J Periodontal Implant Sci.
2013 Feb;43(1):24-29. 10.5051/jpis.2013.43.1.24.
Correlation of expression and activity of matrix metalloproteinase-9 and -2 in human gingival cells of periodontitis patients
- Affiliations
-
- 1Department of Orthodontics, Kyung Hee University School of Dentistry, Seoul, Korea.
- 2Department of Biological Science, Molecular and Cellular Glycobiology Unit, Sungkyunkwan University, Suwon, Korea. chkimbio@skku.edu
- 3College of Art and Sciences, New York University, New York, NY, USA.
- 4Department of Orthodontics, Oral Biology Research Institute, Kyung Hee University School of Dentistry, Seoul, Korea. ygpark@khu.ac.kr
- KMID: 1783674
- DOI: http://doi.org/10.5051/jpis.2013.43.1.24
Abstract
- PURPOSE
Matrix metalloproteinases (MMPs) are capable of degrading extracellular matrix, and they are inducible enzymes depending on an inflammatory environment such as periodontitis and bacterial infection in periodontal tissue. Gingival inflammation has been postulated to be correlated with the production of MMP-2 and MMP-9. The objective of this study was to quantify the expression and activity of MMP-9 and -2, and to determine the correlation between activity and expression of these MMPs in human gingival tissues with periodontitis.
METHODS
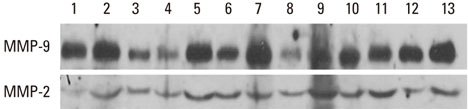
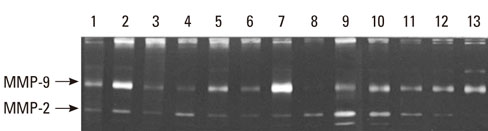
The gingival tissues of 13 patients were homogenized in 500 microL of phosphate buffered saline with a protease inhibitor cocktail. The expression and activity of MMP-2 and -9 were measured by enzyme-linked immunosorbent assay and Western blot analysis, and quantified by a densitometer. For the correlation line, statistical analysis was performed using the Systat software package.
RESULTS
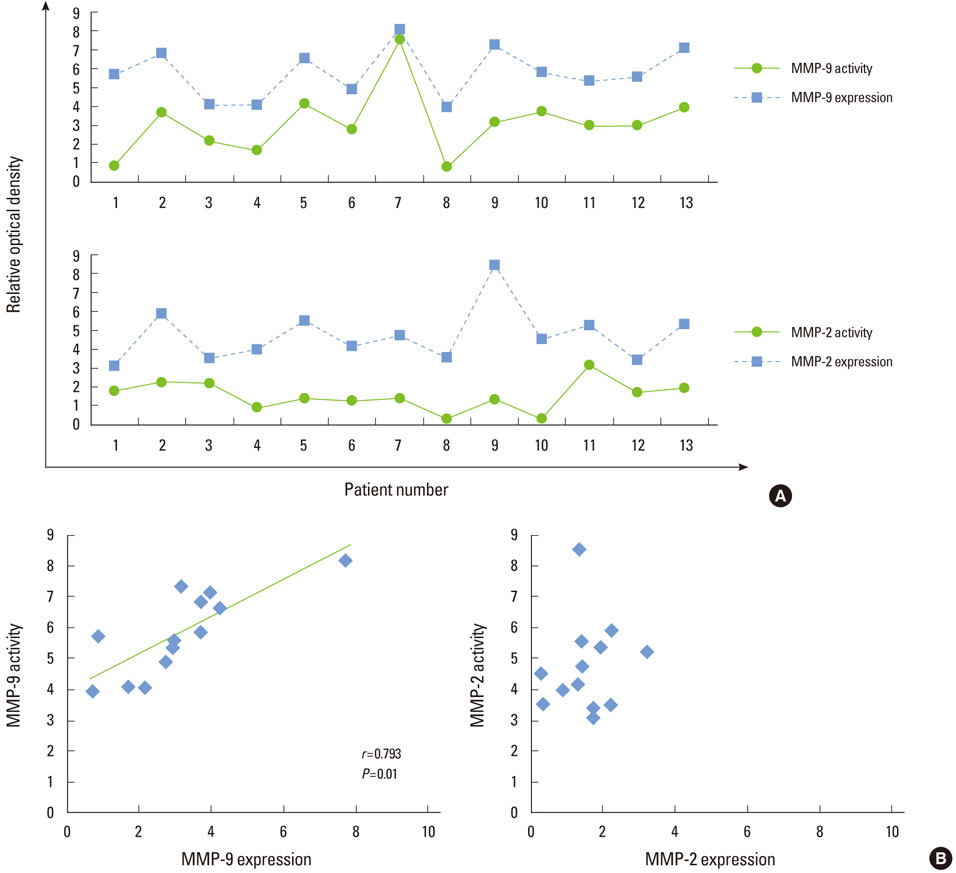
MMP-9 was highly expressed in all gingival tissue samples, whereas MMP-2 was underexpressed compared with MMP-9. MMP-9 activity increased together with the MMP-9 expression level, with a positive correlation (r=0.793, P=0.01). The correlation was not observed in MMP-2.
CONCLUSIONS
The expression of MMP-2 and -9 might contribute to periodontal physiological and pathological processes, and the degree of MMP-9 expression and activity are predictive indicators relevant to the progression of periodontitis.
MeSH Terms
Figure
Reference
-
1. Van Dyke TE, Serhan CN. Resolution of inflammation: a new paradigm for the pathogenesis of periodontal diseases. J Dent Res. 2003. 82:82–90.
Article2. DeCarlo AA Jr, Windsor LJ, Bodden MK, Harber GJ, Birkedal-Hansen B, Birkedal-Hansen H. Activation and novel processing of matrix metalloproteinases by a thiol-proteinase from the oral anaerobe Porphyromonas gingivalis. J Dent Res. 1997. 76:1260–1270.
Article3. Pozo P, Valenzuela MA, Melej C, Zaldivar M, Puente J, Martinez B, et al. Longitudinal analysis of metalloproteinases, tissue inhibitors of metalloproteinases and clinical parameters in gingival crevicular fluid from periodontitis-affected patients. J Periodontal Res. 2005. 40:199–207.
Article4. Vernal R, Dezerega A, Dutzan N, Chaparro A, Leon R, Chandia S, et al. RANKL in human periapical granuloma: possible involvement in periapical bone destruction. Oral Dis. 2006. 12:283–289.
Article5. Sorsa T, Tjaderhane L, Konttinen YT, Lauhio A, Salo T, Lee HM, et al. Matrix metalloproteinases: contribution to pathogenesis, diagnosis and treatment of periodontal inflammation. Ann Med. 2006. 38:306–321.
Article6. Birkedal-Hansen H. Role of cytokines and inflammatory mediators in tissue destruction. J Periodontal Res. 1993. 28(6 Pt 2):500–510.
Article7. Cazalis J, Tanabe S, Gagnon G, Sorsa T, Grenier D. Tetracyclines and chemically modified tetracycline-3 (CMT-3) modulate cytokine secretion by lipopolysaccharide-stimulated whole blood. Inflammation. 2009. 32:130–137.
Article8. Uitto VJ, Overall CM, McCulloch C. Proteolytic host cell enzymes in gingival crevice fluid. Periodontol 2000. 2003. 31:77–104.
Article9. Sorsa T, Tervahartiala T, Leppilahti J, Hernandez M, Gamonal J, Tuomainen AM, et al. Collagenase-2 (MMP-8) as a point-of-care biomarker in periodontitis and cardiovascular diseases. Therapeutic response to non-antimicrobial properties of tetracyclines. Pharmacol Res. 2011. 63:108–113.
Article10. Cox SW, Eley BM, Kiili M, Asikainen A, Tervahartiala T, Sorsa T. Collagen degradation by interleukin-1beta-stimulated gingival fibroblasts is accompanied by release and activation of multiple matrix metalloproteinases and cysteine proteinases. Oral Dis. 2006. 12:34–40.
Article11. Reynolds JJ, Meikle MC. Mechanisms of connective tissue matrix destruction in periodontitis. Periodontol 2000. 1997. 14:144–157.
Article12. Ryan ME, Golub LM. Modulation of matrix metalloproteinase activities in periodontitis as a treatment strategy. Periodontol 2000. 2000. 24:226–238.
Article13. Sorsa T, Tjaderhane L, Salo T. Matrix metalloproteinases (MMPs) in oral diseases. Oral Dis. 2004. 10:311–318.
Article14. Ingman T, Sorsa T, Michaelis J, Konttinen YT. Matrix metalloproteinases-1, -3, and -8 in adult periodontitis in situ. An immunohistochemical study. Ann N Y Acad Sci. 1994. 732:459–461.15. Corotti MV, Zambuzzi WF, Paiva KB, Menezes R, Pinto LC, Lara VS, et al. Immunolocalization of matrix metalloproteinases-2 and -9 during apical periodontitis development. Arch Oral Biol. 2009. 54:764–771.
Article16. Tsagareli ZG, Shishniashvili TE, Gogiashvili LE, Kvachadze TI, Khimshiashvili NB. The level of matrix metalloproteinases and type IV collagen in the gingival mucosa under different clinical forms of periodontitis in pre- and pubertal periods and their prognostic value. Georgian Med News. 2012. (206):25–29.17. Rai B, Kharb S, Jain R, Anand SC. Biomarkers of periodontitis in oral fluids. J Oral Sci. 2008. 50:53–56.
Article18. Nishikawa M, Yamaguchi Y, Yoshitake K, Saeki Y. Effects of TNFalpha and prostaglandin E2 on the expression of MMPs in human periodontal ligament fibroblasts. J Periodontal Res. 2002. 37:167–176.
Article19. Reynolds JJ, Hembry RM, Meikle MC. Connective tissue degradation in health and periodontal disease and the roles of matrix metalloproteinases and their natural inhibitors. Adv Dent Res. 1994. 8:312–319.
Article20. Emingil G, Kuula H, Sorsa T, Atilla G. Gingival crevicular fluid matrix metalloproteinase-25 and -26 levels in periodontal disease. J Periodontol. 2006. 77:664–671.
Article21. Tervahartiala T, Pirila E, Ceponis A, Maisi P, Salo T, Tuter G, et al. The in vivo expression of the collagenolytic matrix metalloproteinases (MMP-2, -8, -13, and -14) and matrilysin (MMP-7) in adult and localized juvenile periodontitis. J Dent Res. 2000. 79:1969–1977.
Article22. Aiba T, Akeno N, Kawane T, Okamoto H, Horiuchi N. Matrix metalloproteinases-1 and -8 and TIMP-1 mRNA levels in normal and diseased human gingivae. Eur J Oral Sci. 1996. 104:562–569.
Article23. Shapiro SD, Senior RM. Matrix metalloproteinases. Matrix degradation and more. Am J Respir Cell Mol Biol. 1999. 20:1100–1102.24. Márton IJ, Kiss C. Protective and destructive immune reactions in apical periodontitis. Oral Microbiol Immunol. 2000. 15:139–150.
Article25. Kesanakurti D, Chetty C, Bhoopathi P, Lakka SS, Gorantla B, Tsung AJ, et al. Suppression of MMP-2 attenuates TNF-α induced NF-κB activation and leads to JNK mediated cell death in glioma. PLoS One. 2011. 6:e19341.
Article26. Maeso G, Bravo M, Bascones A. Levels of metalloproteinase-2 and -9 and tissue inhibitor of matrix metalloproteinase-1 in gingival crevicular fluid of patients with periodontitis, gingivitis, and healthy gingiva. Quintessence Int. 2007. 38:247–252.27. Murphy G, Docherty AJ, Hembry RM, Reynolds JJ. Metalloproteinases and tissue damage. Br J Rheumatol. 1991. 30:Suppl 1. 25–31.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The effect of periodontal flap surgery on Matrix metalloproteinases (MMPs) and Tissue inhibitors of matrix metalloproteinase-1 (TIMP-1) levels in gingival crevicular fluids of periodontitis patients
- Expression of mRNA for matrix metalloproteinases and tissue inhibitors of metalloproteinases in human gingival and periodontal ligament fibroblasts treated with lipopolysaccharide from Prevotella intermedia
- The expressions of inflammatory factors and tissue inhibitor of matrix metalloproteinase-2 in human chronic periodontitis with type 2 diabetes mellitus
- Expression of Matrix metalloproteinase-1 between Simple Chronic Periodontitis and Type 2 Diabetes associated Chronic Periodontitis on Protein level
- The influence of type 2 diabetes mellitus on the expression of inflammatory mediators and tissue inhibitor of metalloproteinases-2 in human chronic periodontitis