: clinical correlation for cisplatin resistance of ovarian carcinoma. Cancer Res. 1995. 55:5276–5282.16. Kang SM, Park MS, Chang J, Kim SK, Kim H, Shin DH, et al. A feasibility study of adenosine triphosphate-based chemotherapy response assay (ATP-CRA) as a chemosensitivity test for lung cancer. Cancer Res Treat. 2005. 37:223–227.
Article17. Ahn SG, Jeong J, Choi SK, Hwang SH, Lee SA, Jung WH, et al. Correlation of early systemic recurrence with in vitro adenosine triphophate-based chemotherapy response assay in stage II and III breast cancer patients treated with doxorubicin-based chemotherapy. J Breast Cancer. 2011. 14:Suppl 1. S50–S56.18. Yamamoto Y, Iwase H. Clinicopathological features and treatment strategy for triple-negative breast cancer. Int J Clin Oncol. 2010. 15:341–351.
Article19. Freedman GM, Anderson PR, Li T, Nicolaou N. Locoregional recurrence of triple-negative breast cancer after breast-conserving surgery and radiation. Cancer. 2009. 115:946–951.
Article20. Konecny G, Crohns C, Pegram M, Felber M, Lude S, Kurbacher C, et al. Correlation of drug response with the ATP tumorchemosensitivity assay in primary FIGO stage III ovarian cancer. Gynecol Oncol. 2000. 77:258–263.
Article21. Kuerer HM, Sahin AA, Hunt KK, Newman LA, Breslin TM, Ames FC, et al. Incidence and impact of documented eradication of breast cancer axillary lymph node metastases before surgery in patients treated with neoadjuvant chemotherapy. Ann Surg. 1999. 230:72–78.
Article22. Rouzier R, Pusztai L, Delaloge S, Gonzalez-Angulo AM, Andre F, Hess KR, et al. Nomograms to predict pathologic complete response and metastasis-free survival after preoperative chemotherapy for breast cancer. J Clin Oncol. 2005. 23:8331–8339.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Treatment with Cisplatin and Etoposide Chemotherapy in Patient with Metastatic Breast Cancer
- Chemotherapy in Breast Cancer
- The evidence for adjuvant taxanes in early breast cancer
- Gemcitabine and Vinorelbine Combination Chemotherapy in Anthracycline- and Taxane-pretreated Advanced Breast Cancer
- Predictive Factors for Non-Response to Neoadjuvant Chemotherapy for Breast Cancer